US Pharm. 2014;39(4):44-46.
ABSTRACT: Anaphylaxis is a serious, potentially fatal medical emergency that requires prompt treatment with epinephrine. Patients at risk for anaphylaxis are commonly prescribed an epinephrine auto-injector (EAI). The FDA has assigned BX ratings to EAIs, indicating that sufficient data are not available to determine therapeutic equivalence. It is important that pharmacists understand that patients will have received training from their physician on the proper use of the EAI they have been prescribed. As each of the marketed EAIs differ in appearance, design, and administration, dispensing a different EAI than prescribed could possibly lead to confusion by users during an anaphylactic event.
Anaphylaxis is a severe, potentially fatal allergic reaction that can occur rapidly after exposure to a specific allergen.1 Typically affecting two or more organ systems (e.g., skin, respiratory, gastro-intestinal), anaphylaxis is characterized clinically by symptoms that may include flushing, itching, swelling, tightness of the throat and chest, and abdominal pain.2
Anaphylaxis Overview
Triggers for anaphylaxis include exposure to specific foods (e.g., peanuts, milk, shellfish), drugs (e.g., beta-lactam antibiotics), biologicals (e.g., monoclonal antibodies), insect stings (e.g., bees, wasps, ants), and latex.2 Exercise and other physical factors (e.g., cold air) have also been associated with anaphylaxis.2
The lifetime prevalence of anaphylaxis is estimated to be between 0.05% and 2%, but it is thought to be greater because of the level of underreporting that occurs.3 In the United States, between 1.2% and 16.8% of the population may experience anaphylaxis during their lifetime, resulting in death in 0.002% of patients.4 In children and adolescents, estimates for the incidence of anaphylaxis range from 0.2 to 10.5 episodes per 100,000 person-years.5,6 However, these numbers may not reflect the current burden of anaphylaxis, as rates of food allergy in children tripled between 1997 and 2007.7 Furthermore, prevalence of severe food allergy in children has been estimated at 3.1%.8 Guidelines recommend that patients experiencing anaphylaxis receive prompt treatment with intramuscular (IM) epinephrine as the preferred route of administration.1,9 Fatalities have occurred in patients who either never received treatment with epinephrine or were not treated in a timely fashion.10,11
Epinephrine Auto-Injectors
Patients at risk for an acute episode of anaphylaxis are commonly prescribed epinephrine auto-injectors (EAIs), which are used to administer epinephrine by IM injection into the anterolateral thigh.2 There are currently four EAIs marketed in the U.S. (see TABLE 1): EpiPen/EpiPen Jr (Mylan Specialty LP, Basking Ridge, NJ); Auvi-Q (sanofi-aventis US LLC, Bridgewater, NJ); Adrenaclick (Amedra Pharmaceuticals LLC, Horsham, PA); and Epinephrine injection, USP auto-injector (Lineage Therapeutics, Horsham, PA), the authorized generic to Adrenaclick.12-15 Each of the EAIs administers a single dose of either 0.3 or 0.15 mg of epinephrine. The 0.3-mg dose is intended for patients who weigh 30 kg or more, and the 0.15-mg dose is intended for patients who weigh 15 to 30 kg.12-15
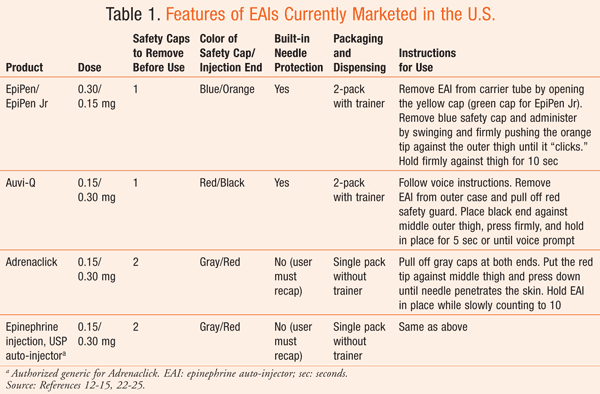
Some of the EAIs currently marketed in the U.S. are dispensed as a two-pack rather than as a single EAI.12,13 This is an important distinguishing feature, as 16% to 36% of patients are estimated to require more than 1 dose of epinephrine during an anaphylactic episode.16,17 The need for more than 1 dose of epinephrine has been shown to be directly associated with the severity of the anaphylactic reaction.17
Epinephrine does not have any absolute contraindications when used in a life-threatening situation.12-15 However, administration of a therapeutic dose of epinephrine may cause transient, systemic adverse reactions in some patients. These include moderate anxiety, apprehensiveness, difficulty breathing, restlessness, dizziness, headache, nausea and vomiting, palpitations, pallor, sweating, tremor, and weakness. Also, potentially fatal arrhythmias have been reported in patients with underlying cardiac disorders. Finally, rapid increases in blood pressure have produced cerebral hemorrhage, and angina may occur in patients with coronary artery disease.12-15
