US Pharm. 2011;36(8):HS17-HS26.
Viral hepatitis remains a major cause of morbidity and mortality in the United States and worldwide.1 Viruses identified as causing viral hepatitis include hepatitis A to E. Of these viruses, hepatitis B virus (HBV) and hepatitis C virus (HCV) are of particular concern due to prevalence, the ability to cause chronic infection, and the complications associated with disease progression. HBV and HCV convey an increased risk for the development of liver cirrhosis and hepatocellular carcinoma (HCC), and HCV is the leading cause of liver transplantation.2,3 Identification and subsequent management of HBV and HCV are central to preventing long-term complications. This review focuses on the diagnosis and therapeutic options available for HBV and HCV.
HEPATITIS B
Infection with HBV may either be self-limited or progress to chronic infection. More than 350 million people globally and 800,000 to 1.4 million people in the U.S. are chronically infected with HBV.4 Many patients who are carriers of HBV will not progress to hepatic complications, but 15% to 40% will go on to develop grave ramifications of the disease, and all chronically infected individuals create a reservoir for disease transmission.2
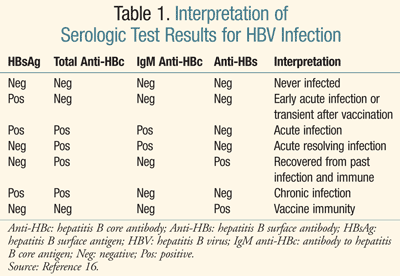
Early in the course of chronic HBV infection, serum HBV DNA levels are elevated and hepatitis B e-antigen (HBeAg) is present in serum. Presence of HBeAg indicates high levels of viral replication. In patients who become chronically infected, hepatitis B surface antigen (HBsAg) and HBV DNA persist (TABLE 1).5
Therapeutic agents for the treatment of chronic HBV can achieve sustained suppression of HBV replication.4,5 The short-term goal of treatment is to achieve initial response in terms of HBeAg seroconversion, HBV DNA suppression, and alanine transaminase (ALT) normalization. Long-term goals of therapy are to prevent hepatic decompensation, prevent progression to cirrhosis and/or HCC, and prolong survival.6 For HBeAg-positive or HBeAg-negative chronic HBV patients, treatment is advised when the HBV DNA level is ³20,000 IU/mL (105 copies/mL) and serum ALT is >2 times the upper normal limit.2
There are currently seven therapeutic agents approved for the treatment of chronic HBV in the U.S. (TABLE 2). Two agents are interferons (IFNs), including standard IFN-alfa-2b and pegylated IFN (peg-IFN)-alfa-2a. The five other agents are nucleoside/nucleotide analogues (NAs) and include tenofovir, lamiv-udine, adefovir, entecavir, and telbivudine. Peg-IFN, tenofovir, or entecavir is preferred as initial therapy.2

Therapeutic Agents: Hepatitis B
Interferon: IFNs have antiviral activity but primarily confer antiproliferative and immunomodulatory effects. Because of this, there are minimal reports of HBV resistance to IFNs. An additional advantage to IFNs may be the potential for shorter duration of treatment.7 IFN is only available as a subcutaneous injection. Peg-IFN is the IFN of choice because this formulation has shown greater sustained viral suppression and requires less frequent administration versus standard IFN. Of the available peg-IFNs, only peg-IFN-alfa-2a (Pegasys) is FDA approved for HBV.2
Adverse effects include an initial influenza-like illness, including fever, chills, headache, malaise, and myalgia.8,9 Other noted adverse effects include anxiety, irritability, depression, and suicidal tendency. IFN has myelosuppressive effects, and patients should be monitored closely for potential neutropenia or thrombocytopenia. Contraindications and precautions that warrant consideration include history of suicidal tendency, active psychiatric or autoimmune illness, severe leukopenia or thrombocytopenia, concurrent severe systemic disorders, or decompensated cirrhosis. Administration of acetaminophen or a nonsteroidal anti-inflammatory drug (NSAID) such as ibuprofen prior to IFN injection may lessen the incidence of flulike symptoms. Patients may also tolerate IFN better with bedtime administration.8 Patients should be educated to report any signs and symptoms of depression in order to assess whether discontinuation of IFN is warranted or if psychotropic medication is a viable option for the duration of treatment.
