US Pharm. 2014;39(6):HS2-HS7.
ABSTRACT: Diabetic foot disease is a broad spectrum of foot disorders involving the cutaneous, soft-tissue, and osseous structures. Acute conditions may manifest as deeply infected wounds with extensive structural damage necessitating immediate medical attention to avoid amputation. Peripheral neuropathy, tissue ischemia, and foot biomechanics play a role in the development of acute ulcerative conditions. Some common offending agents in acute infections include gram-positive cocci and gram-negative bacilli. Early recognition and treatment by a multidisciplinary healthcare team are necessary for complete therapeutic resolution in patients who present with acute conditions. Pharmacists play an integral role in assessing the appropriateness of therapeutic regimens, as well as in educating patients upon discharge in order to prevent exacerbation of the condition.
Diabetic foot disease (DFD) is a spectrum of disorders involving the cutaneous, soft-tissue, and osseous structures of the foot. This includes the development of foot ulcers and infections that may develop into limb-threatening conditions. Early recognition and management of acute DFD are critical for the prevention of disease progression and amputation. This review is intended to educate pharmacists regarding the proper management of acute DFDs according to current guidelines.
Epidemiology
The CDC has estimated that 25.8 million people in the United States have diabetes, and the prevalence of foot ulcers in this population is 12%.1,2 Foot ulcers account for the greatest percentage of hospital admissions among diabetes patients and are the leading cause of nontraumatic lower-extremity amputations in the U.S.2 The 20-year cumulative incidence of lower-limb ulcers in patients with type 1 diabetes was estimated to approach 10%, with 5% of cases requiring amputation.3 Mortality following amputation was estimated to range from 13% to 40% at 1 year, 35% to 65% at 3 years, and 39% to 85% at 5 years.3 Aside from the great degree of morbidity and substantial emotional distress, acute DFD is associated with a considerable financial burden. A Swedish study estimated costs to range from $18,000 to $34,000 (with amputation) from time of diagnosis to healing or death.4
Risk Factors and Pathophysiology
Diabetic Foot Ulcers (DFUs): The development of DFUs involves many etiological factors that are important to monitor in diabetes patients. Peripheral neuropathy (PN), which is present to some degree in >50% of diabetes patients aged >60 years, is one of the most important factors in the development of DFUs.5 Profound PN leads to loss of protective sensation, and consequently to increased vulnerability to physical and thermal trauma. PN has been shown to increase the risk of DFU development sevenfold.3 Irregular mechanical loading of the foot that results in excessive plantar and heel pressure is another causative factor.6,7 Biomechanics, such as structural foot abnormalities (calluses and hammertoes) and reduced joint mobility, further alters the mechanical loading of the plantar surface and increases the risk of DFUs.7 Physical trauma, especially when repetitive, also increases the risk. In one study involving 669 subjects with DFUs, 21% of ulcers were linked to rubbing from footwear, 11% to injuries, 4% to cellulitis complicating tinea pedis, and 4% to self-inflicted trauma (e.g., cutting toenails).8
Peripheral arterial disease (PAD), which is a major contributor to the development of DFU, is approximately twice as prevalent in diabetes patients.6 PAD reduces blood flow and ulcer healing, leading to tissue ischemia and a worsened DFU prognosis.9 One large study found that the ankle-brachial index—a common assessment of vascular dysfunction in PAD diagnosis—was strongly linked to the risk of foot ulceration.10 Management of fasting plasma glucose is also critical in diabetes patients; uncontrolled hyperglycemia has been shown to interfere with wound healing, resulting in endothelial dysfunction and development of PN.7 One study showed that long diabetes duration (>10 years) and poor glycemic control (A1C >9%) were associated with DFUs.10
