US Pharm. 2014;39(2):HS13-HS16.
ABSTRACT: Cardiac catheterization is a common procedure that is performed in hospitals throughout the United States and worldwide. The procedure can be used for diagnosis and treatment of cardiovascular disease. Pharmacists should be aware of the benefits and risks involved in cardiac catheterization and the associated drug therapy in order to help determine appropriate treatment for patients.
Cardiac catheterization is a common procedure that is performed in hospitals throughout the United States and worldwide. Cardiac catheterization can be used for diagnosis and assessment of cardiovascular disease. Additionally, it can be used therapeutically in order to open an occluded blood vessel and has become an alternative to surgery in some patients.1 Pharmacotherapy plays a large role before, during, and after cardiac catheterization. The goal of this article is to provide practicing pharmacists an overview of the most common reasons for cardiac catheterization, describe the procedure and its associated risks, and discuss the drugs that need be to administered as part of the procedure.
Purpose of Cardiac Catheterization
Cardiac catheterization can be used for the diagnosis and treatment of cardiovascular disease. Diagnostic cardiac catheterization can be utilized to assess the presence and severity of cardiac disease and may help in diagnosis in those patients who present with inconclusive results with noninvasive testing.1 IV radiopaque contrast dye can be administered just prior to the procedure in order to visualize the coronary vessels and can help determine the severity of coronary artery disease (CAD).2 During a right heart catheterization, vascular resistance is measured by attaining pressures in the heart, blood oxygen saturations, and the degree of tricuspid/pulmonic stenosis. This can indicate the presence and severity of pulmonary hypertension.1 During a left heart catheterization, mitral and aortic valvular functions are assessed by measuring the left ventricular pressure, left ventricular function, vascular resistance, and coronary arterial anatomy.1
Therapeutic cardiac catheterization, such as percutaneous coronary intervention (PCI) can be used in patients with occluded arteries or blood vessels. Vascular access is obtained percutaneously via the femoral, brachial, or radial vessels.3 A guidewire is positioned past the stenosis through the lumen and a small angioplasty balloon catheter is advanced over the guidewire. The balloon catheter is then positioned into the artery across the stenotic area and inflated. This inflation expands the stenotic area, thus restoring blood flow.3 The balloon is then deflated, withdrawn, and exchanged for a stent, which remains in place.3
According to the practice guidelines of the American Heart Association (AHA), the American College of Cardiology Foundation (ACCF), and the Society of Cardiovascular Angiography and Interventions (SCAI), PCI is indicated for patients with any of the following cardiac conditions: acute myocardial infarction; stable angina pectoris unrelieved by medical therapy, unstable angina, or angina pectoris following a coronary artery bypass graft (CABG); symptomatic restenosis after previous PCI; and unsuitable or high-risk coronary anatomy that could result in death.4 PCI is contraindicated in the following patients: those with bleeding disorders, multiple PCI restenosis, noncompliance with pre- and post-PCI instructions, and inability to tolerate dual antiplatelet therapy following the procedure.3
Other types of therapeutic cardiac catheterization include those for the treatment of congenital heart defects and for regaining lost hemodialysis access. The use of cardiac catheterization in patients with congenital heart defects may allow for the avoidance of surgery, thus reducing recovery time and duration of hospitalization.5 Cardiac catheterization can also be used for patients with thrombosed hemodialysis access. The use of this approach can prevent the use of temporary hemodialysis catheters, thus allowing for the preservation of venous segments for future access creation.3
