US Pharm. 2014;39(2):57-60.
ABSTRACT: The prevalence of hypertension in pediatrics is a serious topic that has continued to gain exposure among the healthcare community. Many conversations about the health status of the American population, particularly the growing concern surrounding trends in obesity, have brought new focus to the prevalence of hypertension in children. As a result, healthcare practitioners, principally pharmacists, must be adequately prepared to engage with and assist other members of the healthcare team in this realm. The article summarizes information pertinent to facilitating clinical recommendations regarding the safe and effective use of the treatment options available for this special population.
Pediatric hypertension (HTN) has become a growing problem over the past decade. It is estimated that 3% to 5% of the pediatric population is currently affected.1 The steady increase in the occurrence of HTN within pediatric patients has been associated with childhood obesity, the advancement in diagnosing childhood HTN, and the implementation of a national databank established by the National High Blood Pressure Education Program (NHBPEP) Working Group on Children and Adolescents.1-4 The development of HTN among pediatric patients raises several concerns for healthcare providers. These concerns focus on an increased risk of developing cardiovascular disease (CVD) as well as on risk associated with developing sustained HTN in early adulthood patients.4,5 The following summarizes recommendations for consideration by pharmacy practitioners as they engage in the treatment of children who may be battling HTN.
Etiology
There are two types of HTN—primary and secondary. Primary HTN may be due to family history of HTN or cardiovascular disease.4 Secondary HTN, however, is caused by another medical condition, such as renal disease, heart disease, or an endocrine system disorder. In the pediatric population, primary HTN is more commonly linked to adolescent patients (>12 years of age) and is often associated with obesity and family history of HTN or cardiovascular disease. The normal blood pressure (BP) range associated with HTN in pediatric patients varies based on age, sex, and height. Secondary HTN is more common in preadolescent children and is caused by an underlying disorder.4
Screening, Diagnostics, and Symptoms
Children 3 years of age and older should have their BP measured each time they present for a healthcare visit. It is recommended that children younger than 3 years have their BP measured if they have a previous history of certain conditions, such as prematurity or other neonatal complications, congenital heart disease, recurrent urinary tract infections, renal disease, a solid organ or bone marrow transplant, or elevated intracranial pressure.4
BP Measurement Techniques
The recommended method of BP measurement in children is auscultation (listening to the internal sounds of the body, usually using a stethoscope).4 However, a standard clinical sphygmomanometer with stethoscope should be used to confirm HTN when readings are ≥95th percentile.2 Children and adolescents awaiting a BP measurement should avoid stimulant drug use and food and drink containing caffeine, sugar, and chocolate for 30 minutes prior to the measurement. Sitting quietly for 5 minutes with feet flat on the floor, back supported, and right arm supported with cubital fossa at heart level prior to the reading is suggested.
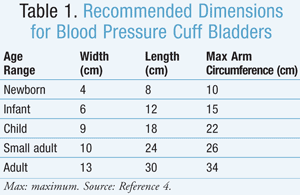
An appropriately sized cuff should have an inflatable bladder width of at least 40% of the arm circumference at a point midway between the elbow and shoulder.4 BP cuff sizes are listed in TABLE 1. If a cuff is too small, BP measurements may be incorrect and overestimated.4 In this case, the next largest cuff size should be used so as to minimize the cuff-size effect on the BP measurement.4 Normal BP for children and adolescents is defined as systolic (SBP) and diastolic (DBP) blood pressure <90th percentile for sex, age, and height (e.g., refer to the Blood Pressure Levels for Boys and Girls by Age and Height Percentile) because it provides a more precise reading according to body size.4,6
Classification
It is important to distinguish pediatric HTN from other types of HTN. Pediatric hypertension is diagnosed based on average SBP and/or DBP that is ≥95th percentile for sex, age, and height on three or more office visits.4 An average SBP or DBP level ≥90th percentile, but <95th percentile, is classified as prehypertensive.4 An adolescent with a BP of ≥120/80 mmHg is also considered prehypertensive, despite a DBP reading <90th percentile.4
Pulmonary arterial hypertension (PAH) is a disorder in which the pulmonary arteries constrict, causing an increase in resistance to blood flow that results in abnormally high blood pressure in the pulmonary arteries. PAH is defined as a mean pulmonary arterial pressure >25 mmHg at rest or >30 mmHg during exercise.6 Symptoms, which include dyspnea, fatigue, chest pain, and peripheral edema, appear gradually. While HTN refers to the systemic pressure throughout the body, it is not to be assumed that PAH and HTN will coexist in a patient.