Diabetic Foot Infections (DFIs): DFIs develop in the presence of an ulcer or traumatic injury. A DFI typically begins superficially and spreads to deeper structures, leading to deep abscess, chronic ulcers, cellulitis, and osteomyelitis. Acute soft-tissue infections usually involve necrotizing fasciitis, malodorous discharge, and tissue loss; the presence of cyanotic discoloration usually signifies vascular interference.7 Acute DFIs warrant immediate therapeutic intervention to prevent gangrene and amputation. Etiological microorganisms of DFIs include gram-positive cocci (GPC), gram-negative bacilli (GNB), and anaerobic organisms. Staphylococcus aureus and beta-hemolytic streptococci are the most common organisms in mild-to-moderate DFIs; they most commonly occur in patients who have not had antibiotics in the past month.11 Chronic infections are typically mixed GPC and GNB. The presence of ischemic and necrotic tissue is associated with obligate anaerobes. Methicillin-resistant S aureus and Pseudomonas aeruginosa infections usually occur in patients with chronic hospitalization and drug-resistant conditions.11,12
Clinical Presentation and Diagnostic Approach
Because acute DFD can rapidly progress to a limb-threatening condition, it is imperative for clinical pharmacists and healthcare professionals (HCPs) to assess the extent of disease and develop an appropriate patient-directed treatment algorithm. Early recognition of the patient’s acute condition through the use of proper diagnostic tools is essential. Upon patient presentation, the following must be assessed: physical presentation of the foot ulcer, presence of wound infection, tissue ischemia, and neuropathy or neuropathic osteoarthropathy.6,13
The Meggitt-Wagner classification is a well-known and validated system that HCPs can use to stage DFUs.14 This classification has been shown to be a good predictor of ulcer prognosis and determinant of whether amputation is warranted.15 MRI is the gold standard for detection of soft-tissue lesions, whereas x-ray may be appropriate when osseous involvement is suspected.11,16
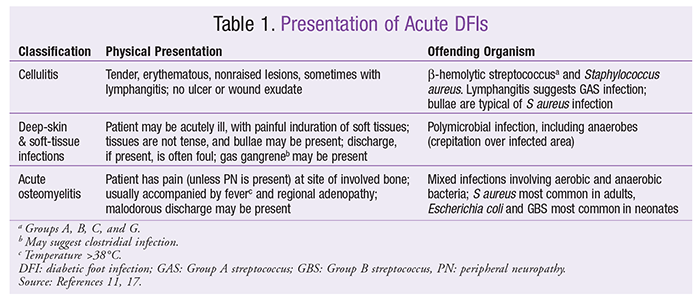
Rapid progression of an acute DFI may lead to a limb-threatening condition; thus, early recognition of the infection’s severity is important. A patient with a wound infection usually presents with classic signs and symptoms of inflammation (tenderness, erythema, warmth, and/or pain).11 A purulent, potentially malodorous discharge from the ulcer suggests a more extensive infection.11 Acute DFI typically manifests as cellulitis, deep-skin and soft-tissue infections, or acute osteomyelitis (
TABLE 1
).17 According to the most recent Infectious Diseases Society of America guideline, a tissue culture must be obtained before empirical therapy is initiated, and the wound must be appropriately cleaned and debrided before specimens are obtained.11 A patient presenting with a local infection associated with signs of systemic inflammatory response syndrome must be hospitalized for critical care.11 Blood cultures are recommended for patients with signs of acute systemic illness.11
In the presence of neuropathy and insufficient vascularization, some of the classic signs and symptoms of a foot infection (pain and erythema) may be false-negative, so evaluation for tissue ischemia and PN is critical in acute DFD patients.11 The absence of pedal pulses may indicate lower-extremity ischemia.18 The patient’s vascular status should be determined via diagnostic tools such as Doppler ultrasound and tissue percutaneous oximetry.16 Tissue ischemia can be diagnosed as tissue percutaneous oximetry <30 mmHg or ankle blood pressure <50 mmHg.15 The ankle-brachial systolic pressure index is an unreliable assessment of ischemia in diabetes patients.18 The 10-g Semmes-Weinstein monofilament test and the Neuropathy Disability Score are useful diagnostic tools for neurologic examination in the clinical setting, and both may be used.19
Treatment
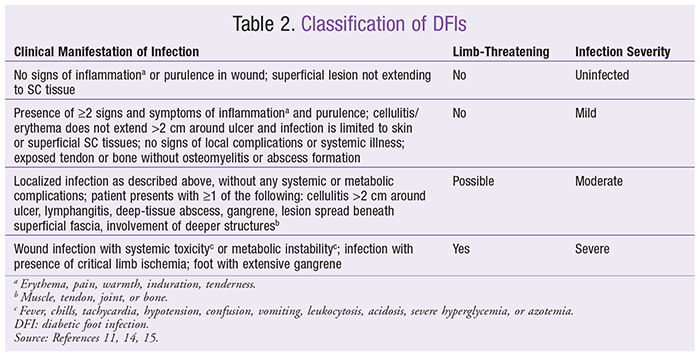
Most hospitalizations in acute DFD are due to acute infection of a foot ulcer or exacerbation of a previous DFI.5 Because of the complexity of an acute DFD and its potentially life-threatening exacerbations, management requires prompt action by a multidisciplinary team.11 The healthcare team must first assess the foot disease to classify the wound and determine whether there is an infection. The DFD classification in
TABLE 2
