After a new drug entity enters the pharmaceutical marketplace, it is always wise to keep abreast of the current literature documenting postmarketing randomized clinical trials (RCTs) and surveillance reports, in addition to case studies, so that the clinical experience of others, including evidence-based regimens, may be considered when making recommendations for, and monitoring patients receiving, a particular pharmaceutical. Ongoing, lifelong continuing education is not only helpful in practice with regard to therapeutic outcomes and patient safety, but also imperative from a risk-management point of view, for both the individual practitioner and the health care organization. This column focuses on postmarketing evidence and current clinical considerations regarding rasagiline for treatment of Parkinson’s disease (PD), particularly as compared with selegiline, another monoamine oxidase type-B (MAO-B) inhibitor used in the treatment of PD.
Rasagiline
In 2006, the FDA approved rasagiline (Azilect) for the treatment of PD (TABLE 1), a chronic, progressive neurodegenerative condition caused by the destruction of brain cells that produce dopamine.1 Owing to this neuronal loss, slowing of neurotransmission renders patients incapable of initiating and controlling normal body movements, a state characteristically manifesting as resting tremor, rigidity, bradykinesia, and postural instability; these symptoms are often described by patients and caregivers as shaking, stiff muscles, slow movements, and falling. PD affects about 0.4% of people over 40 years, 1% of people 65 years or older, and 10% of people 80 years or older; the mean age at onset is about 57 years.2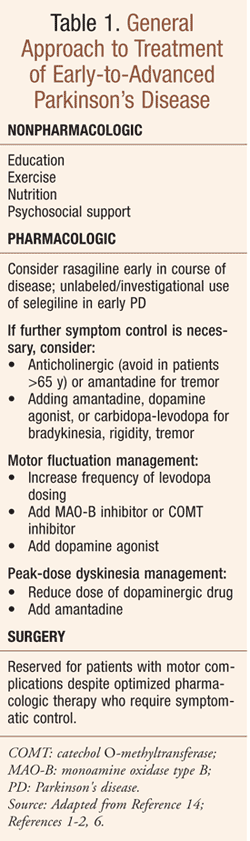
A new molecular entity when it was introduced, rasagaline had shown the ability to block the breakdown of dopamine as an irreversible and selective second-generation MAO-B inhibitor.1,3 Compared with selegiline (Eldepryl), the only other MAO-B inhibitor for PD, rasagiline is five times more potent; unlike selegiline, rasagiline is not metabolized to an amphetamine-like substance, so that insomnia is not an issue as with selegiline oral tablet or capsule (insomnia 12%) and orally disintegrating tablets (insomnia 7%).3-6 Further, while selegiline has demonstrated neuroprotective properties in experimental laboratory models, the amphetamine metabolites may interfere with or neutralize such neuroprotective effects.7 Rasagiline was proposed to have a neuroprotective effect similar to that of selegiline, which was introduced in 1996 (see below, Clinical Controversy).8
Evidence Supports Safety and Efficacy
As with selegiline, PD symptoms are mildly improved with rasagiline.9-12 Early clinical studies have shown that rasagiline is associated with improved outcomes in patients with early PD, in addition to its ability to reduce “off” time (periods of the day when the drug is not working optimally, causing worsening of parkinsonian symptoms) in patients with moderate-to-advanced PD with motor fluctuations.3 Compared with patients on placebo, PD patients on rasagiline showed significantly less worsening of their condition on a rating scale that measures the ability to perform mental and motor tasks as well as daily living activities.1 Two other studies compared rasagiline effects with placebo when taken concomitantly with levodopa in patients with more advanced PD; patients using rasagiline and concomitant levodopa had significantly less time per day with relatively poor function and mobility as compared with patients on levodopa and placebo.1 Of note, the efficacy of rasagiline for the management of patients with motor fluctuations appears similar to that of the catechol O-methyltransferase (COMT) inhibitor entacapone; both agents are considered first-line interventions for motor fluctuations.11,13 Rasagiline is FDA approved for initial monotherapy in idiopathic early PD and as an adjunct to levodopa in advanced disease. 1,3,6
Rasagiline Therapy: Early in Course of Disease
For monotherapy in idiopathic PD, rasagiline 1 mg once daily is recommended in both geriatric and adult patients. As an adjunct with levodopa, 0.5 mg once daily is the initial recommended dose, increased to 1 mg once daily based on response and tolerability.5-6,14 No dosage adjustment is necessary in mild renal impairment; dosage adjustment is not defined in moderate-to-severe renal impairment.5 In mild hepatic impairment, rasagiline is dosed 0.5 mg once daily; use is not recommended in moderate-to-severe hepatic impairment. Owing to the potential risk of hypertensive crisis, concomitant use of foods high in tyramine (>150 mg) should be avoided.5,6 Rasagiline should be tapered to discontinuation to avoid abrupt withdrawal.5