Tenofovir: Tenofovir disoproxil fumarate is an NA with potent antiviral activity against both HBV and HIV. Despite a similar chemical structure, tenofovir has shown to have a higher barrier to development of drug resistance than adefovir. Tenofovir is generally well tolerated, but has been reported to cause Fanconi syndrome, renal insufficiency, osteomalacia, and decreased bone density.10 Serum creatinine levels should be checked every 12 weeks, and thus patients should be educated on the importance of following up with laboratory work in order to monitor renal function.
Lamivudine: Lamivudine is a synthetic NA with activity against HBV and HIV.11 Lamivudine is potent in preventing HBV replication and improving liver disease; however, resistance may develop.7 Factors contributing to lamivudine resistance include duration of treatment, high pretreatment serum HBV DNA levels, and high levels of residual virus after initiation of treatment.12 Due to potential resistance, initiation of lamivudine treatment should only be considered when the use of an alternative antiviral agent with a higher genetic barrier to resistance is not available or appropriate.11 The most common adverse effects include gastrointestinal (GI) effects such as nausea, vomiting, and diarrhea. Less common yet serious adverse events reported include lactic acidosis and hepatomegaly. Patients should therefore be educated on signs and symptoms such as muscle weakness, fatigue, shortness of breath, and abdominal pain.11
Adefovir: Adefovir dipivoxil is an NA that is active against wild-type HBV and lamivudine-resistant HBV. However, its anti-HBV activity is less potent than lamivudine’s, so it is not a recommended first-line therapy.2 In patients with lamivudine-resistant HBV, adefovir dipivoxil should be used in combination with lamivudine to maintain selective pressure and limit development of adefovir resistance.2 Nephrotoxicity has been reported in patients with compensated liver disease after continued adefovir therapy; therefore, it is recommended to check serum creatinine every 12 weeks.13 Consider modifying treatment if HBV DNA remains above 1,000 copies/mL with continued therapy. As with lamivudine, patients should be educated about the signs and symptoms of lactic acidosis and the importance of following up with providers to monitor renal function.
Entecavir: Entecavir is an NA that is more potent than lamivudine and adefovir. Lower rates of resistance in treatment-naïve patients have been reported, and entec-avir maintains activity against lamivudine-resistant HBV mutants. The most common adverse reactions are headache, fatigue, dizziness, and nausea. The drug should be administered on an empty stomach to optimize absorption.14
Telbivudine: Telbivudine is prone to high rates of resistance development, and telbivudine resistance usually confers resistance to lamivudine as well. Therefore, telbivudine monotherapy has a limited role in the treatment of HBV. Cases of myopathy and peripheral neuropathy have been reported, but the overall safety profile is similar to that of lamivudine.2,4
Patient Monitoring
Patients receiving IFNs require blood counts and liver function tests monitored every 4 weeks. Thyroid-stimulating hormone and HBV DNA levels should be evaluated every 12 weeks. All of the NAs have a warning for lactic acidosis and severe hepatomegaly with steatosis. For those receiving NA therapy, clinicians should monitor liver function tests every 12 weeks and HBV DNA levels every 12 to 24 weeks. If patients are HBeAg-positive at initiation of treatment, HBeAg/anti-HBe should be evaluated every 24 weeks during treatment regardless of treatment regimen. HBsAg should be evaluated every 6 to 12 months in those who have HBeAg-negative serology and undetectable serum HBV DNA levels.2 In addition, all NAs should be renally dose-adjusted when creatinine clearance is <50 mL/min.10,11,13-15
HEPATITIS C
An estimated 3.2 million persons in the U.S. have chronic HCV infection, and 8,000 to 10,000 people die every year from HCV-related liver disease. Of people infected with HCV, 75% to 85% develop chronic infection. Many go on to develop liver damage resulting in liver failure and ultimately death. HCV is the leading cause of cirrhosis and liver cancer, and remains the most common reason for liver transplantation in the U.S.3,16