, which is based on often-used and validated classification systems for DFIs, is useful for determining whether an infection is limb-threatening (severe) or non–limb-threatening (mild-to-moderate).11 Uninfected wounds should not be cultured or treated with antibiotics. Mild infections are usually treated with oral antibiotics on an outpatient basis for 1 to 2 weeks; moderate infections usually involve either oral or parenteral antibiotics for 2 to 4 weeks.11 Immediate hospitalization is warranted for all patients whose infection is severe or complicated by critical limb ischemia.11
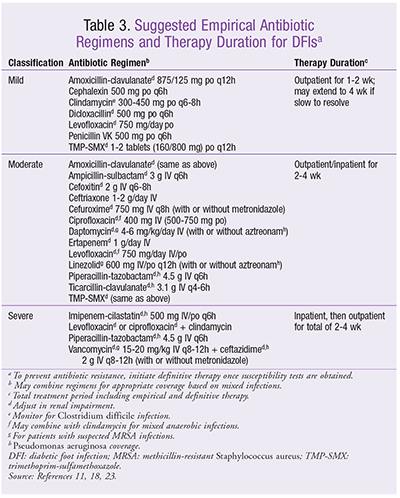
Upon hospitalization, it is important to keep the patient medically stabilized (fluids, electrolytes, insulin, etc.). After site-appropriate deep culturing, empiric parenteral therapy should be initiated with broad-spectrum antibiotics (
TABLE 3
) and must be reevaluated daily. Once cultures are analyzed, more definitive therapy should be initiated. When possible, the patient may be switched to oral therapy for a total of 2 to 4 weeks of antibiotic therapy (including initial parenteral therapy). If there is no improvement or the condition worsens, reculturing and broadening of antibiotic therapy should be considered.11 Pharmacists play an integral role in this process by working with other HCPs to select the most appropriate empirical therapy, dosage, and route of administration, as well as by monitoring the pharmacodynamic and pharmacokinetic properties of these medications.
Urgent surgical consultation may be necessary when there is suspicion of limb ischemia and an extensive limb-threatening infection that is nonresponsive to parenteral antibiotic therapy. In the presence of critical limb ischemia and in patients with a history of PAD, revascularization may be initiated after recognition of the infection (within 1-2 days).11 Amputation should be considered in patients with necrotizing fasciitis, gas gangrene, extensive soft-tissue loss, or severe osteomyelitis.20 Amputation is reserved for prevention of a life-threatening condition, and the decision to perform this procedure should be made only after a thorough patient-provider discussion.18
Many studies suggest that it is important to monitor blood glucose levels in hospitalized patients.11,18,21 In particular, because hyperglycemia is associated with poor wound healing, a correction would help eradicate the infection and increase the likelihood of a favorable outcome.11,21
Offloading techniques should be employed in all patients to prevent iatrogenic foot complications. Wound care, which includes incision, drainage, and debridement, should be performed on DFD patients to enable optimal healing of the ulcer.11 Sharp debridement of devitalized tissue may be necessary in noninfected neuropathic wounds.18 Dressings should be applied to promote wound healing, and appropriate selection is based on the type of wound: alginates, foams, or hydrocolloids for exudative wounds; films for dry wounds; hydrogels and saline gauze for dry or necrotic wounds.11 Granulocyte colony-stimulating factors such as Apligraf and Dermagraft have been shown to reduce the need for operative procedures and may be used as adjunctive therapy for wound closure in noninfected, nonischemic patients.11
Follow-up and Prevention
Appropriate discharge procedures should be in place to reduce the risk of readmittance for an acute exacerbation of DFD. Hospitalized patients who are discharged and initiated on oral antibiotics must complete their regimen and follow up as directed so that therapeutic improvement of the condition can be evaluated. Upon discharge, the pharmacist must counsel patients on the importance of therapy adherence and potential side effects. Patients with PAD must optimize their therapy and be educated about risk factors (lipid abnormalities, smoking) in order to prevent future acute DFD. Pharmacists should also discuss foot-trauma preventive measures, such as daily inspections, appropriate footwear, and prompt reporting of any lesions, with patients. Blood glucose monitoring is critical for preventing acute DFD. In the United Kingdom Prospective Diabetes Study, a 1% mean reduction in hemoglobin A1C was associated with a 25% reduction in microvascular complications and a 36% reduction in amputations.22
Pharmacists must counsel patients on appropriate glucose monitoring, instill an attitude of medication adherence, and work with physicians to optimize the patient’s diabetes therapy. Therapeutic lifestyle changes, such as individualized diet and exercise, should be discussed with the patient. Lastly, it is important to involve the patient’s family and caregivers to facilitate a dynamic treatment approach.