BX Ratings as They Apply to EAIs
The FDA has assigned EAIs a BX rating, the Orange Book code given to drug products for which data reviewed are insufficient to make a determination of therapeutic equivalence to other pharmaceutically equivalent products.18 Drug products are considered to be therapeutically equivalent when there are no known or suspected problems with bioequivalence or when potential problems have been resolved by in vivo and/or in vitro evidence supporting bioequivalence.18 Only products that have been assigned an “A” code are considered therapeutically equivalent and, therefore, are the only products expected to produce the same clinical effect and safety profile as the prescribed product.18
Under specific conditions, pharmacists in 21 states can substitute a drug product despite a BX rating.19 These conditions include meeting the state’s definition of an equivalent drug product; substituting a therapeutically equivalent drug product in the economic interest of the patient; deeming a product to be therapeutically equivalent on the basis of the pharmacist’s professional judgment; having a drug listed in the state’s or community pharmacy’s formulary of generic and brand name products; substituting a drug product when requested by a prescriber or purchaser; and substituting a drug product that does not meet adequate standards on the basis of the pharmacist’s professional judgment.19
Epinephrine auto-injectors differ not only in appearance but also in design and method of administration (TABLE 1). In addition, each EAI has different design features that are continually examined (e.g., change in color of safety cap, addition of yellow arrow pointing to black injection tip) in order to evaluate which minor alterations may enhance correct usage during an emergency.20 Patients experiencing anaphylaxis require immediate administration of epinephrine,1,9 so proper training and education on the use of the specific EAI prescribed to a patient by his or her physician is critical to enable immediate use during an anaphylactic episode.
Patients and caregivers who are familiar with a given EAI are at increased risk for confusion during an anaphylactic episode if they are switched to another EAI without receiving appropriate training on how to administer the product.21 For example, some EAIs require the patient or caregiver to remove two caps before use, while other EAIs require the removal of only one cap. These differences may lead patients who are familiar with a one-cap EAI and are then switched to a two-cap EAI to potentially be confused regarding proper injection during an anaphylactic episode.21 Additionally, patients and caregivers may be exposed to accidental needle sticks if the caps are removed out of order.21 Similarly, patients and caregivers familiar with EAIs that provide needle protection after use may be exposed to accidental needle sticks when switched to an EAI that requires recapping.
Different EAIs have specific techniques for administration; for example, some EAIs use a “swing and firmly push” method, while others employ a “press and hold” method.22,23 Considering emergency use, BX ratings, differences between EAIs, and the potential for confusion between marketed products, it is important that pharmacists not exchange a prescribed EAI for another marketed EAI without discussing this with the patient’s physician first. Patients would also require training on the new device.
Conclusion
EAIs are the current recommended first-line treatment for patients at risk for anaphylaxis, and it is critical that patients be properly trained and familiar with the device on which they may need to rely during an emergency. Pharmacists should have an understanding of BX ratings in the context of the products they may dispense to patients and be aware of the potential consequences of sending patients or caregivers home with an EAI for which they have not received adequate training.
REFERENCES
1. Sampson HA, Muñoz-Furlong A, Campbell RL, et al. Second
symposium on the definition and management of anaphylaxis: summary
report—second National Institute of Allergy and Infectious Disease/Food
Allergy and Anaphylaxis Network symposium. Ann Emerg Med. 2006;47:373-380.
