US Pharm. 2013;38(2):38-42.
ABSTRACT: Sudden cardiac death (SCD) has been associated with diabetes, although it is unclear whether diabetes is a risk factor for SCD. Diabetes is a risk factor for common preventable comorbidities associated with SCD, including coronary artery disease (CAD), myocardial infarction, and heart failure. Researchers are seeking ways to measure susceptibility to SCD, but because of SCD’s multifactorial development, most likely no single test will be able to identify at-risk individuals. Until tests with proven predictive value are available, preventive efforts should focus on slowing the progression or development of cardiovascular diseases that frequently cause SCD. This can be accomplished by promptly initiating ACE inhibitors, angiotensin receptor blockers, beta-blockers, antiplatelet agents (aspirin), and possibly statin therapy for CAD, hypertension, and/or hypercholesterolemia. SCD prevention strategies for the general population are the same as those for patients with diabetes.
Sudden cardiac death (SCD) is an event that can occur in asymptomatic individuals, as well as in those with advanced cardiovascular (CV) disease.1 SCD typically manifests as a structural abnormality coupled with a disturbance in cardiac electrical activity that leads to fatal arrhythmias.1,2 However, in 5% to 10% of SCD cases, no definable structural cardiac abnormality exists.3 Patients may either be asymptomatic or experience symptoms including palpitations, chest pain or discomfort, dyspnea, fatigue, or syncope.3-5 Sudden cardiac arrest (SCA), in which an electrical malfunction causes the heart to stop abruptly, may occur in these patients; if the heart is not quickly shocked back into rhythm, SCD ensues. At least 90% to 95% of SCA cases in the community setting end in death before the patient receives medical assistance.3,6
In the United States, the annual number of SCDs is estimated to be between 250,000 and 300,000, with a predominance in males and an increased incidence with age for both genders.1,3,6,7 SCD is the first cardiac event in approximately 55% of men and 68% of women, while in other cases SCD follows multiple cardiac events.2,6 Unfortunately, the prevalence of SCD is difficult to determine since there previously was no unified definition of the condition. In general, SCD is defined as unexpected death occurring within a specific time frame after initial onset of cardiac symptoms.8 Depending upon the study being reviewed for SCD, the time frame between symptom onset and death varies greatly, from less than 1 hour up to 24 hours.
The most common preventable comorbidities associated with SCD include coronary artery disease (CAD), myocardial infarction (MI), and heart failure (HF).1 Since diabetes is a risk factor for these comorbidities, it has been deemed to have an association with SCD. The association between diabetes and SCD may involve a combination of macrovascular and microvascular complications that can affect the electrical system controlling cardiac rhythm, thus increasing the propensity for SCD.9,10 With the number of diabetes patients growing in epidemic proportions (approximately 29.9 million in the U.S.), there is an urgent need to determine whether having diabetes increases one’s risk for SCD.11
Macrovascular Disease
Approximately 80% of SCD cases have underlying CAD; 90% of cases have arrhythmias which may be caused by CAD.5,8,12-14 CAD exhibits a direct role in SCD, although the exact mechanism provoking the condition is unclear and may be multifactorial. The study with perhaps the most insight into the relationship between SCD and macrovascular disease is the Framingham Heart Study, in which long-term trends in CAD and SCD were evaluated. A Framingham multivariate risk index combined several CV risk factors (e.g., systolic blood pressure, total cholesterol, relative weight) with other CAD risk factors (e.g., age, smoking, heart rate [HR], vital capacity, ECG abnormalities) to predict SCD risk.9 The predicted risk of SCD increased with the addition of each risk factor. However, at 15-year follow-up, the number of SCDs was significantly lower than was predicted through the risk index. Thus, the Framingham risk index may be an acceptable tool overall for risk prediction in the general population, but perhaps less useful for individual risk prediction until more specific predictors of SCD are identified.3
Cardiac Autonomic Neuropathy (CAN): Chronic hyperglycemia contributes to progressive autonomic neural dysfunction.15 Patients with CAN typically experience resting tachycardia and less HR variability during exercise.10 CAN is characterized initially by an early increase of cardiac sympathetic activity, which leads to apoptosis and myocardial injury.15 Patients with diabetes-related CAN have increased mortality compared with patients with nondiabetes-related CAN, and they are more likely to develop arrhythmias, cardiomyopathy, silent ischemia, and subsequent SCD.9,10,15 Patients would benefit from learning how to recognize less distinct signs of ischemia, such as dyspnea with or without cough, extreme fatigue, and sudden onset of nausea and vomiting. In the Diabetes Control and Complications Trial, intensive glycemic control slowed CAN’s development and progression and reduced the progression of atherosclerosis and microvascular disease in patients with type 1 diabetes (T1DM).1,9,16 This has not been demonstrated in type 2 diabetes (T2DM) patients, however. Additionally, SCD was not directly assessed in this trial. Furthermore, it has since been found that glycemic control that is too intensive may increase mortality.17 Data are inconclusive regarding intensive glycemic control and reduced risk of CAN in patients with T2DM. However, if possible, intensive glycemic control should be recommended in T1DM patients to reduce the risk of CAN.