White-coat hypertension, defined as elevated blood pressure in a clinical setting, is caused by general anxiety associated with an office visit. Patients who have BP readings >95th percentile in a physician’s office but whose average BP in a nonclinical setting is <90th percentile, are considered to have white-coat HTN.6
As with adult classification of HTN, pediatric HTN is divided into two stages—stage I and stage II. Patient symptoms such as headache, dizziness, and altered vision are not stage-specific. Stage I HTN is defined as BP levels ranging from the 95th percentile to 5 mmHg above the 99th percentile.6 Stage II HTN represents BP levels >5 mmHg above the 99th percentile.6
Nonpharmacologic Treatment
Lifestyle modifications may result in decreased blood pressure and an overall decrease in the cardiovascular risk factors.7 The Dietary Approach to Stop Hypertension (DASH) diet is often initiated in patients with an increased risk of cardiovascular events, and it should be considered in symptomatic pediatric patients starting at 12 months of age.8 The diet promotes a nutrition plan high in fruits and vegetables, nonfat dairy products, fiber, and low amounts of daily sodium consumption (1.2 g/day for 4 to 8 years of age and 1.5 g/day for older children).8
In addition to dietary modifications, weight loss in overweight patients and an increase in daily physical exercise are also beneficial.4 The NHBPEP suggests limiting sedentary activity throughout the day and conducting 30 to 60 minutes of physical exercise on 3 to 4 days during the week.9 Scheduled physical exercise can be achieved by participating in sports or family-based activities such as walking, biking, and household chores.4
Pharmacologic Treatment
Antihypertensive medication in children and adolescents should be initiated in the presence of symptomatic HTN, end-organ damage (e.g., left ventricular hypertrophy, retinopathy, proteinuria), secondary HTN, stage I HTN that does not respond to lifestyle changes, and stage II HTN.4 The treatment goal for pediatric HTN is to reduce the DBP to at least the 90th percentile for age and sex.10 In accordance with NHBPEP recommendations, pharmacologic therapy should be initiated in a stepwise approach. Initiation on a single antihypertensive agent should begin at the lowest effective dose.11
All of the major classes of antihypertensive medications can be used in children to some degree. Legislation such as the FDA Modernization Act of 1997 (FDAMA) and the Best Pharmaceuticals for Children Act (BPCA) of 2002 have led to the study and approval of antihypertensive medications for use in the pediatric population (TABLE 2).11-13
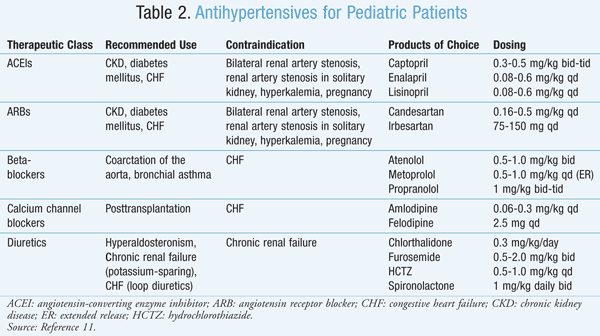
Angiotensin-Converting Enzyme Inhibitors (ACEIs): In comparison with other classes of antihypertensive agents, ACEIs have the most evidence supporting their use in the pediatric population when treating HTN.14 Several studies have been conducted with lisinopril in the pediatric population.15 The drug has FDA approval for pediatric HTN patients aged 6 to 16 years. Adverse effects associated with the use of lisinopril include dry cough, hypotension, hyperkalemia, acute renal failure, and increased serum creatinine.
Angiotensin Receptor Blockers (ARBs): Several studies have demonstrated the efficacy of ARBs in the treatment of HTN in children. Irbesartan has been studied by von Vigier et al and Franscini et al with 20 and 44 patients, respectively.16,17 Both studies found a significant decrease in arterial BP with irbesartan. In general, ARBs tend to be well tolerated by patients, with cough, headache, dizziness, and hyperkalemia reported as the most common adverse effects.
Calcium Channel Blockers: Amlodipine and felodipine have been studied in children aged 6 to 18 years. Amlodipine was found to significantly lower SBP by 6.9 mmHg with the 2.5-mg dose and by 8.7 mmHg with the 5-mg dose.18 The once-daily dosing and low adverse-effect profile make amlodipine an appealing choice. Felodipine was found to be not as efficacious on reducing SBP.19 The most common adverse effects of calcium channel blockers include headache, flushing, dizziness, and peripheral edema.20
Beta-Blockers: Propranolol, the first beta-blocker to be used in the U.S., is the most common beta-blocker used in children.21 In one study, mean SBP declined by 26 mmHg and mean DBP decreased by 20 mmHg using an average dose of 2.5 mg/kg daily.21 Fatigue is most frequently seen at the beginning of therapy and usually resolves in 4 to 6 weeks postinitiation.14
Diuretics: Despite the fact that diuretics are commonly recommended for use in pediatric HTN, to date no large controlled studies have been performed. Hydrochlorothiazide and chlorthalidone have been reported as successful therapies within the pediatric community.11 The adverse effects associated with diuretic use include hyperkalemia, hypernatremia, and gynecomastia in males.22
Role of the Pharmacist
Pharmacists can assist patients by aiding in monitoring BP, promoting lifestyle modifications, verifying therapeutic doses, and working collaboratively with physicians to identify treatment plans that befit each patient. Ultimately, pharmacists serve as a resource to patients through effective counseling, medication therapy management, and their efforts to promote, improve, and monitor successful patient outcomes.
