US Pharm. 2014;39(7):19-26.
ABSTRACT: Seasonal influenza is a substantial cause of morbidity and mortality worldwide. Prevention of infection is key, and influenza vaccination is recommended for all individuals aged 6 months and older. When used early and appropriately, antiviral agents can be helpful in decreasing disease burden. Pharmacists play a crucial role in optimizing prevention through advocating and delivering influenza immunizations and recommending antiviral therapy when indicated.
Seasonal influenza causes significant respiratory illness, resulting in approximately 25,000 deaths in the United States each year. Influenza is characterized by fever that is often abrupt in onset, myalgia, sore throat, headache, and nonproductive cough.1 While symptoms are usually self-limiting and resolve in a few days, complications—including secondary bacterial infections such as pneumonia—can be severe. This article will discuss the use of vaccination strategies for prevention and antivirals for treatment.
History
Knowledge of the natural history of influenza is important for understanding the ongoing significance of the influenza vaccine. Influenza is an RNA virus with three antigen types: A, B, and C. Most clinical illness is caused by types A and B. Influenza A is also associated with subtypes based on two surface antigens: hemagglutinin, which is responsible for viral attachment to cells, and neuraminidase, which is responsible for viral penetration into cells. The genetic change of these surface antigens over time leads to antigenic shifts and drifts that, if transmitted from person to person, can result in a pandemic or epidemic, respectively. Influenza epidemics and pandemics that resulted in millions of deaths and caused widespread respiratory illness have been documented as far back as the 1500s. The last major pandemic seen in the U.S. was the novel influenza A (H1N1) virus in 2009.2
Influenza is typically most prevalent during the fall and winter months.2 The CDC publishes weekly surveillance data indicating the prevalence of influenza illness across the U.S., as well as additional information on influenza-related deaths, hospitalizations, antigenic characterization, and antiviral resistance patterns.3
Influenza Vaccine
Immunization against influenza infection is paramount in reducing disease burden. Currently, there are three types of influenza vaccine available in the U.S., including trivalent and quadrivalent inactivated influenza vaccines (IIVs) and live attenuated influenza vaccine (LAIV) ( TABLE 1 ).
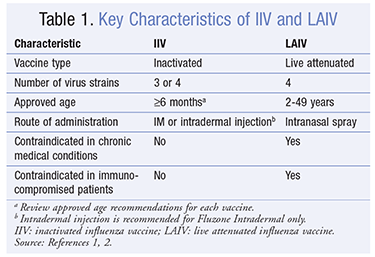
The trivalent inactivated influenza vaccine (TIV) first became available in the 1940s.1 The vaccine contains three inactivated viruses (two type A and one type B), one or more strains of which are replaced each year to provide protection based on surveillance of influenza disease and anticipated activity.2 Several pharmaceutical manufacturers are responsible for developing and preparing TIV. Inactivated virus has historically been grown in chicken eggs; however, new recombinant technology has enabled the development of a vaccine (RIV) that is egg-free, an option for those with anaphylaxis to egg proteins. In 2013, a new quadrivalent vaccine was marketed for the first time in the U.S. that confers broader protection by covering an additional influenza B virus strain.2,4
New preparations of IIV are available, including a high-dose formulation for persons aged >65 years and an intradermal vaccine. Currently, no preference for these agents has been indicated by the Advisory Committee on Immunization Practices.1 Immunizers should observe dosing and administration recommendations and age restrictions for individual vaccines.
Dosing of IIV in adults is 0.5 mL given as an IM injection once yearly during influenza season; pediatric dosing is 0.25 mL IM in children aged 6 to 35 months. When receiving the influenza vaccine for the first time, children aged 6 months through 8 years should receive a second dose 1 month after the first dose is administered to help elicit an appropriate immune response.1,2
In otherwise healthy individuals aged <65 years, TIV is approximately 90% effective in preventing illness.1,2 Although efficacy significantly declines with advancing age, patients aged >65 years still receive benefit through prevention of hospitalizations and death.
