US Pharm. 2014;39(5):39-43.
ABSTRACT: Varicella-zoster virus is responsible for causing a primary varicella infection (chickenpox) and a secondary herpes zoster infection (shingles). Although varicella typically manifests as a mild disease in otherwise healthy children, it can also manifest as a moderate-to-severe disease, most notably in immunocompromised and adult hosts. Acyclovir is the antiviral agent of choice for the management of varicella infections. However, in acyclovir-resistant varicella infections, foscarnet is the recommended antiviral agent for use. Routine vaccination with Varivax has been very effective in reducing chickenpox incidence in the United States; it confers immunity and also reduces clinical manifestation of the illness.
Varicella-zoster virus (VZV) is a double-stranded DNA virus that belongs to the Alpha-herpesvirinae subfamily of the Herpesviridae family of eight herpes viruses. It is responsible for an infection that manifests as separate illnesses in two phases of the human life cycle: 1) a primary infection, varicella (chickenpox), typically in childhood; and 2) a secondary herpes zoster (HZ) infection (shingles) through reactivation of the latent virus in the central nervous system (CNS), typically in the elderly. This article aims to review the epidemiology, clinical manifestations, complications, management, and prevention of childhood chickenpox, including recent advances in therapeutic management of varicella and CDC recommendations.
Epidemiology
Varicella is a highly contagious infection that occurs only in humans, with a secondary attack rate of up to 90% in susceptible (seronegative) household contacts.1-3 Prior to the development of the varicella vaccine in 1995, chickenpox was reported to be endemic in the United States, with approximately 4 million cases, resulting in about 11,000 hospitalizations and 100 deaths per year.1,4,5 In temperate climates, it exhibits an annual epidemic trend, peaking in late winter to spring.1,2 Varicella is primarily a disease of childhood with approximately 90% of the cases, 67% of varicella hospitalizations, and about 50% of related deaths reported in children.2-4,6 Historically, children between the ages of 1 and 4 years accounted for 39% of cases, while children aged 5 to 9 years and adults ≥20 years accounted for 38% and 7%, respectively (National Health Interview Survey data, 1990-1994).5
Transmission and Pathophysiology
Varicella is primarily transmitted human-to-human via the nasopharynx or conjunctiva, through mucosal inoculation with droplets and aerosols from VZV- or HZ- infected persons. The infectious period occurs within 1 to 2 days before the onset of rash (through nasopharyngeal secretions) or within 5 to 7 days after appearance of rash (through skin lesions). This period of contagiousness can be extended in immunocompromised persons.1-2,7,8 The virus has an average incubation period of 14 to 16 days (range: 10-21 days).1-3,7
After inoculation, there is a primary viremia phase where the virus disseminates and multiplies in the viscera and reticuloendothelial tissues. A secondary viremia phase occurs at about 14 days post infection where there is a viral spread to the nasopharyngeal surface and the skin, leading to the appearance of typical maculopapular vesicular rashes.2,3,7 These vesicular rashes are highly concentrated with the virus and are very contagious until crusted. The virus can also infiltrate the CNS and become dormant in the dorsal root and cranial nerve ganglia, to be reactivated at a later period in the human life cycle as HZ infection.7 While there is a favorable outcome with the primary infection, children with underlying HIV infection showed a higher incidence of the secondary zoster infection in early stages of life.7,8
Clinical Manifestations and Laboratory Findings
Varicella is characterized by a generalized itchy, blister-like rash, usually associated with fever, headache, malaise, myalgia, and loss of appetite.1-3,7 The rash first appears as macules, then progresses through the papular and vesicular stages, before crusting over within a short period of time (within 24-48 hours). The lesions typically start on the face and scalp and then progress to the trunk and extremities, appearing throughout the body at different stages of development.3,7
Varicella infection can lead to secondary complications from either viruses or bacteria. The most common complication is secondary bacterial infections, especially infections caused by group A beta-hemolytic streptococci or staphylococci. These infections can manifest as impetigo, cellulitis, erysipelas, and skin lesions from staphylococcal exfoliative toxin.3,7 Extracutaneous secondary complications include pneumonia, osteomyelitis, myocarditis, sepsis, necrotizing fasciitis, and CNS involvement, which is the most common in children.