REFERENCES
1. CD
C. National Diabetes Fact Sheet: National Estimates and General Information on Diabetes and Prediabetes in the United States, 2011. Atlanta, GA: U.S. Department of Health and Human Services, CDC; 2011.
2. Currie CJ, Morgan CL, Peters JR. The epidemiology and cost of inpatient care for peripheral vascular disease, infection, neuropathy, and ulceration in diabetes. Diabetes Care. 1998;21:42-48.
3. Reiber GE, Lipsky BA, Gibbons GW. The burden of diabetic foot ulcers. Am J Surg. 1998;176(suppl 2A):5S-10S.
4. Tennvall GR, Apelqvist J, Eneroth M. Costs of deep foot infections in patients with diabetes mellitus. Pharmacoeconomics. 2000;18:225-238.
5. Singh N, Armstrong DG, Lipsky BA. Preventing foot ulcers in patients with diabetes. JAMA. 2005;293:217-228.
6. Younes NA, Ahmad AT. Diabetic foot disease. Endocr Pract. 2006;12:583-592.
7. Altman MI, Altman KS. The podiatric assessment of the diabetic lower extremity: special considerations. Wounds. 2000;12(6 suppl B):64B-71B.
8. Macfarlane RM, Jeffcoate WJ. Factors contributing to the presentation of diabetic foot ulcers. Diabet Med. 1997;14:867-870.
9. Gregg EW, Sorlie P, Paulose-Ram R, et al. Prevalence of lower-extremity disease in the US adult population ≥40 years of age with and without diabetes: 1999-2000 National Health and Nutrition Examination Survey. Diabetes Care. 2004;27:1591-1597.
10. Lavery LA, Armstrong DG, Vela SA, et al. Practical criteria for screening patients at high risk for diabetic foot ulceration. Arch Intern Med. 1998;158:157-162.
11. Lipsky BA, Berendt AR, Cornia PB, et al. 2012 Infectious Diseases Society of America clinical practice guideline for the diagnosis and treatment of diabetic foot infections. Clin Infect Dis. 2012;54:e132-e173.
12. Clayton W Jr, Elasy TA. A review of the pathophysiology, classification, and treatment of foot ulcers in diabetic patients. Clin Diabetes. 2009;27:52-58.
13. Wukich DK, Armstrong DG, Attinger EC, et al. Inpatient management of diabetic foot disorders: a clinical guide. Diabetes Care. 2013;36:2862-2871.
14. Wagner FW Jr. The dysvascular foot: a system for diagnosis and treatment. Foot Ankle. 1981;2:64-122.
15. Van Acker K, De Block C, Abrams P, et al. The choice of diabetic foot ulcer classification in relation to the final outcome. Wounds. 2002;14:16-25.
16. Ranachowska C, Lass P, Korzon-Burakowska A, Dobosz M. Diagnostic imaging of the diabetic foot. Nucl Med Rev Cent East Eur. 2010;13:18-22.
17. Bronze MS. Diabetic foot infections. Medscape. http://emedicine.medscape.com/article/237378-overview. Accessed March 3, 2014.
18. American Diabetes Association. Consensus Development Conference on Diabetic Foot Wound Care: 7–8 April 1999, Boston, Massachusetts. Diabetes Care. 1999;22:1354-1360.
19. Meijer JW, van Sonderen E, Blaauwwiekel EE, et al. Diabetic neuropathy examination: a hierarchical scoring system to diagnose distal polyneuropathy in diabetes. Diabetes Care. 2000;23;750-753.
20. Aragón-Sánchez J. Seminar review: a review of the basis of surgical treatment of diabetic foot infections. Int J Low Extrem Wounds. 2011;10:33-65.
21. Umpierrez GE, Hellman R, Korytkowski MT, et al; Endocrine Society. Management of hyperglycemia in hospitalized patients in non-critical care setting: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2012;97:16-38.
22. Effect of intensive blood-glucose control with metformin on complications in overweight patients with type 2 diabetes (UKPDS 34). UK Prospective Diabetes Study (UKPDS) Group. Lancet. 1998;352:854-865.
23. Micromedex Healthcare Series [Internet database]. Greenwood Village, CO: Thompson Micromedex. www.micromedexsolutions.com. Accessed March 3, 2014.
To comment on this article, contact rdavidson@uspharmacist.com.