Silent Ischemia: Silent ischemia is a painless condition in which narrowed or blocked arteries prevent oxygen-rich blood from reaching the heart. Diabetes patients, particularly those with CAN, are at increased risk for developing silent ischemia, which eventually may lead to SCD secondary to arrhythmias.8,10 However, no studies clearly demonstrate the risk of SCD due to silent ischemia in patients with diabetes.
QT-Interval Prolongation: The Rotterdam Heart Study and the Oregon Sudden Unexpected Death Study provided evidence that prolonged QT intervals heighten the risk of SCD in the general population.1,4,5,18,19 QT-interval prolongation is common in diabetes, particularly in CAN patients, although the mechanism behind this association is unclear.8,19 QT-interval prolongation has been indicated as an independent predictor of mortality and has been associated with a suspected increase in risk of SCD in diabetes patients; however, further research is necessary to determine the association between QT prolongation and SCD in patients with diabetes.8,9
HF: Systolic dysfunction—particularly left ventricular ejection fraction (LVEF) below 30% to 35%—increases the risk of SCD in the general population, but little is known about the effect of diastolic dysfunction on SCD risk.1,4,8 Patients with less severe HF are more likely to die from SCD, whereas those with more severe HF (i.e., New York Heart Association [NYHA] Class IV) are more likely to die from pump failure.4,20 However, since the NYHA classification can rapidly fluctuate between Class I and Class IV, this approach only estimates functional consequences, so the usefulness of HF classification as a marker for risk of SCD is minimal. In a post hoc analysis of the Valsartan in Acute Myocardial Infarction Trial, LVEF was a strong predictor of SCD, which increased by 21% with every 5% reduction in ejection fraction.20,21 Left ventricular systolic dysfunction is established as a strong predictor of SCD.6,21
Microvascular Disease
Microvascular disease caused by diabetes may result in autonomic neuropathy, which may lead to QT-interval prolongation. Optimal glycemic control reduces the incidence of microvascular disease, but at the risk of increased hypoglycemic events.8,16,17,22 Frequent hypoglycemia has been indicated as a potential contributor to SCD.15
Hypoglycemia
Intensive glycemic control may reduce the risk of diabetes complications, but it also increases the incidence of hypoglycemia.8,16,17,22 Even a single hypoglycemic event can impair hormonal and autonomic responses to subsequent events.8,17 Once hormonal and autonomic responses to hypoglycemia are impaired, hypoglycemia unawareness or asymptomatic hypoglycemia may ensue. This impaired response may induce transient QT prolongation and promote a reduced threshold for arrhythmias and, subsequently, SCD.8,15
Prothrombotic State
Based on autopsy reports, thrombus, plaque disruption, or both occur in more than 50% of SCD cases.14 When stable atherosclerotic plaques fissure, platelet activation and aggregation occur, resulting in thrombosis.14 Plaque rupture occurs more often in women, particularly those of advanced age.14 Distribution of coronary artery lesions does not appear to play a substantial role in SCD development. However, acute thrombosis was observed two to three times more often in smokers than in nonsmokers, likely because of an increase in platelet adhesiveness.14 SCD was much more likely to occur in current cigarette smokers, underscoring the importance of tobacco cessation. The increased risk of SCD development in diabetes patients through early coronary atherosclerosis and thrombosis appears to be theoretical.9,10
Genetic Factors
Family history may be a significant risk factor for SCD, according to the Paris Prospective Study I (PPSI), a 23-year analysis of CV outcomes in 7,746 asymptomatic middle-aged men.23 In this study, diabetes and parental history of SCD were identified as independent risk factors for SCD.1,23,24 Multifactorial statistical analyses of PPSI indicated that the relative risk (RR) for development of SCD in offspring was 1.8 if only one parent experienced SCD; however, the RR rose to 9.4 if both parents had a positive family history.4,13,23,24 Thus, it appears that the risk of SCD is heightened in diabetes patients and in patients with a family history of SCD. Currently, researchers are examining ways to measure genetic susceptibility to SCD, but because of the multifactorial development of SCD, it is likely that no single test will be able to identify at-risk individuals.