CNS involvements range from mild complications like cerebellar ataxia to serious complications such as Reye syndrome, an acute illness characterized by rapidly progressive encephalopathy and fatty degra-dation of the liver. The syndrome presents as severe vomiting and confusion, encephalitis, meningitis, and vasculitis, which can lead to stroke.9 Varicella infection is usually more severe in immunocompromised patients with an increased risk of dissemination and infection to other organs.3,8,10
Laboratory testing is not necessary for most cases of varicella infection, but it is routinely used for suspected disseminated infection. It may also be utilized in atypical cases. VZV antibodies can be detected via direct fluorescence (DF). The virus from skin lesions, or cerebrospinal fluid in cases of CNS involvement, can be detected via polymerase chain reaction (PCR).2,7,10
Serologic testing for VZV antibodies is not recommended for the diagnosis of VZV but can be useful in determining if an individual is at risk.2 A positive immunoglobulin G (IgG) and IgM suggest a primary VZV infection. A positive IgG, which can persist for years, and a negative IgM imply vaccination or a primary infection, conferring a protective immunity against reinfection.2 A negative IgG and negative IgM indicate the absence of prior exposure to VZV and nonimmunity. A negative reading does not rule out a VZV infection. IgM commercial assays are prone to both false-positive and false-negative results; IgG commercial assays are prone to false-negative results (10%-20%).2 Retesting should be done in 2 to 3 weeks if a negative reading is obtained.
Treatment
Strategies for the management of varicella infection are aimed at symptom management and prevention of secondary complications.3,7 Treatment options include supportive measures, antiviral therapy, varicella-zoster immune globulin (human) (VZIG or VariZIG), and management of secondary complications. Due to varicella’s high contagiousness, all persons admitted as inpatients with VZV or HZ infection should be placed on airborne plus contact precaution to reduce risk of transmission.7,10 All close contacts of infected persons without evidence of immunity should receive immunization or VariZIG, preferably within 72 hours, but up to 5 days, post exposure.10
Symptomatic Measures: As a self-limiting disease, varicella infection in otherwise healthy children can often be treated solely with symptomatic management and prevention of secondary complications. Good hygiene (e.g., daily baths, preferably with antibacterial soap) and thorough skin care are important for the prevention of secondary bacterial infections.1 The child’s fingernails should also be cut short in an effort to prevent secondary bacterial skin infections. Further measures of supportive therapy include OTC medications such as acetaminophen for fever, topical agents that include calamine, daily lukewarm soaks with colloidal oatmeal, and oral or topical antihistamines; the latter three will provide management and a soothing relief of the pruritus.1-3 Aspirin should be avoided in varicella-infected children due to its association with Reye syndrome; acetaminophen should be the antipyretic of choice in children, with appropriate dosing (TABLE 1).1,7

Antiviral Therapy: In otherwise healthy children <12 years of age, varicella is often of benign manifestation and self-limiting; routine antiviral therapy may not provide much clinical benefit and is therefore not routinely recommended in this population. But it may help prevent secondary complications in at-risk populations including immunocompromised children and children on immunosuppressive therapy.1-3,7,10-13 Vidarabine and interferon-alfa were the initial antiviral agents studied in at-risk populations with varicella infections, but were later replaced by acyclovir, a safer and more effective therapy.2