HCV can cause both acute and chronic hepatitis. The acute process does not require treatment but frequently progresses to chronic infection. Chronic HCV infection often follows a progressive course over many years and can ultimately result in cirrhosis, HCC, and the need for liver transplantation.17 The diagnosis of HCV infection is based on serum testing of antibody to HCV (anti-HCV) and HCV RNA. There are six known genotypes (1-6). Genotyping is recommended before initiation of treatment, since duration of treatment and response to therapy differ based on the genotype.3 Genotypes 2 and 3 are more likely to respond to therapy; however, genotype 1 is the most common cause of HCV in the U.S.3
Treatment Approach
The goal of antiviral therapy in patients with chronic HCV is to eradicate HCV RNA, which is predicted by attainment of a sustained virological response (SVR), defined as an undetectable HCV RNA at week 24 of treatment. SVR is associated with a 99% chance of being HCV RNA negative during long-term follow-up, and attaining SVR has been associated with decreases in all-cause mortality, liver-related death, need for liver transplantation, HCC rates, and liver-related complications.17 Undetectable virus at the end of either a 24- or 48-week course of therapy is referred to as an end-of-treatment response (ETR). Following ETR and treatment discontinuation, any return to HCV RNA serum positivity is defined as virological relapse.3
Therapeutic Agents: Hepatitis C
Standard pharmacotherapy for chronic HCV infection is the combination of a peg-IFN-alfa and ribavirin (Copegus). There are two available peg-IFNs for the treatment of HCV, peg-IFN-alfa-2b (Peg-Intron) and peg-IFN-alfa-2a (Pegasys). In clinical trials, peg-IFN-alfa has demonstrated superiority over standard IFN-alfa.3
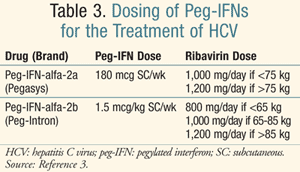
For genotypes 1 and 4, the recommended treatment duration with peg-IFN plus ribavirin is 48 weeks. Of note, the doses of peg-IFNs differ (TABLE 3).3 Treatment may be discontinued in patients who do not achieve an early virological response (EVR) defined as a >2 log reduction in HCV RNA at week 12 of treatment. Patients who do not achieve a complete EVR at 12 weeks should be retested at week 24. Treatment should be discontinued if HCV RNA remains positive. Clinicians should consider extending therapy to 72 weeks for patients with genotype 1 infection whose HCV RNA test becomes negative between weeks 12 and 24. For genotype 2 and 3 HCV infections, treatment with peg-IFN plus ribavirin should be administered for 24 weeks, using a ribavirin dose of 800 mg and the same peg-IFN doses as genotypes 1 and 4. When treatment continues through 24 weeks and measurement of HCV RNA is negative, follow-up testing for HCV RNA should be completed after another 24 weeks to evaluate for SVR. Patients with HCV-related cirrhosis who achieve an SVR regardless of the genotype should continue to be monitored every 6 to 12 months for the development of HCC. Due to limited prevalence, genotypes 5 and 6 are underrepresented in trials of peg-IFN and ribavirin; thus, the guidelines for the management of HCV from the American Association for the Study of Liver Diseases (AASLD) do not provide specific dosing recommendations.3
The most common adverse effects associated with peg-IFN use are flulike side effects such as fatigue, headache, fever, and rigors, which occurred in more than half of the patients in clinical trials. Psychiatric side effects, including depression, irritability, and insomnia, occurred in 22% to 31% of patients.8,18
Ribavirin is renally eliminated, and use is not recommended when creatinine clearance is <50 mL/min. The most common side effect is hemolytic anemia. Other adverse effects include mild lymphopenia, hyperuricemia, itching, rash, cough, and nasal stuffiness.19 Ribavirin is reported to cause fetal death and fetal abnormalities in animals (teratogenic), and thus anyone receiving the drug should use strict contraceptive methods both during treatment and for a period of 6 months thereafter.19 It is important to monitor symptoms of anemia such as shortness of breath, fatigue, and change in stool color and to obtain a regular CBC while taking ribavirin.