Pharmacotherapy
Antithrombotic prophylaxis is an important aspect of diagnostic cardiac catheterization and PCI. Current adjunctive antithrombotic therapy prior to the procedure includes aspirin, a P2Y12 inhibitor (a chemoreceptor for adenosine diphosphate [ADP]), and anticoagulant therapy in combination with a glycoprotein (GP) IIb/IIIa receptor antagonist. Of note, patients undergoing cardiac catheterization for diagnostic purposes with no intervention will require only aspirin and an anticoagulant with GP IIb/IIIa receptor–antagonist therapy. A P2Y12 inhibitor is administered if there is a high likelihood of stent placement during the diagnostic procedure.6 Aspirin produces its antiplatelet effects by inhibiting thromboxane A2 production via irreversible inhibition of cyclooxygenase in platelets.7 Patients already on aspirin therapy and undergoing catheterization should take a total of 81 to 325 mg of aspirin before the procedure. Patients not on aspirin therapy should receive 325 mg of nonenteric-coated aspirin. In all patients, aspirin 81 mg should be continued indefinitely.4
P2Y12 Inhibitors: The P2Y12 inhibitors, also known as the thienopyridines, produce their effect by blocking the ADP-P2Y12 receptors on platelets to inhibit platelet aggregation.7 Several new drugs in this class have been introduced to the market in recent years, including prasugrel and ticagrelor. These agents have been incorporated into the most recent guidelines from the ACCF/AHA/SCAI.4 A loading dose of clopidogrel 600 mg, prasugrel 60 mg, or ticagrelor 180 mg is administered to the patient prior to PCI. Patients with bare-metal or drug-eluting stent placement should continue P2Y12 inhibitor therapy for 1 year with clopidogrel 75 mg daily, prasugrel 10 mg daily, or ticagrelor 90 mg twice daily (TABLE 1).4 Of note, prasugrel should be avoided in patients with history of stroke or transient ischemic attack (TIA) because of the increased risk of bleeding. Patients with a drug-eluting stent and low bleeding risk may be advised to continue therapy up to 15 months. Patients undergoing PCI without stent placement should continue P2Y12 therapy for 14 to 28 days. The combination of aspirin plus a P2Y12 inhibitor is known as dual-antiplatelet therapy (DAPT) and is recommended for all patients to prevent premature thrombosis with both types of stent placement.4

Heparins and Bivalirudin: Anticoagulation, generally with unfractionated heparin (UFH), is also recommended (TABLES 2 and 3).4,7 Additionally, a GP IIb/IIIa receptor antagonist should be coadministered with UFH to reduce the risk of thrombus formation at the puncture site, on the coronary guidewire, and in indwelling cardiac catheters.4 UFH binds to antithrombin and inhibits multiple factors in the blood-clotting cascade.8 UFH should be started preprocedure with a bolus dose of 50 to 70 U/kg followed by a continuous infusion to achieve and maintain therapeutic activated clotting time (ACT) between 200 and 250 seconds, per ACCF/AHA/SCAI guidelines.8,9 When UFH is given without a GP IIb/IIIa receptor antagonist, the bolus dose should be increased to 70 to 100 U/kg followed by continuous infusion to achieve and maintain therapeutic ACT between 250 and 350 seconds.4 UFH should be discontinued immediately after catheterization is complete.7 In some cases, patients are already receiving anticoagulation with enoxaparin, and this medication can be continued in place of UFH, along with a GP IIb/IIIa receptor antagonist.
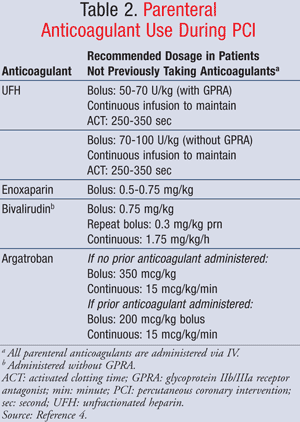
Editor's Note 3/21/14: Bolus dose for Argatroban corrected to 350 mcg/kg (not mg/kg).