Acyclovir, an antiviral agent used in the management of varicella, is available in both oral and IV formulations. It is an acyclic analogue of guanosine that must be phosphorylated by viral thymidine kinase and subsequently by host cell enzymes into its active triphosphate form for its antiviral activity through inhibition of viral DNA polymerase (FIGURE 1). Acyclovir is the antiviral agent of choice in the treatment of varicella and should be initiated within 24 hours of the onset of rash.1-3,7 It has been proven to shorten both the duration and severity of chickenpox by decreasing viral shedding and the formation of new lesions, as well as by accelerating lesion healing in both healthy and immunologically compromised children.1-3,7
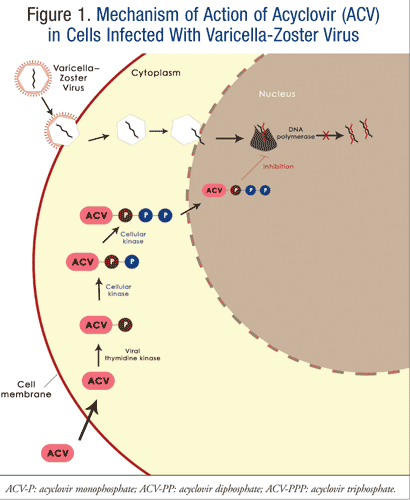
Alternative antiviral agents for varicella include valacyclovir, the L-valine ester of acyclovir that is converted to acyclovir after oral administration, and famciclovir. Both agents are clinically used for varicella infections; however, lack of sufficient clinical efficacy trials prevents the use of these drugs as first-line agents for this indication. Additionally, famciclovir is not FDA-approved for varicella.2 Valacyclovir has approximately 55% bioavailability and therefore requires less frequent dosing in contrast to oral acyclovir, which has a low oral bioavailability; only about 10 to 20% of the oral acyclovir dose becomes bioavailable.
Treatment with antiviral agents is optional in otherwise healthy children ≤12 years of age, but the American Academy of Pediatrics (AAP) recommends antiviral therapy in adolescents (children >12 years of age); children >12 months who are on long-term salicylate therapy; children with chronic pulmonary or skin problems; and children receiving aerosolized corticosteroids. IV acyclovir is recommended to prevent or treat disseminated varicella in immunocompromised children, including those receiving oral corticosteroids ≥2 mg/kg/day (or prednisolone 20 mg/day equivalent) for ≥2 weeks; in HIV-infected persons; and in those receiving immunosuppressive therapy. IV therapy unfortunately requires hospitalization, posing a risk of nosocomial infections. Alternatively, high-dose oral acyclovir or valacyclovir may be used for mildly immunocompromised patients.7
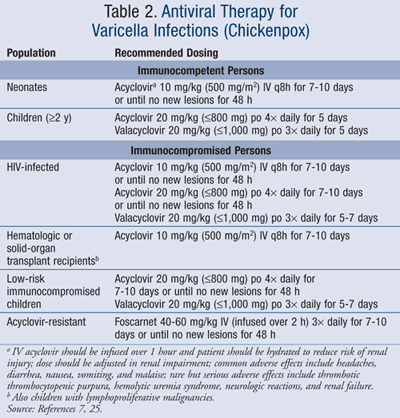
TABLE 2 summarizes the recommended agents and dosing for management of varicella infection in children and adolescents.7 Acyclovir resistance has only been reported in rare cases, often in immunocompromised patients. It is more often reported in HIV-infected persons, in whom acyclovir may be used long term, thereby creating a selection for thymidine kinase–negative or mutant virus.7,13,14 In this case, the virus fails to produce thymidine kinase, or produces a genetically altered form of the enzyme, which is necessary for the initial phosphorylation of the drug.14 Acyclovir-resistance can be clinically defined when patients fail to respond to acyclovir therapy.2 Thus, children with acyclovir-resistant varicella infection should be treated with foscarnet, a pyrophosphate analogue that, unlike acyclovir, does not require further phosphorylation for activity.15 It has direct action on viral DNA polymerase and acts by selectively inhibiting pyrophosphate–binding sites on viral DNA polymerase.15 Moreover, foscarnet is only available via IV route.