As with the majority of agents for Parkinson’s disease (TABLE 2), the potential to cause dyskinesias, hallucinations, and orthostatic hypotension (particularly with levodopa) may occur with rasagiline as well.1,6 Orthostasis is usually most problematic during the first 2 months of therapy. While rasagiline appears to be well tolerated, elderly patients may be more prone to treatment-emergent adverse cardiovascular and psychiatric effects.3 Of note, rasagiline is considered a hazardous agent so that appropriate precautions for handling and disposal are required.6

Clinical Controversy
While it is commonly inferred that MAO-B inhibitors have disease-modifying effects, much debate and scientific interest continue in this regard.12,14 In a large, placebo-controlled, 18-month, delayed-start clinical trial, the initiation of rasagiline monotherapy early in idiopathic PD was associated with less functional decline than when treatment was delayed for 9 months.15 Better long-term outcomes were attributed to a disease-modifying effect (as opposed to a symptomatic effect).14 It is not known if this is a class effect of the MAO-B inhibitors; however, currently there are no treatments clinically proven to arrest or delay the neuronal loss associated with PD.12
Melanoma Risk: Awaiting Study Data
During rasagiline’s development, melanoma was diagnosed in a small number of patients treated with this drug. While the FDA has concluded that data available to date do not establish that the drug is the causative agent, it appears that compared to the general population, patients with Parkinson’s disease have an increased risk for this form of skin cancer.1,6 Data from the postmarket phase IV study (see RESOURCES) are awaited to address this question. Product labeling recommends that patients undergo periodic dermatologic examinations; bruising (2%), alopecia, skin carcinoma, and vesiculobullous rash have been reported in patients receiving rasagiline.1,6 Of note, seborrheic dermatitis is common in patients with PD.2

Beneficial Addition to the Formulary
The management of PD is likely to continue to change as more information is obtained regarding the pathoetiology of PD and new therapeutic interventions.12 Until then, Chen and Ly suggest that rasagiline may be a beneficial addition to the formulary for monotherapy for early PD or as an alternative for patients with a history of intolerance to selegiline.3 These researchers propose rasagaline as an addition to the formulary based on the following reasons:
• Rasagiline has demonstrated clinical efficacy as monotherapy in patients in early PD and was associated with improved quality of life.16
• Rasagiline has shown beneficial effects as an adjunct to levodopa in patients with moderate-to-advanced disease and motor fluctuations.10,11
• Rasagiline and entacapone have both shown similar reductions in off-time hours compared with placebo as an adjunct to levodopa with or without additional antiparkinsonian drugs (e.g., amantadine, dopamine agonists) in patients with moderate-to-advanced disease and motor fluctuations.11
• Unlike selegiline, which is metabolized to amphetamine derivatives, rasagiline is biotransformed to the nonamphetamine compound aminoindan. Safety and tolerability data show rasagiline appearing to be well tolerated, with infrequent adverse cardiovascular or psychiatric effects.3
• The long-term (more than one year) data suggest that rasagiline continues to provide a benefit (e.g., less symptom progression, slower functional decline) over several years, although the data are limited.17
Summary
Pharmacists need to be confident in recommending one agent over another in the management of PD. While results of a phase IV postmarket study are pending and in the absence of head-to-head comparator trials, no firm conclusions can be drawn regarding the superiority of rasagiline when compared with selegiline, dopamine agonists, carbidopa-levodopa, COMT inhibitors, or other agents used in PD.12 Pharmacists need to consider and rely on current clinical factors until additional data are available. Long-term clinical trials are necessary in order to determine whether or not neuroprotective or disease-modifying effects of rasagiline observed in preclinical experiments will translate into clinically meaningful results.12REFERENCES
1. FDA approves new treatment for Parkinson’s disease.
News & Events. U.S. Food and Drug Administration. Updated April 8,
2013.
www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/2006/ucm108654.htm.
