US Pharm. 2011;36(6):12-15.
When patients ask for assistance with selection of an appropriate nonprescription product, it is vital for the pharmacist to be aware of the various label precautions and warnings. Several groups of products present potential problems for patients with a history of kidney disease or prostate enlargement.
Kidney Disease
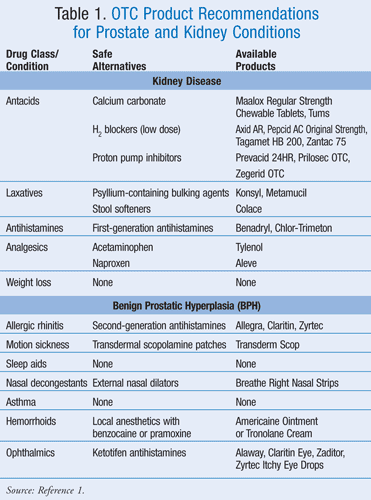
Certain groups of nonprescription medications present precautions in patients with kidney problems, usually due to potential toxicity if the medications cannot be renally eliminated. Fortunately, there are alternatives for each that the pharmacist can recommend (TABLE 1).
Acid Reducers: Antacids containing more than 50 mEq of magnesium or 25 mEq of potassium per maximum recommended daily dose must carry a warning recommending that patients speak to a physician before use if they have kidney disease.1 This warning is meant to prevent hypermagnesemia or hyperkalemia in patients with a hampered ability to clear these ions. The warning is found on such products as Alka-Seltzer Gold (344 mg of potassium bicarbonate per tablet) and Maalox Advanced Regular Strength Suspension (200 mg of magnesium hydroxide per teaspoon).2,3
A kidney warning is also found on histamine-2 (H2) blockers such as Pepcid AC Maximum Strength (20 mg of famotidine per tablet) and Zantac 150 (150 mg of ranitidine per tablet).4,5 The pharmacist can recommend alternative products without kidney warnings for these patients. Calcium carbonate is considered safe for kidney patients when used as directed. It is the ingredient in such products as Tums and Maalox Regular Strength Chewable Tablets.1
Should the patient wish to purchase a H2 blocker, the pharmacist can recommend low-dose options such as Zantac 75 (75 mg of ranitidine per tablet) or Tagamet HB 200, Axid AR, or Pepcid AC Original Strength (each 10 mg of famotidine per tablet). Proton pump inhibitors (e.g., Prilosec OTC, Prevacid 24HR, Zegerid OTC) are free of kidney warnings, so they would also be safe for kidney patients when used as directed.1
Laxatives: Laxatives also carry a warning against use by kidney patients without the advice of a physician if they contain more than 50 mEq of magnesium or 25 mEq of potassium per maximum recommended daily dose.1 Kidney warnings appear on such liquid products as Phillips’ Milk of Magnesia (1,200 mg magnesium hydroxide per tablespoon), Phillips’ Concentrated MOM (2400 mg magnesium hydroxide per tablespoon), and Phillips’ M-O (300 mg of magnesium hydroxide per teaspoon).6-8 Kidney warnings are also found on solid products such as Phillips’ Caplets (500 mg of magnesium per caplet) and Phillips’ Chewable Tablets (311 mg of magnesium hydroxide per tablet).9,10 Kidney disease warnings are required on laxatives containing polyethylene glycol 3350, such as MiraLAX and Dulcolax Balance.11,12
When patients with kidney disease request assistance with constipation, the pharmacist should ask whether they are on a fluid-restricted diet, as may be the case with patients on dialysis. If not, it is prudent to first recommend that the patient increase intake of fluids, exercise, and supplement the diet with high fiber foods, as well as ingesting psyllium-containing bulking agents, such as Konsyl or Metamucil. The pharmacist can also recommend stool softeners, such as those containing docusate (e.g., Colace).1
Antihistamines: Nonprescription second-generation antihistamines all carry a warning against unsupervised use if the patient has kidney disease, including Claritin, Zyrtec, and the newly switched Allegra OTC. Their chief advantage in the treatment of allergic rhinitis is the relatively lower incidence of sedation when compared to first-generation products. However, when patients with kidney disease ask the pharmacist for assistance in treating allergic rhinitis, it is critical to recommend such first-generation products as Chlor-Trimeton or Benadryl. In these cases, the pharmacist must point out the potential for drowsiness when using them. Patients should be cautioned against driving a car or operating heavy machinery while using them, and to avoid concomitant use of medications that also cause sedation, such as sedatives or tranquilizers.