REFERENCES
1. Redwine KM, Acosta AA, Poffenbarger T, et al. Development of hypertension in adolescents with prehypertension. J Pediatr. 2012;160:98-103.
2. Luma GB, Spiotta RT. Hypertension in children and adolescents. Am Fam Physician. 2006;73:1558-1568.
3. Juonala M, Magnussen CG, Berenson GS, et al. Childhood adiposity, adult adiposity, and cardiovascular risk factors. N Engl J Med. 2011;365:1876-1885.
4. National High Blood Pressure Education Program Working
Group on High Blood Pressure in Children and Adolescents. The fourth
report on the diagnosis, evaluation, and treatment of high blood
pressure in children and adolescents. Pediatrics. 2004;114:555-576.
5. Williams CL, Hayman LL, Daniels SR, et al.
Cardiovascular health in childhood: a statement for health professionals
from the Committee on Atherosclerosis, Hypertension, and Obesity in the
Young (AHOY) of the Council on Cardiovascular Disease in the Young,
American Heart Association. Circulation. 2002;106:143-160.
6. McLaughlin VV, Archer SL, Badesch DB, et al. ACCF/AHA
2009 expert consensus document on pulmonary hypertension: a report of
the American College of Cardiology Foundation Task Force on Expert
Consensus Documents and the American Heart Association developed in
collaboration with the American College of Chest Physicians, American
Thoracic Society, Inc, and the Pulmonary Hypertension Association. J Am Coll Cardiol. 2009;53:1573-1619.
7. Riley M, Bluhm B. High blood pressure in children and adolescents. Am Fam Physician. 2012;85:693-700.
8. Torrance B, McGuire KA, Lewanczuk R, McGavock J.
Overweight, physical activity and high blood pressure in children: a
review of the literature. Vasc Health Risk Manag. 2007;3:139-149.
9. Sorof J, Daniels S. Obesity hypertension in children: a problem of epidemic proportions. Hypertension. 2002;40:441-447.
10. Sadowski RH, Falkner B. Hypertension in pediatric patients. Am J Kid Dis. 1996;27:305-315.
11. Lurbe E, Cikova R, Cruickshank JK, et al. Management
of high blood pressure in children and adolescents: recommendations of
the European Society of Hypertension. J Hypertens. 2009;27:1719-1742.
12. FDA Modernization Act, 1997. PL 105-115. Enacted November 21, 1997.
13. Best Pharmaceuticals for Children Act, 2002. PL 107-109. Enacted January 4, 2002.
14. Meyers RS, Siu A. Pharmacotherapy review of chronic pediatric hypertension. Clin Ther. 2011;33:1331-1356.
15. Soffer B, Zhang Z, Miller K, et al. A double-blind,
placebo-controlled, dose-response study of the effectiveness and safety
of lisinopril for children with hypertension. Am J Hyperten. 2003;16:795-800.
16. von Vigier RO, Zberg PM, Teuffel O, et al. Preliminary
experience with the angiotensin II receptor antagonist irbesartan in
chronic kidney disease. Eur J Pediatr. 2000;159:590-593.
17. Franscini LM, von Vigier RO, Pfister R, et al.
Effectiveness and safety of the angiotensin II antagonist irbesartan in
children with chronic kidney disease. Am J Hypertens. 2002;15:1057-1063.
18. Flynn JT, Newburger JW, Daniels SR, et al. A
randomized, placebo-controlled trial of amlodipine in children with
hypertension. J Pediatr. 2004;145:353-359.
19. Trachtman H, Frank R, Mahan JD, et al. Clinical trial of extended-release felodipine in pediatric essential hypertension. Pediatr Nephrol. 2003;18:548-553.
20. Sahney S. A review of calcium channel antagonists in the treatment of pediatric hypertension. Pediatr Drugs. 2006;8:357-373.
21. Griswold WR, McNeal R, Mendoza SA, et al. Propranolol as an antihypertensive agent in children. Arch Dis Child. 1978;53:594-596.
22. Jeunemaitre X, Chatellier G, Kreft-Jais C, et al. Efficacy and tolerance of spironolactone in essential hypertension. Am J Cardiol. 1987;60:820-825.
To comment on this article, contact rdavidson@uspharmacist.com.