Another option for immunization of healthy, nonpregnant individuals aged 2 to 49 years is LAIV. LAIV is administered as a single-dose nasal spray, with half of the dose delivered into each nostril. Replication of the virus in the nasopharyngeal mucosa is required for the vaccine to be effective. Studies in children have shown improved efficacy of LAIV over TIV, but evidence supporting improved efficacy in adults is limited.1,2 Quadrivalent LAIV is available in the U.S. and replaced the trivalent formulation during the 2013–2014 season.
Contraindications to the influenza vaccine are few. Patients with a history of severe allergic reaction to egg protein, other vaccine components, or a prior dose of any influenza vaccine should not receive IIV or LAIV. In addition to age restrictions, other contraindications to LAIV include asthma or other chronic pulmonary disorders; cardiovascular, renal, hepatic, neurologic/neuromuscular, hematologic, or metabolic disorders; immunosuppression; and pregnancy. Caution should be exercised in administering influenza vaccine to patients with moderate-to-severe illness with or without fever. In patients with a history of Guillain-Barré syndrome within 6 weeks of a prior dose of influenza vaccine, caution should be used when administering subsequent doses.2
Research is ongoing for new technologies to improve vaccine efficacy and delivery. Other non–egg-based technology using cell cultures and recombinant DNA is in development. Adjuvant vaccine to augment immune response is currently being investigated.4 Studies involving new delivery systems, including self-vaccination using microneedle patches, show promise for acceptability and use.5
Antiviral Medications
Although vaccination is the preferred method for prevention of illness caused by the influenza virus, antivirals can play an adjunctive role in prophylaxis. Antivirals are also used to treat both uncomplicated and complicated illness.6 Currently, two antiviral classes are FDA-approved for influenza prophylaxis and treatment: adamantanes and neuraminidase inhibitors. The adamantanes include amantadine and rimantadine. Although the mechanism of action is not fully elucidated, the adamantanes appear predominantly to block the uncoating of the viral RNA within infected host cells and prevent its replication by interfering with the function of the transmembrane domain of the viral M2 protein.7 The neuraminidase inhibitors include oseltamivir (Tamiflu) and zanamivir ( Relenza), and the antiviral mechanism of action is through inhibition of influenza virus neuraminidase affecting viral shedding within the respiratory tract.8,9
Antiviral Resistance
Although both classes of antivirals carry indications for prophylaxis and treatment of influenza, adamantanes carry high (>99%) levels of resistance to influenza A(H1N1) and A(H3N2) and are not active against influenza B.6 Accordingly, the CDC states that these agents are considered resistant to the current circulating viruses for the 2013–2014 season, and it does not recommend their use in treatment and prophylaxis.6 Based on current resistance data, it is assumed that this will also be the case for the 2014–2015 influenza season. Therefore, the adamantanes will not be discussed in this article.
According to CDC surveillance data, at the close of the 2013–2014 influenza season, the neuraminidase inhibitors oseltamivir and zanamivir are reported to have a resistance rate of 1.2% and 0%, respectively.3 An annual review of surveillance data during the influenza season is imperative, as resistance patterns and rates are ever evolving. This information should be used to guide decisions regarding preferred agents for prophylaxis and treatment.