New Developments
Boceprevir (Victrelis) and telaprevir (Incivek) are oral protease inhibitors that were approved in 2011 for the treatment of hepatitis C (genotype 1) in combination with peg-IFN and ribavirin.20,21 They target the NS3/4A protease responsible for processing proteins essential for the HCV replication cycle. In clinical trials leading to their approval, both agents showed increased rates of SVR in treatment-naïve patients with genotype 1 HCV and in chronic genotype 1 HCV–infected patients with prior treatment failure when used with the combination of a peg-IFN-alfa and ribavirin compared to peg-IFN-alfa and ribavirin alone.22-25
Of note to clinicians, both medications have the potential for significant drug–drug interactions. Boceprevir is a potent inhibitor and partial substrate of the hepatic isoenzymes CYP3A4 and CYP3A5, and it is also an inhibitor and substrate of the efflux transporter P-glycoprotein (P-gp).20 Telaprevir is an inhibitor and substrate of the hepatic isoenzyme CYP3A4. In vitro studies suggest that telaprevir has a low potential to induce CYP2C, CYP3A, and CYP1A enzymes. Telaprevir is also an inhibitor and substrate of P-gp.21 The most common adverse events reported in early trial data included incidence of anemia with boceprevir and pruritus and rash with telaprevir.26 Other potential concerns include cost of the medications and barriers to the development of HCV resistance. It is too early to realize the clinical impact; however, these agents are a promising oral therapy option that will add to the limited treatment armamentarium against HCV.
CO-INFECTION: HBV/HIV
Co-existent HIV infection has been estimated to be present in 10% to 15% of patients with chronic HBV.27 The indications for HBV therapy are the same as in HIV-negative patients who meet criteria for chronic HBV treatment; however, clinicians may elect to treat earlier, given the accelerated course of HBV and increased risk of cirrhosis in HIV-infected individuals.7,28 Patients who are not on highly active antiretroviral therapy (HAART), and are not anticipated to require HAART in the near future, should be treated with an antiviral therapy that does not target HIV, such as peg-IFN or adefovir. Patients receiving treatment for both HBV and HIV should be prescribed a HAART regimen that consists of NA backbones that are effective against both viruses. The preferred agents are lamivudine plus tenofovir or emtricitabine plus tenofovir.2
If tenofovir cannot be used due to contraindications, alternative agents include entecavir, peg-IFN, adefovir with lamivudine, or adefovir with emtricitabine.2,29 When HAART regimens are altered, drugs that are effective against HBV should not be discontinued without substituting another drug that has activity against HBV, unless the patient has achieved HBV treatment goals.29 Clinicians should be aware of the potential reactivation of HBV and subsequent consequences if agents active against this virus are abruptly discontinued.2,29
CONCLUSION
Hepatitis B and C remain challenging infectious diseases responsible for significant morbidity and mortality. Available treatments are not always effective and are fraught with potential complications. Patients should be counseled extensively on their medications and monitored closely for adverse effects and efficacy of treatment. Pharmacists can make a significant impact in these disease states by optimizing appropriate and safe medication use while providing important education to their patients.
REFERENCES
1. Gonzalez SA, Keeffe EB. Chronic viral hepatitis: epidemiology, molecular biology, and antiviral therapy. Front Biosci. 2011;16:225-250.
2. Lok AS, McMahon BJ. Chronic hepatitis B: update 2009. Hepatology. 2009;50:661-662.
3. Ghany MG, Strader DB, Thomas DL, Seeff LB; American Association for the Study of Liver Diseases. Diagnosis, management, and treatment of hepatitis C: an update. Hepatology. 2009;49:1335-1374.
4. Weinbaum CM, Williams I, Mast EE, et al. Recommendations for identification and public health management of persons with chronic hepatitis B virus infection. MMWR Recomm Rep. 2008;57:1-20.