Risk Prediction
It is understood that most patients with underlying CAD are at greater risk for SCD, and most efforts to stratify SCD risks have focused on patients with known CAD. However, there are no standardized recommendations for predicting risk in patients who have no underlying structural heart disease. Since SCD is multifactorial, prediction of individual risk is more difficult.3 Researchers have reviewed numerous markers for potential predictors of fatal arrhythmias, including QT dispersion, HR variability, and inflammatory markers such as C-reactive protein; however, they have not performed well as predictors of SCD.3 Diabetes, while currently recognized as a clinical risk marker, is not used for risk stratification to predict SCD.6 Only left ventricular systolic dysfunction is an established clinical risk predictor of SCD, but it is an effective predictor only in patients with a substantial reduction in LVEF.6 In an effort to elucidate the prevalence and risk of SCD, the National Heart, Lung, and Blood Institute and the Heart Rhythm Society have developed a unified definition for future studies (TABLE 1).6

Prevention Strategies
Because of the sudden and unpredictable nature of SCD, risk stratification continues to be an area of interest. The Framingham study revealed a 49% decline in SCD between 1990 and 1999 versus between 1950 and 1969, possibly because of the increased use of preventive agents and/or devices for CV morbidity and mortality.25 Automated external defibrillators, which became available in the 1970s, may have contributed to the decline in SCD prevalence.25 Implantable cardioverter defibrillators (ICDs), which were not routinely used until the 1990s, are recommended to prevent SCD in high-risk patients (e.g., prior MI, left ventricular dysfunction).1,4,9,26 Although the cost-effectiveness of ICDs as a population-based prevention strategy has been questioned, these devices remain a mainstay of SCD prevention.6
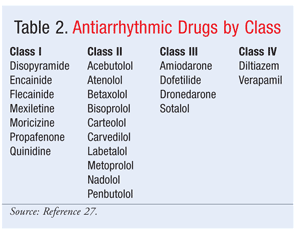
Theoretically, since the vast majority of deaths attributed to SCD involve underlying CAD, controlling the risk factors should indirectly reduce SCD incidence. In fact, many methods of SCD prevention have focused on slowing the progression or development of CV diseases that frequently cause SCD.6,26 Since patients with diabetes and those who experience SCD share risk factors such as CAD, hypercholesterolemia, and hypertension, controlling the comorbid conditions associated with diabetes should, by default, lessen the risk of SCD in diabetes patients. Pharmacologic agents believed to improve CV longevity include ACE inhibitors, beta-blockers, and antiplatelet agents (aspirin).9,26 Drugs that modulate the renin-angiotensin-aldosterone system, such as ACE inhibitors and angiotensin receptor blockers, are thought to have indirect antiarrhythmic effects and therefore may be useful for preventing SCD.27 Beta-blockers—in particular, carvedilol—prevent SCD in CAD and HF.27 Statins may help reduce SCD incidence by slowing the progression of CV disease; however, few data demonstrate an overall beneficial effect on SCD prevention.1 Interestingly, Class 1 antiarrhythmics (TABLE 2) are not recommended for SCD prevention, based on unexpected outcomes of the Cardiac Arrhythmia Suppression Trial (CAST) and CAST II, in which CAD patients using these agents had an increased risk of death.14,27 Class IV antiarrhythmics have limited use in preventing SCD, but Class III may be considered for this purpose.27 Nonpharmacologic prevention strategies include weight reduction, dietary education, smoking cessation, stress management, physical activity, and ICDs (in high-risk patients).1,4,6,9,14,26
Conclusion
It is difficult to ascertain whether diabetes is a direct risk factor for SCD. The notion that diabetes results in enhanced susceptibility to SCD is generally accepted, but is not evidence based. Therefore, although diabetes is recognized as a clinical risk marker for SCD, it is not currently used for risk stratification. If the incidence of SCD is greater in patients with diabetes, the root cause remains unclear. Overall, mortality remains high, most likely owing to the unexpectedness of the event and failure to recognize warning symptoms in a timely manner. Thus, it is critically important to develop prediction and prevention strategies for SCD.
REFERENCES
1. Spooner PM. Sudden cardiac death: influence of diabetes. Diabetes Obes Metab. 2008;10:523-532.
2. Myerburg RJ, Kessler KM, Castellanos A. Sudden cardiac death: structure, function, and time-dependence of risk. Circulation. 1992;85(suppl 1):I2-I10.