Neuraminidase Inhibitors
These agents ( TABLE 2 ) are active against both influenza A and B viruses.6 Treatment with oseltamivir has been shown to reduce the duration of illness symptoms by 1 or 2 days when initiated within 36 hours and 24 hours, respectively.10,11 Oseltamivir treatment also decreases mortality.12

Oseltamivir is available as oral capsules and suspension.8 In 2012, the FDA changed this agent’s indications and usage to include treatment of patients aged >2 weeks.8 However, the use of oral oseltamivir in infants aged <14 days is recommended by the CDC and the American Academy of Pediatrics.6,13 Zanamivir is available as a powder for oral inhalation; it is not recommended for use in individuals with respiratory disease, including asthma and chronic obstructive pulmonary disease, and it is contraindicated in persons with an allergy to milk proteins.6,9 Treatment with oral zanamivir may be initiated in patients as young as 7 years and is not recommended in severe, complicated illness.6,9 In the event of an oseltamivir-resistant virus in a severely ill hospitalized patient, IV zanamivir is available for compassionate use via an emergency investigational new drug request to the manufacturer or enrollment in an ongoing phase III clinical trial.14 In a study addressing future resistance, the efficacy of combination treatment with oseltamivir and zanamivir was compared with each monotherapy, and combination therapy was found to be less effective.15
Treatment
The greatest clinical benefit from neuraminidase inhibitors occurs with early initiation, especially within 48 hours of onset of influenza illness. Treatment should begin as soon as possible in patients with confirmed or suspected influenza who are hospitalized, have severe or complicated illness, or are at high risk for influenza complications; in these instances, prescribers should not wait for laboratory confirmation of the virus before initiating treatment.6,16 A summary of factors that confer a high risk of complications secondary to influenza infection is given in TABLE 3 . In several observational studies, and more recently in a randomized, controlled trial, oseltamivir was found to shorten the length of illness and virus shedding when the antiviral was received after the 48-hour window, but within 5 days of onset.17

Neuraminidase inhibitors have been associated with a reduction in mortality in hospitalized patients versus no treatment. A meta-analysis comparing neuraminidase inhibitors initiated within 2 days of symptom onset with no treatment found an associated reduction in mortality risk (odds ratio 0.81, 95% CI 0.7-0.93; P = .0024).12 The mortality hazard ratio, which increased with each day’s delay in initiating treatment, still was found to be statistically significant.12 Therefore, it is recommended that neuraminidase inhibitor treatment be initiated as early as possible in any suspected or confirmed influenza illness.
Dosing and Duration
Dosing of neuraminidase inhibitors for oseltamivir and zanamivir in all age groups is summarized in TABLE 4 . Oseltamivir requires a dose adjustment for an estimated creatinine clearance <30 mL/min.8 Dose adjustment is not recommended in obese patients. A cohort study found a poor correlation between BMI >40 kg/m2 and drug clearance.18 Double-dose therapy has been described in the literature in hospitalized patients. A prospective intervention study detected no clinically significant difference in outcomes between 75 mg and 150 mg twice-daily dosing in hospitalized patients with severe influenza A infection.19 A double-blind, randomized, controlled trial investigated the effectiveness of double-dose oseltamivir and found a lack of additional benefit of double-dosing in adult and pediatric patients in a hospital setting.20
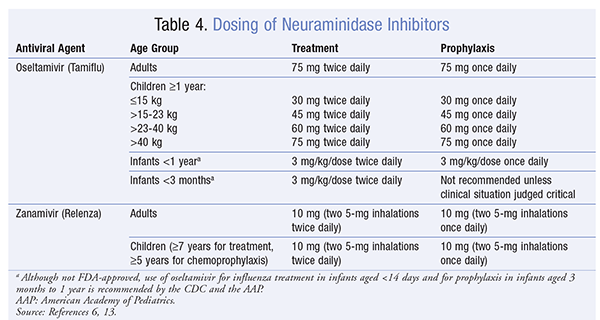
The FDA-recommended duration of treatment with neuraminidase inhibitors is 5 days.8,9 Some experts recommend a longer duration for severely ill hospitalized patients. Clinical judgment should guide the decision to continue therapy beyond the standard 5 days in severely ill hospitalized patients.
Conclusion
Influenza illness is associated with morbidity and mortality, and vaccination is the cornerstone of infection prevention. Pharmacists are of paramount importance in protecting patients through education and advocation of annual immunization. Diligent surveillance of each season’s predicted virus strains and resistance rates for available antivirals positions pharmacists to recommend appropriate vaccine products and proper selection of antivirals, when warranted.
REFERENCES
1. Influenza. In: CDC. Epidemiology and Prevention of Vaccine-Preventable Diseases. 12th ed. Washington, DC: Public Health Foundation; 2012.
2. CDC. Prevention and control of seasonal influenza with vaccines. Recommendations of the Advisory Committee on Immunization Practices—United States, 2013-2014. MMWR
Recomm Rep. 2013;62:1-43.