Prevention and Prophylaxis
Varicella Vaccine: Varivax (Merck & Co., Inc.), a single-agent vaccine containing a live, attenuated (Oka strain) virus, is recommended to prevent varicella infection in healthy persons >12 months of age who do not have any contraindication to the vaccine (TABLE 3).16,17 Routine varicella vaccination occurs between ages 12 and 18 months, with the second dose administered between ages 4 and 6 years.16 ProQuad (Merck & Co., Inc.), a measles/mumps/rubella/varicella (MMRV) combination vaccine, is licensed for use in persons aged 12 months to 12 years with a similar dosing schedule.18

Nonimmunized adolescents should receive two doses of varicella vaccine, with a minimum interval of 4 weeks between doses. The vaccine is safe and well tolerated in healthy children and adolescents. Common adverse effects of Varivax include reactions at the injection-site (i.e., pain, redness, swelling, rash), fever, and generalized rash.19
Since its license, Varivax has been highly effective in reducing incidence of varicella.6,17,19 According to a 14-year prospective study of varicella incidence and long-term effectiveness of the varicella vaccine by Kaiser Permanente Vaccine Study Center, incidence of varicella decreased by about 10-fold from the prevaccine era.19 Effectiveness of Varivax is reported as 85% to 90%.17,20,21 Varivax can also be utilized for postexposure prophylaxis to prevent or reduce the severity of the illness; efficacy is maximized if administered within 72 hours of exposure.10,16-20,22
Varicella-Zoster Immune Globulin: VariZIG, a purified human IgG, provides passive immunity to persons at high risk of severe chickenpox (TABLE 4); it can also prevent or ameliorate clinical varicella if administered within 10 days of exposure.23 Factors to consider before utilizing VariZIG include: 1) lack of evidence of immunity, 2) exposure likely to result in infection, and 3) greater risk for complications than in the general population.23

VariZIG should be administered ideally within 96 hours (up to 10 days) after exposure. VariZIG should not be given concurrently with Varivax. Considering that the passive transfer of immunoglobulin could interfere with the efficacy of live attenuated vaccines such as MMRV, vaccination should be deferred until ≥5 months after VariZIG administration.23 Patients should be monitored for 28 days after VariZIG exposure for signs and symptoms of varicella infection, and antiviral therapy should promptly be initiated if these occur. Common adverse effects of VariZIG include reactions at the injection site and headaches. The recommended dose of VariZIG is 125 IU/10 kg of body weight (≤625 IU). For patients weighing ≤2.0 kg, administer 62.5 IU, and 125 IU for patients weighing 2.1 to 10.0 kg.23
Antiviral Postexposure Prophylaxis: Where VariZIG is not indicated, high-dose oral acyclovir 20 mg/kg/dose (≤800 mg) 4 times daily for 7 days or Varivax may be used as postexposure prophylaxis; efficacy is maximized if treatment is initiated within 24 hours of symptoms.7
Conclusion
Varicella infection (chickenpox) is a common illness of childhood. In otherwise healthy children ≥12 years old, the illness is often mild and can be managed with supportive therapy. If required, antiviral therapy can be initiated within 24 hours of the onset of rash. The most effective way to prevent a varicella outbreak is through universal immunization with varicella vaccine. This strategy not only prevents a severe or complicated form of the disease in healthy children, but it also serves to protect at-risk children including those who are immunologically compromised and cannot receive live vaccines. Furthermore, post exposure can be managed with varicella-zoster immunoglobulin preparation, an antiviral agent, or the vaccine.