Internal Analgesics: If internal analgesic products contain magnesium salicylate in a sufficient quantity so that a patient might ingest more than 50 mEq when taking the recommended daily dose, a kidney warning is required. This includes such products as Percogesic Maximum Strength Backache Relief (formerly known as Momentum), containing 580 mg of magnesium salicylate tetrahydrate per caplet.13 Products containing ibuprofen also carry a kidney disease warning. This includes single-entity products such as Motrin IB, and also combination products containing ibuprofen (e.g., Motrin PM, Advil Migraine).14 Patients with kidney disease who need pain relief could be steered to analgesics containing acetaminophen (e.g., Tylenol) or naproxen (e.g., Aleve).
Weight Loss Products: The manufacturer of prescription Xenical (orlistat) states in its package insert, “Some patients taking Xenical may develop an increased risk for the development of kidney stones. Promptly report any symptoms of back pain or blood in the urine.”15 When orlistat became a nonprescription product under the trade name alli, the FDA required a label instructing patients to ask a doctor before use if they have ever had kidney stones. Alli is the only OTC product that is proven safe and effective for producing weight loss. Thus, there is no safe and effective nonprescription alternative the pharmacist can recommend for patients with a past or present history of kidney stones who wish to lose weight. These patients might be advised to enroll in a reputable weight loss program (e.g., Weight Watchers).
Prostate Enlargement
Several groups of OTC products are troublesome for patients with an enlarged prostate, also known as benign prostatic hyperplasia (BPH).
Allergic Rhinitis Products: First-generation antihistamines carry a label warning patients against use if they have difficulty in urination due to an enlarged prostate gland, unless directed to do so by a physician.1 The products worsen symptoms of BPH and may make it even more difficult to urinate.16,17 Second-generation antihistamines (e.g., Claritin, Zyrtec, Allegra OTC) do not carry a prostate warning and would perhaps be a better choice for patients with allergic rhinitis who have BPH.
Motion Sickness Products: All safe and effective products for motion sickness contain antihistamines, either dimenhydrinate (e.g., Dramamine), cyclizine (e.g., Marezine, Bonine for Kids), or meclizine (e.g., Bonine, Dramamine Less Drowsy Formula).1 Being first-generation antihistamines, they all carry the risk of worsening BPH symptoms.1 No other safe and effective product is available for motion sickness without a prescription. Perhaps the patient would benefit from transdermal scopolamine patches (e.g., Transderm Scop), although they may also present problems for patients with urinary difficulty. The physician would be required to make a need-versus-risk decision before prescribing the product.
Sleep Aids: The only safe and effective nonprescription sleep aids contain diphenhydramine (e.g., Sominex) or doxylamine (e.g., Unisom Sleep-Tabs).1 Both can affect the prostate, and there is no other nonprescription option.1 Herbals (e.g., valerian), dietary supplements (e.g., melatonin), and homeopathics are not known to be safe and effective. A physician referral is the best advice.
Nasal Decongestants: Both topical and oral nasal decongestants carry a warning against use by males who have an enlarged prostate. These products increase muscular tone at the bladder neck, further hampering urination. Worrisome oral nasal decongestant ingredients include pseudoephedrine (e.g., Sudafed) and phenylephrine (e.g., Sudafed PE), whereas topicals of concern include oxymetazoline (e.g., Afrin), phenylephrine (e.g., Neo-Synephrine), and levmetamfetamine (e.g., Vicks VapoInhaler). The safest option is the use of external nasal dilators (e.g., Breathe Right Nasal Strips), which act through mechanical means to dilate the nostrils and present no problem to patients with prostatic enlargement.
Hemorrhoid Products: Hemorrhoid products of concern with an enlarged prostate contain vasoconstrictors, potentially reducing urine flow in a manner similar to oral and topical nasal decongestants. The most common ingredient for which a prostate warning is appropriate is phenylephrine, as found in Preparation H Ointment and Suppositories.18 The manufacturer’s Web site explains that the suppositories are temporarily unavailable due to a supply disruption, but promises that they are “working diligently to get product back on store shelves.”18 As alternative products for patients with hemorrhoids and concomitant BPH, pharmacists might suggest products containing local anesthetics, such as Tronolane Cream (with pramoxine) or Americaine Ointment (with benzocaine).
Asthma Products: Nonprescription products for asthma are not considered to be optimal therapy when compared to the safer and more effective prescription products.1 The nonprescription asthma products contain either ephedrine (tablets) or epinephrine (inhalers). Both ingredients carry the prostate warning, and pharmacists who are asked about alternatives should refer the patient for prescription therapy.
Ophthalmics: There is a curious situation in regard to ophthalmic products and prostate warnings. Single-entity ophthalmic vasoconstrictors for red eye are not required to carry a prostate warning, despite acting in the same manner as nasal decongestants.1 These products include Visine Original (tetrahydrozoline) and Clear Eyes (naphazoline). However, OTC products for allergic conjunctivitis have been marketed since 1995, and they contain naphaz-oline combined with the antihistamine pheniramine (e.g., Naphcon-A, Opcon-A, Visine-A). These products all must carry the prostate warning, suggesting that the addition of the antihistamine was critical for BPH patients. For patients with allergic conjunctivitis, pharmacists can suggest single-entity products containing ketotifen, an antihistamine that does not carry prostate warnings.1 These products include Zaditor, Alaway, Claritin Eye, and Zyrtec Itchy Eye Drops.