Accessed August 26, 2013.
2. Parkinson disease. Merck Manual online. Updated
January 2013.
www.merckmanuals.com/professional/neurologic_disorders/movement_and_cerebellar_disorders/parkinson_disease.html.
Accessed September 1, 2013.
3. Chen JJ, Ly AV. Rasagiline: a second-generation monoamine oxidase Type-B inhibitor for the treatment of Parkinson’s disease. Am J Health Syst Pharm. 2006;63(10):915-928.
4.
Clark MA, Finkel R, Rey JA, eds. Pharmacology. 5th ed. Baltimore, MD: Lippincott Williams & Wilkins; 2012:99,104-105.
5. Epocrates Essentials Version 13.7. www.epocrates.com. Accessed September 2, 2013.
6. Semla TP, Beizer JL, Higbee MD. Geriatric Dosage Handbook. 18th ed. Hudson, OH: Lexi-Comp, Inc; 2013:1591-1594,1995-1996.
7. Bar-Am O, Amit T, Youdim MB. Contrasting
neuroprotective and neurotoxic actions of respective metabolites of
anti-Parkinson drugs rasagiline and selegiline. Neurosci Lett. 2004;355:169-172.
8. Olanow CW, Hauser RA, Jankovic J, et al. A randomized,
double-blind, placebo-controlled, delayed start study to assess
rasagiline as a disease modifying therapy in Parkinson’s disease (the
ADAGIO study): rationale, design, and baseline characteristics. Mov Disord. 2008;23:2194-2201.
9. Parkinson Study Group. A controlled trial of rasagiline in early Parkinson disease: the TEMPO Study. Arch Neurol. 2002;59:1937-1943.
10. Parkinson Study Group. A randomized placebo-controlled
trial of rasagiline in levodopa-treated patients with Parkinson disease
and motor fluctuations: the PRESTO study. Arch Neurol. 2005;62:241-248.
11. Rascol O, Brooks DJ, Melamed E, et al. Rasagiline as
an adjunct to levodopa in patients with Parkinson’s disease and motor
fluctuations (LARGO, Lasting effect in Adjunct therapy with Rasagiline
Given Once daily, study): a randomised, double-blind, parallel-group
trial. Lancet. 2005;365:947-954.
12. Ahlskog JE, Uitti RJ. Rasagiline, Parkinson neuroprotection, and delayed-start trials: still no satisfaction? Neurology. 2010;74(14):1143-1148.
www.ncbi.nlm.nih.gov/pubmed/20368634. Accessed August 26, 2013.
13. Pahwa R, Factor SA, Lyons KE, et al. Quality Standards
Subcommittee of the American Academy of Neurology. Practice parameter:
treatment of Parkinson’s disease with motor fluctuations and dyskinesia
(an evidence-based review): report of the Quality Standards Subcommittee
of the American Academy of Neurology. Neurology. 2006;66:983-995.
14. Chen JJ, Nelson MV, Swope DM. Parkinson’s disease. In: DiPiro JT, Talbert RL, Yee GC, et al, eds. Pharmacotherapy: A Pathophysiologic Approach. 8th ed. New York, NY: McGraw-Hill Inc; 2011:1033-1044.
15. Olanow CW, Rascol O, Hauser R. ADAGIO Study
Investigators. A double-blind, delayed-start trial of rasagiline in
Parkinson’s disease. N Engl J Med. 2009;361:1268-1278.
16. Parkinson Study Group. A controlled, randomized, delayed-start study of rasagiline in early Parkinson disease. Arch Neurol. 2004; 61:561-566.
17. Hauser RA, Lew MF, Hurtig HI, et al. Early treatment
with rasagiline is more beneficial than delayed treatment start in the
long-term management of Parkinson’s disease. Mov Disord. 2005;20(suppl 10):S75.
To comment on this article, contact rdavidson@uspharmacist.com