3. CDC.
FluView. www.cdc.gov/flu/weekly. Accessed April 25, 2014.
4.
Nafziger AN, Pratt DS. Seasonal influenza vaccination and technologies. J
Clin
Pharmacol. 2014;54:719-731
5. Norman JJ,
Arya JM, McClain MA, et al.
Microneedle patches: usability and acceptability for self-vaccination against influenza. Vaccine. 2014;32:1856-1862.
6. CDC. Influenza antiviral medications: summary for clinicians. www.cdc.gov/flu/professionals/antivirals/summary-clinicians.htm. Accessed March 29, 2014.
7.
Katzung BG, Masters SB, Trevor AJ. Basic & Clinical Pharmacology. 12th ed. New York, NY: McGraw Hill; 2012:886-887.
8. Tamiflu (
oseltamivir) product information. South San Francisco, CA: Genentech,
Inc; January 2013.
9.
Relenza (
zanamivir) product information. Research Triangle Park, NC: GlaxoSmithKline; December 2011.
10. Nicholson KG, Aoki FY,
Osterhaus AD, et al. Efficacy and safety of
oseltamivir in treatment of acute influenza: a
randomised controlled trial. Neuraminidase Inhibitor Flu Treatment Investigator Group. Lancet. 2000;355:1845-1850.
11.
Treanor JJ, Hayden FG,
Vrooman PS, et al. Efficacy and safety of the oral neuraminidase inhibitor
oseltamivir in treating acute influenza: a randomized controlled trial. US Oral Neuraminidase Study Group. JAMA. 2000;283:1016-1624.
12.
Muthuri SG,
Venkatesan S, Myles PR, et al. Effectiveness of neuraminidase inhibitors in reducing mortality in patients admitted to hospital with influenza A H1N1pdm09 virus infection: a meta-analysis of individual participant data. Lancet
Respir Med. 2014;2:395-404.
13. American Academy of Pediatrics Committee on Infectious Diseases. Recommendations for prevention and control of influenza in children, 2013–2014. Pediatrics. 2013;132:e1089-e1104.
14. CDC. Intravenous influenza antiviral medications and CDC considerations related to investigational use of intravenous
zanamivir for 2013-2014 influenza season. www.cdc.gov/flu/professionals/antivirals/intravenous-antivirals.htm. Accessed April 29, 2014.
15. Duval X, van der
Werf S,
Blanchon T, et al. Efficacy of
oseltamivir-zanamivir combination compared to each
monotherapy for seasonal influenza: a randomized placebo-controlled trial.
PLoS Med. 2010;7:e1000362.
16.
Viasus D,
Paño-Pardo JR,
Pachón J, et al. Timing of
oseltamivir administration and outcomes in hospitalized adults with pandemic 2009 Influenza A(H1N1) virus infection. Chest. 2011;140:1025-1032.
17. Fry AM,
Goswami D,
Nahar K, et al. Efficacy of
oseltamivir treatment started within 5 days of symptom onset to reduce influenza illness duration and virus shedding in an urban setting in Bangladesh: a
randomised placebo-controlled trial. Lancet Infect Dis. 2014;14:109-118.
18.
Pai MP,
Lodise TP Jr.
Oseltamivir and
oseltamivir carboxylate pharmacokinetics in obese adults: dose modification for weight is not necessary.
Antimicrob Agents
Chemother. 2011;55:5640-5645.
19. Lee N,
Hui DS,
Zuo Z, et al. A prospective intervention study on higher-dose
oseltamivir treatment in adults hospitalized with influenza A and B infections.
Clin Infect Dis. 2013;57:1511-1519.
20. South East Asia Infectious Disease Clinical Research Network. Effect of double dose
oseltamivir on clinical and
virological outcomes in children and adults admitted to hospital with severe influenza: double blind
randomised controlled trial. BMJ. 2013;346:f3039.
To comment on this article, contact rdavidson@uspharmacist.com.