5. McMahon BJ. Epidemiology and natural history of hepatitis B. Semin Liver Dis. 2005;25(suppl 1):3-8.
6. Liaw YF, Leung N, Kao JH, et al. Asian-Pacific consensus statement on the management of chronic hepatitis B: a 2008 update. Hepatol Int. 2008;2:263-283.
7. Papatheodoridis GV, Manolakopoulos S. EASL clinical practice guidelines on the management of chronic hepatitis B: the need for liver biopsy. J Hepatol. 2009;51:226-227.
8. Pegasys (peginterferon alfa-2a) package insert. Nutley, NJ: Hoffmann-La Roche Inc; February 2011.
9. Intron A (interferon alfa-2b) package insert. Kenilworth, NJ: Schering-Plough; 2008.
10. Viread (tenofovir) package insert. Foster City, CA: Gilead Sciences, Inc; October 2010.
11. Epivir (lamivudine) package insert. Research Triangle Park, NC: Glaxo-SmithKline; January 2011.
12. Lai CL, Leung N, Teo EK, et al. A 1-year trial of telbivudine, lamivudine, and the combination in patients with hepatitis B e antigen-positive chronic hepatitis B. Gastroenterology. 2005;129:528-536.
13. Hepsera (adefovir dipivoxil) package insert. Foster City, CA: Gilead Sciences, Inc.; October 2009.
14. Baraclude (entecavir) package insert. Princeton, NJ: Bristol-Myers Squibb; December 2010.
15. Tyzeka (telbivudine) package insert. East Hanover, NJ: Novartis Pharmaceuticals Corporation; March 2011.
16. Workowski KA, Berman S; CDC. Sexually transmitted diseases treatment guidelines, 2010. MMWR Recomm Rep. 2010;59:1-110.
17. Swain MG, Lai MY, Shiffman ML, et al. A sustained virologic response is durable in patients with chronic hepatitis C treated with peginterferon alfa-2a and ribavirin. Gastroenterology. 2010;139:1593-1601.
18. PegIntron (peginterferon alfa-2b) package insert. Kenilworth, NJ: Schering Corporation; 2006.
19. Copegus (ribavirin) package insert. South San Francisco, CA: Genentech USA Inc; December 2010.
20. Victrelis (boceprevir) package insert. Whitehouse Station, NJ: Merck & Co., Inc; May 2011.
21. Incivek (telaprevir) package insert. Cambridge, MA: Vertex Pharmaceuticals, Inc; May 2011.
22. Bacon BR, Gordon SC, Lawitz E, et al. Boceprevir for previously treated chronic HCV genotype 1 infection. N Engl J Med. 2011;364:1207-1217.
23. Poordad F, McCone J Jr, Bacon BR, et al. Boceprevir for untreated chronic HCV genotype 1 infection. N Engl J Med. 2011;364:1195-1206.
24. Marcellin P, Forns X, Goeser T, et al. Telaprevir is effective given every 8 or 12 hours with ribavirin and peginterferon alfa-2a or -2b to patients with chronic hepatitis C. Gastroenterology. 2011;140:459-468.
25. McHutchison JG, Everson GT, Gordon SC, et al. Telaprevir with peg-interferon and ribavirin for chronic HCV genotype 1 infection. N Engl J Med. 2009;360:1827-1838.
26. Pawlotsky JM. The results of phase III clinical trials with telaprevir and boceprevir presented at the Liver Meeting 2010: a new standard of care for hepatitis C virus genotype 1 infection, but with issues still pending. Gastroenterology. 2011;140:746-754.
27. Strader DB. Understudied populations with hepatitis C. Hepatology. 2002;36(suppl 1):S226-S236.
28. Soriano V, Puoti M, Peters M, et al. Care of HIV patients with chronic hepatitis B: updated recommendations from the HIV-Hepatitis B Virus International Panel. AIDS. 2008;22:1399-1410.
29. Panel on Antiretroviral Guidelines for Adults and Adolescents. Guidelines for the use of antiretroviral agents in HIV-1-infected adults and adolescents. Department of Health and Human Services. January 10, 2011. www.aidsinfo.nih.gov/
To comment on this article, contact rdavidson@uspharmacist.com.