Conclusion
When patients with kidney disease, a history of kidney stones, or BPH require assistance in choosing an appropriate nonprescription product, informed pharmacists can direct them to products that do not present problems or can refer them to a physician when appropriate.
PATIENT INFORMATION

Some nonprescription products may not be cleared from your body and become toxic, while others may cause kidney stones. If males have prostate enlargement that is common with aging and often slows the flow of urine, there is also a set of products that can further slow the urine flow or even shut it off completely.
Kidney Problems
Many medications are eliminated from your body in your urine. In order for this to occur as it should, you must have adequate urinary flow from kidneys that produce the normal amount of urine daily. However, many patients have any of several kidney diseases that cause problems producing an adequate quantity of urine. Physicians may alter the dose of prescription medications when prescribing them to take the altered ability to eliminate them into account. The doctor may reduce the dose or prescribe it less often (such as once daily instead of twice).
However, when you wish to purchase nonprescription products, physicians are not usually involved in that decision. Therefore, you cannot know how to adjust the dose of nonprescription medications that might be dangerous. In these cases, the label simply warns against use if you have kidney disease. Pharmacists can assist you in locating this warning when it is present and can also recommend alternative products that do not present problems to patients with kidney disease. Examples of products that may present problems include acid reducers, laxatives, antihistamines, internal analgesics, and alli for weight loss.
Prostate Problems
Several groups of nonprescription products cause problems for men with urinary difficulty due to an enlarged prostate, usually due to a condition known as benign prostatic hyperplasia (BPH). Most often, the products have the potential to cause a further reduction in urine flow. If the man uses them without medical advice, he may have to be catheterized to empty the bladder. You should first ask your physician if the products might present problems. Prostate issues must be seen by a physician to rule out more serious conditions such as prostate cancer or prostate infection (prostatitis).
If you do not have a physician or are unable to contact your health care provider, you may ask the pharmacist for a recommendation. Nonprescription products presenting potential problems for men with BPH include some antihistamines for the common cold or allergic rhinitis, motion sickness products, sleep aids, nasal decongestants, some hemorrhoid products, asthma products, and some eye drops. Once again, your pharmacist can assist you in discovering whether this warning is present on a product you wish to purchase, and can direct you to a more suitable product.
REFERENCES
1. Pray WS. Nonprescription Product Therapeutics. 2nd ed. Baltimore, MD: Lippincott Williams & Wilkins; 2006.
2. Maalox Advanced Regular Strength. Novartis Consumer Health, Inc. www.maaloxus.com/index.shtml?
3. Alka-Seltzer Gold. Bayer HealthCare LLC. www.alka-seltzer.com/as/as_
4. Pepcid AC. Johnson & Johnson. www.pepcid.com/maximum-strength-pepcid-ac. Accessed April 28, 2011.
5. Maximum Strength Zantac 150. Boehringer Ingelheim Pharmaceuticals, Inc. www.zantacotc.com/products/
6. Phillips’ Milk of Magnesia. Bayer HealthCare LLC. http://phillipsrelief.com/
7. Phillips’ Concentrated MOM. Bayer HealthCare LLC. http://phillipsrelief.com/
8. M-O. Bayer HealthCare LLC. http://phillipsrelief.com/
9. Phillips’ Caplets. Bayer HealthCare LLC. http://phillipsrelief.com/
Accessed April 28, 2011.
10. Phillips’ Chewable Tablets. Bayer HealthCare LLC. http://phillipsrelief.com/
11. MiraLAX. Schering-Plough HealthCare Products, Inc. www.miralax.com/miralax/hcp/
12. Dulcolax Balance. Boehringer Ingelheim Pharmaceuticals, Inc. www.dulcolaxusa.com/consumer/
13. Percogesic. Drugstore.com. www.drugstore.com/percogesic-maximum-strength-
14. Motrin IB. McNeil-PPC, Inc. www.motrin.com/page.jhtml?id=/
15. Xenical. Genentech, Inc. www.gene.com/gene/
16. Enlarged prostate. MedlinePlus. www.nlm.nih.gov/medlineplus/
17. Prostate enlargement. National Kidney and Urologic Diseases Information Clearinghouse. http://kidney.niddk.nih.gov/
18. Preparation H products. Pfizer Consumer Healthcare. www.preparationh.com/hemorrhoid_ medications/suppositories.asp. Accessed April 28, 2011.
To comment on this article, contact rdavidson@uspharmacist.com.