If UFH or enoxaparin is contraindicated, as in
heparin-induced thrombocytopenia, bivalirudin alone or argatroban along
with a GP IIb/IIIa receptor antagonist can be substituted.4
Bivalirudin, a direct thrombin inhibitor, should always be administered
alone due to increased risk of bleeding events associated with use in
combination with a GP IIb/IIIa receptor antagonist. Bivalirudin alone is
also recommended in place of UFH in patients at risk for bleeding
events. It is initiated with an IV bolus of 0.75 mg/kg followed by 1.75
mg/kg/h continuous infusion. An additional bolus of 0.3 mg/kg can be
administered if needed. Bivalirudin should be discontinued immediately
after PCI completion.4,7 Of note, if fondaparinux is chosen
as the anticoagulant prior to PCI, it should not be the only
anticoagulant administered. An additional anticoagulant with anti-IIa
activity will need to be administered in combination.4
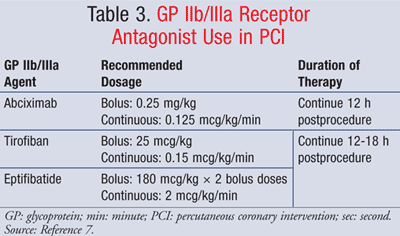
GP IIb/IIIa Receptor Antagonists: A GP IIb/IIIa receptor antagonist should also be started prior to PCI. These drugs block platelet aggregation by inhibiting the cross-linking of platelets at the IIb and IIIa receptor sites.7 There are several options within this class. Recommended GP IIb/IIIa receptor antagonists include abciximab, tirofiban, and eptifibatide (TABLE 3).7 Hospitals generally will not carry all of the GP IIb/IIIa receptor antagonists on formulary due to cost. Efficacy between the individual drugs in the class is comparable.
Abciximab is started at a bolus dose of 0.25 mg/kg followed by 0.125 mcg/kg/min continuous infusion. Tirofiban can also be used in place of abciximab, with a bolus dose of 25 mcg/kg followed by 0.15 mcg/kg/min continuous infusion. Eptifibatide is another option and should be initiated with a bolus of 180 mcg/kg, followed by 2 mcg/kg per minute continuous infusion. A second bolus of 180 mcg/kg is administered 10 minutes after the first bolus. Both tirofiban and eptifibatide require dosage reduction in patients with renal disease.4,10 Abciximab should be continued for 12 hours postprocedure, while eptifibatide or tirofiban should be continued for 12 to 18 hours.7 Other supportive therapies that may be used during catheterization and PCI include IV fluids for hydration and blood pressure stabilization, antihypertensives, vasodilators, and sedatives/anxiolytics.6
Complications
As technology and experience with cardiac catheterization has evolved, the risk of complications associated with the procedure has decreased. The complication rate varies based on diagnostic versus interventional use of catheterization (TABLE 4).3 Complications can range from minor issues with no long-term effects to major complications that require emergency care or surgical intervention and can lead to irreversible damage or death. However, the risk of major complications is under 1%.3 Major complications may include myocardial infarction, cerebrovascular events/stroke, cardiac perforation, and cardiac arrhythmias. Patients with comorbid conditions such as left ventricular dysfunction, valvular heart disease, a prior CABG, congestive heart failure, and renal insufficiency are at higher risk of complications.
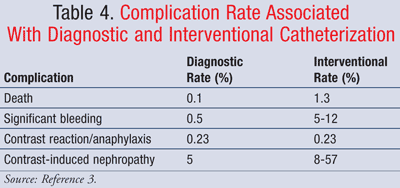
Local vascular complications are the most common type of complication associated with cardiac catheterization. They are also the single greatest source of morbidity. Some of these complications include vessel thrombosis, distal embolization, and bleeding. Vessel thrombosis and embolization usually occur when a piece of plaque or a clot imperceptibly breaks free after contact with the catheter. Poorly controlled bleeding at the entry site is associated with a poorly placed puncture, vessel lacerations, excess anticoagulant use, and unskillful technique with a closure device. This can lead to hemorrhage and hematoma formation, usually within 12 hours of the procedure, and may require surgical interventions and blood transfusions. Patients may also experience false aneurysms or arteriovenous fistulas that can appear days to weeks after the procedure and require surgical interventions.10
While an important and required part of cardiac catheterization, the use of contrast can also lead to complications such as allergic or anaphylactic reactions and nephropathy in some patient populations. Those with penicillin, seafood, or atopic allergies or past reactions to contrast are at a higher risk for complications. Premedicating these patients with glucocorticoids, H1 antihistamines, and H2 antihistamines can decrease the risk of such complications. Additionally, ionic contrast should be avoided and replaced with low- or iso-osmolar nonionic contrast.