3. Podrid PJ, Myerburg RJ. Epidemiology and stratification of risk for sudden cardiac death. Clin Cardiol. 2005;28(11 suppl 1):I3–I11.
4. Govil AK, Gupta MD, Girish MP, Tyagi S. Prediction and prevention in sudden cardiac death. Apollo Med. 2011;8:228-237.
5. Chugh SS. Early identification of risk factors for sudden cardiac death. Nat Rev Cardiol. 2010;7:318-326.
6. Fishman GI, Chugh SS, DiMarco JP, et al. Sudden cardiac death
prediction and prevention: report from a National Heart, Lung, and Blood
Institute and Heart Rhythm Society Workshop. Circulation. 2010;122:2335-2348.
7. State-specific mortality from sudden cardiac death–United States, 1999. MMWR Morb Mortal Wkly Rep. 2002;51:123-126.
8. Bergner DW, Goldberger JJ. Diabetes mellitus and sudden cardiac death: what are the data? Cardiol J. 2010;17:117-129.
9. El-Atat FA, McFarlane SI, Sowers JR, Bigger JT. Sudden cardiac death in patients with diabetes. Curr Diab Rep. 2004;4:187-193.
10. El-Menyar AA. Dysrhythmia and electrocardiographic changes in
diabetes mellitus: pathophysiology and impact on the incidence of sudden
cardiac death. J Cardiovasc Med (Hagerstown). 2006;7:580-585.
11. CDC. Number (in millions) of civilian, noninstitutionalized
persons with diagnosed diabetes, United States, 1980–2010.
www.cdc.gov/diabetes/statistics/prev/national/figpersons.htm. Accessed
September 9, 2012.
12. Thomas KE, Josephson ME. The role of electrophysiology study in risk stratification of sudden cardiac death. Prog Cardiovasc Dis. 2008;51:97-105.
13. Arking DE, Chugh SS, Chakravarti A, Spooner PM. Genomics in sudden cardiac death. Circ Res. 2004;94:712-723.
14. Zipes DP, Wellens HJ. Sudden cardiac death. Circulation. 1998;98:2334-2351.
15. Pop-Busui R. Cardiac autonomic neuropathy in diabetes: a clinical perspective. Diabetes Care. 2010;33:434-441.
16. The effect of intensive treatment of diabetes on the development
and progression of long-term complications in insulin-dependent diabetes
mellitus. N Engl J Med. 1993;329:977-986.
17. Adler GK, Bonyhay I, Failing H, et al. Antecedent hypoglycemia
impairs autonomic cardiovascular function: implications for rigorous
glycemic control. Diabetes. 2009;58:360-366.
18. Hofman A, Breteler MM, van Duijn CM, et al. The Rotterdam Study: 2010 objectives and design update. Eur J Epidemiol. 2009;24:553-572.
19. Chugh SS, Reinier K, Singh T, et al. Determinants of prolonged QT
interval and their contribution to sudden death risk in coronary artery
disease: the Oregon Sudden Unexpected Death Study. Circulation. 2009;119:663-670.
20. Al-Khatib SM, Sanders GD, Bigger JT, et al. Preventing tomorrow’s
sudden cardiac death today: part I: current data on risk stratification
for sudden cardiac death. Am Heart J. 2007;153:941-950.
21. Solomon SD, Zelenkofske S, McMurray JJ, et al. Sudden death in
patients with myocardial infarction and left ventricular dysfunction,
heart failure, or both. New Engl J Med. 2005;352:2581-2588.
22. Intensive blood-glucose control with sulphonylureas or insulin
compared with conventional treatment and risk of complications in
patients with type 2 diabetes (UKPDS 33). Lancet. 1998;352:837-853.
23. Jouven X, Desnos M, Guerot C, Ducimetiere P. Predicting sudden death in the population: the Paris Prospective Study I. Circulation. 1999;99:1978-1983.
24. Balkau B, Jouven X, Ducimetière P, Eschwège E. Diabetes as a risk factor for sudden death. Lancet. 1999;354:1968-1969.
25. Fox CS, Evans JC, Larson MG, et al. Temporal trends in coronary
heart disease mortality and sudden cardiac death from 1950 to 1999: the
Framingham Heart Study. Circulation. 2004;110:522-527.
26. Ganz LI. Primary prevention of sudden cardiac death. Curr Cardiol Rep. 2004;6:339-347.
27. Das MK, Zipes DP. Antiarrhythmic and nonantiarrhythmic drugs for sudden cardiac death prevention. J Cardiovasc Pharmacol. 2010;55:438-449.
To comment on this article, contact rdavidson@uspharmacist.com.