2. Simons FE, Sheikh A. Anaphylaxis: the acute episode and beyond. BMJ. 2013;346:f602.
3. Lieberman P, Camargo CA Jr, Bohlke K, et al.
Epidemiology of anaphylaxis: findings of the American College of
Allergy, Asthma and Immunology Epidemiology of Anaphylaxis Working
Group. Ann Allergy Asthma Immunol. 2006;97:596-602.
4. Neugut AI, Ghatak AT, Miller RL. Anaphylaxis in the United States: an investigation into its epidemiology. Arch Intern Med. 2001;161:15-21.
5. Bohlke K, Davis RL, DeStefano F, et al. Epidemiology of
anaphylaxis among children and adolescents enrolled in a health
maintenance organization. J Allergy Clin Immunol. 2004;113:536-542.
6. Macdougall CF, Cant AJ, Colver AF. How dangerous is
food allergy in childhood? The incidence of severe and fatal allergic
reactions across the UK and Ireland. Arch Dis Child. 2002;86:236-239.
7. Branum AM, Lukacs SL. Food allergy among children in the United States. Pediatrics. 2009;124:1549-1555.
8. Gupta RS, Springston EE, Warrier MR, et al. The
prevalence, severity, and distribution of childhood food allergy in the
United States. Pediatrics. 2011;128:e9-e17.
9. Simons FE, Ardusso LR, Bilò MB, et al. 2012 update:
World Allergy Organization guidelines for the assessment and management
of anaphylaxis. Curr Opin Allergy Clin Immunol. 2012;12:389-399.
10. Bock SA, Muñoz-Furlong A, Sampson HA. Fatalities due to anaphylactic reactions to foods. J Allergy Clin Immunol. 2001;107:191-193.
11. Bock SA, Muñoz-Furlong A, Sampson HA. Further fatalities caused by anaphylactic reactions to food, 2001-2006. J Allergy Clin Immunol. 2007;119:1016-1018.
12. EpiPen/EpiPen Jr (epinephrine auto-injectors 0.3/0.15 mg) package insert. Basking Ridge, NJ: Mylan Specialty LP; 2012.
13. Auvi-Q (epinephrine injection, USP) package insert. Bridgewater, NJ: sanofi-aventis US LLC; 2012.
14. Adrenaclick (epinephrine injection, USP auto-injector) package insert. Horsham, PA: Amedra Pharmaceuticals LLC; 2013.
15. Epinephrine injection, USP auto-injector package insert. Horsham, PA: Lineage Therapeutics Inc; 2013.
16. Kelso JM. A second dose of epinephrine for anaphylaxis: how often needed and how to carry. J Allergy Clin Immunol. 2006;117:464-465.
17. Korenblat P, Lundie MJ, Dankner RE, Day JH. A
retrospective study of epinephrine administration for anaphylaxis: how
many doses are needed? Allergy Asthma Proc. 1999;20:383-386.
18. FDA Center for Drug Evaluation and Research. Approved Drug Products With Therapeutic Equivalence Evaluations. 33rd ed. Silver Spring, MD: US Department of Health and Human Services; 2013.
19. State regulations on generic substitution. Pharm/Prescrib Lett. 2006;22:220901.
20. Bakirtas A, Arga M, Catal F, et al. Make-up of the epinephrine autoinjector: the effect on its use by untrained users. Pediatr Allergy Immunol. 2011;22:729-733.
21. Ram FSF, Hoare K, Arroll B, Hoare S. Epinephrine
self-administration in anaphylactic emergencies: comparison of commonly
available autoinjectors. J Asthma Allergy Educators. 2012;3:178-181
22. EpiPen/EpiPen Jr (epinephrine auto-injectors 0.3/0.15 mg) patient information. Basking Ridge, NJ: Mylan Specialty LP; 2012.
23. Auvi-Q (epinephrine injection, USP) trainer instructions. Bridgewater, NJ: sanofi-aventis US LLC; 2012.
24. Adrenaclick (epinephrine injection, USP auto-injector) patient information. Horsham, PA: Amedra Pharmaceuticals LLC; 2013.
25. Epinephrine injection, USP auto-injector patient information. Horsham, PA: Lineage Therapeutics Inc; 2013.
To comment on this article, contact rdavidson@uspharmacist.com.