Certain patient populations may also experience contrast-related nephropathy, including those with a history of diabetes, renal failure, and volume depletion. Patients who receive larger volumes of contrast are also at increased risk for nephrotoxicity. These patients should receive IV hydration pre- and postprocedure, and the amount of contrast used during the procedure should be minimized. Serum creatinine should be monitored in these patients for 3 to 5 days after catheterization. N-acetylcysteine can also be administered pre- and postprocedure as a protective strategy; however, clinical studies show questionable value.6,10
Conclusion
Cardiac catheterization is a common procedure that is generally well tolerated. Pharmacists should be aware of the benefits and risks associated with the procedure and associated drug therapy. Pharmacists in the community setting are in a position to answer patients’ questions regarding the cardiac catheterization procedure and the need for adherence with DAPT following hospital discharge. Pharmacists in hospital settings can play a role in guiding drug choices and developing a drug formulary of antiplatelets and anticoagulants for the procedure. These pharmacists can also ensure that patients are discharged on appropriate antiplatelet therapy to prevent future cardiovascular events.
REFERENCES
1. Lange RA, Hillis LD. Chapter 17. Cardiovascular testing. In: DiPiro JT, Talbert RL, Yee GC, et al, eds. Pharmacotherapy: A Pathophysiologic Approach. 8th ed. New York, NY: McGraw-Hill; 2011.
2. Scanlon P, Faxon D, Smith S Jr, et al. ACC/AHA
guidelines for coronary angiography: executive summary and
recommendations. A report of the American College of Cardiology/American
Heart Association Task Force on Practice Guidelines (Committee on
Coronary Angiography) developed in collaboration with the Society for
Cardiac Angiography and Interventions. Circulation. 1999;99:2345-2357.
3. Kern MJ. Interventional Cardiac Catheterization Handbook. 3rd ed. Philadelphia, PA: Elsevier Saunders; 2013.
4. Levine GN, Bates ER, Blankenship JC, et al. 2011
ACCF/AHA/SCAI guideline for percutaneous coronary intervention: a report
of the American College of Cardiology Foundation/American Heart
Association Task Force on Practice Guidelines and the Society for
Cardiovascular Angiography and Interventions. Circulation. 2011;124:e574-e651.
5. Krasemann T, Qureshi S. Treatment of congenital heart defects using interventional catheterization. Cardiology. 2007;26:97-106.
6. Bashore TM, Balter S, Barac A, et al. 2012 American
College of Cardiology Foundation/Society for Cardiovascular Angiography
and Interventions expert consensus document on cardiac catheterization
laboratory standards update. J Am Coll Cardiol. 2012;59:2221-2305.
7. Spinler SA, De Denus S. Chapter 24. Acute coronary syndromes. In: Wells BG, ed. Pharmacotherapy: A Pathophysiologic Approach. 8th ed. New York, NY: McGraw-Hill; 2011.
8. Zehnder JL. Chapter 34. Drugs used in disorders of coagulation. In: Katzung BG, Masters SB, Trevor AJ, eds. Basic & Clinical Pharmacology. 12th ed. New York, NY: McGraw-Hill; 2012.
9. O’Gara PT, Kushner FG, Ascheim DD, et al. 2013 ACCF/AHA
guideline for the management of ST-elevation myocardial infarction: a
report of the American College of Cardiology Foundation/American Heart
Association Task Force on Practice Guidelines. Circulation. 2013;127:e362-e425.
10. Baim DS, Simon DI. Chapter 3. Complications and the optimal use of adjunctive pharmacotherapy. In: Baim DS, ed. Grossman’s Cardiac Catheterization, Angiography, and Intervention. 7th ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2006.
To comment on this article, contact rdavidson@uspharmacist.com.