REFERENCES
1. Sagraves R, Morrison Y. Varicella-zoster virus: recent therapeutic advances. J Pediatr Health Care. 1995;9:81-86.
2. Arvin AM. Antiviral therapy for varicella and herpes zoster. Semin Pediatr Infect Dis. 2002;13:12-21.
3. Heininger U, Seward JF. Varicella. Lancet. 2006;368:1365-1376.
4. Wharton M. The epidemiology of varicella-zoster virus infections. Infect Dis Clin N Am. 1996;10:571-581.
5. American Academy of Pediatrics. Committee on Infectious Diseases. Varicella vaccine update. Pediatrics. 2000;105:136-141.
6. Nguyen HQ, Jumaan AO, Seward JF. Decline in mortality
due to varicella after implementation of varicella vaccination in the
United States. N Engl J Med. 2005;352:450-458.
7. Cohen JI, Brunell PA, Straus SE, Krause PR. Recent advances in varicella-zoster virus infection. Ann Intern Med. 1999;130:922-932.
8. Gershon AA, Mervish NF, LaRussa PF, et al.
Varicella-zoster virus infection in children with underlying human
immunodeficiency virus infection. J Infect Dis. 1997;176:1496-1500.
9. Belay ED, Bresee JS, Holman RC, et al. Reye’s syndrome in the United States from 1981 through 1997. N Engl J Med. 1999;340:1377-1382.
10. Pergam SA, Limaye AP. Varicella zoster virus (VZV) in solid organ transplant recipients. Am J Transplant. 2009;9(suppl 4):108-115.
11. Zuckerman RA, Limaye AP. Varicella zoster virus (VZV) and herpes simplex virus (HSV) in solid organ transplant patients. Am J Transplant. 2013;13(suppl 3):55-66.
12. Roderick M, Finn A, Ramanan AV. Chickenpox in the immunocompromised child. Arch Dis Child. 2012;97:587-589.
13. Margo KL, Shaughnessy AF. Antiviral drugs in healthy children. Am Fam Physician. 1998;57:1073-1077.
14. Boivin G, Edelman CK, Pedneault L, et al. Phenotypic
and genotypic characterization of acyclovir-resistant varicella-zoster
viruses isolated from persons with AIDS. J Infect Disease. 1994;170:68-75.
15. Gérard L, Salmon-Céron D. Pharmacology and clinical use of foscarnet. Int J Antimicrob Agents. 1995;5:209-217.
16. Marin M, Giiris D, Chaves SS, et al. Prevention of
varicella: recommendations of the Advisory Committee on Immunization
Practices (ACIP). MMWR Recomm Rep. 2007;56:1-40.
17. Vasquez M, LaRussa PS, Gershon AA, et al. The effectiveness of the varicella vaccine in clinical practice. N Engl J Med. 2001;344:955-960.
18. ProQuad (measles, mumps, rubella and varicella virus
vaccine live) package insert. Whitehouse Station, NJ: Merck & Co,
Inc; February 2014.
19. Skull SA, Wang EEL. Varicella vaccination—a critical review of the evidence. Arch Dis Child. 2001;85:83-90.
20. Baxter R, Ray P, Tran TN, et al. Long-term effectiveness of varicella vaccine: a 14-year, prospective cohort study. Pediatrics. 2013;131:1389-1396.
21. Grose C. Varicella vaccination of children in the United States: assessment after the first decade 1995-2005. J Clin Virol. 2005;3:89-95.
22. Bate J, Chisholm J, Heath PT, et al. PEPtalk: postexposure prophylaxis against varicella in children with cancer. Arch Dis Child. 2011;96:841-845.
23. Updated recommendations for Use of VariZIG—United States, 2013. Centers for Disease Control and Prevention (CDC). MMWR Morb Mortal Wkly Rep. 2013;62:574-576.
24. Epocrates Rx Online [online database]. San Mateo, CA:
Athenahealth; 2014. www.online.epocrates.com. Accessed February 9, 2014.
25. Micromedex Drug Information [online database]
www.micromedexsolutions.com. Greenwood Village, CO: Truven Health
Analytics; 2014. Accessed February 9, 2014.
To comment on this article, contact rdavidson@uspharmacist.com.





