US Pharm. 2011;36(4):HS2-HS6.
Optic neuropathies are disorders of the optic nerve involving degeneration of the nerve.1 Optic neuropathy should not be confused with optic neuritis. Both can lead to vision problems; optic neuritis involves inflammation of the optic nerve while optic neuropathy refers to damage from any cause. Optic neuritis is one of the many causes of optic neuropathy.
Optic neuropathies can be either hereditary or acquired. Acquired etiologies of optic neuropathy include ischemic, nutritional, and toxic types.1,2 Drug-induced optic neuropathy is of the toxic type. Mechanisms of drug-induced optic neuropathy include mitochondrial dysfunction, disruption of blood flow to the optic nerve, and unknown mechanisms.2
Common symptoms of optic neuropathy include decreased vision in the central field, which is typically bilateral but may be unilateral in some cases; visual-field defects; and swelling of the optic nerve.3 The onset is typically slow and painless.2
In drug-induced optic neuropathy, dechallenge (withdrawal) of the offending drug can lead to relief of symptoms.3,4 Since many of the medications known to cause optic neuropathy are common ones, it is important for pharmacists to be aware of this adverse effect in order to refer patients with these symptoms in a timely manner and avoid further, permanent damage.
Drugs That Can Cause Optic Neuropathy
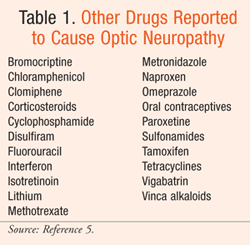
Many commonly prescribed drugs have been implicated in optic neuropathy. Discussed here are the agents that have the most supporting information: phosphodiesterase type 5 (PDE-5) inhibitors, amiodarone, linezolid, ethambutol, and isoniazid. Other drugs reported to cause optic neuropathy are listed in TABLE 1.5
PDE-5 Inhibitors: This class of drugs has revolutionized the treatment of erectile dysfunction (ED) in males. Currently there are three agents available—sildenafil (Viagra), tadalafil (Cialis), and vardenafil (Levitra). They exert their action by selectively inhibiting PDE type 5. Considered first-line agents for the treatment of ED, they are very commonly prescribed, with approximately 40 million males worldwide being treated for sexual dysfunction with PDE-5 inhibitors.6 Although sildenafil, tadalafil, and vardenafil are specific for PDE 5, these agents have been shown to have inhibitory effects on PDE 6, an isoenzyme expressed in the rods and cones of the eye. It is theorized that inhibition of PDE 6 results in the many visual disturbances associated with this drug class.7-9 The PDE-5 inhibitors account for approximately 19% of the FDA reports of drug-associated ocular toxicities, making it the most commonly reported drug class associated with this toxicity.10
NAION: Postmarketing surveillance has linked the PDE-5 inhibitors to nonarteritic anterior ischemic optic neuropathy (NAION). This potential relationship has raised controversy. Since 2000, there have been at least 20 case reports published in the literature describing PDE-5 inhibitor use and suspected NAION.11,12 Most of the published literature is related to sildenafil use since this was the first agent in this class to market (sildenafil was introduced in 1998, 5 years prior to tadalafil and vardenafil). To date, there have been no published case reports involving vardenafil.
NAION is a fairly uncommon ocular disorder in the general population that occurs suddenly, is usually painless, and results in an irreversible partial loss of vision.11,13 Although it is the most common acute optic neuropathy in people over the age of 50 years, there are only between 1,500 and 6,000 cases reported annually in the United States.14 The etiology of NAION is not well understood, but it is believed to be caused by a shortage of blood supply to the optic nerve. A well-established risk factor for the development of NAION occurs in individuals who have a small disc and a small or absent physiologic cup (i.e., when the optic nerve is nearly as large as the opening in the back of the eye and the optic disc appears “crowded”), also known as disc at risk or crowded disc. Other associated risk factors include diabetes, hypertension, hypercholesterolemia, atherosclerosis, ischemic heart disease, sleep apnea, and nocturnal hypotension. Many of these risk factors are also associated with ED.8,11
Of the NAION cases reported, most occurred within 24 hours after administration, usually upon waking in the morning. Patients presented with blurred vision and loss of visual field.15 In addition, most patients were found to have crowded disc as a risk factor along with various cardiovascular diseases, which are considered to be risk factors for both NAION and ED.
In a retrospective cohort study utilizing the National Veterans Health Administration’s pharmacy and clinical databases from 2004 to 2005, a 2-year cohort of men (approximately 4 million) who were at least 50 years of age without a prior history of optic nerve disease were evaluated.16 Approximately 11.5% of these patients were dispensed a PDE-5 inhibitor; the majority were prescribed sildenafil 100 mg. From this cohort, 0.09% of patients had a diagnosis of new ischemic optic neuropathy, with a relative risk of 1.02 (95% confidence interval [CI], 0.92-1.12).
Another retrospective study evaluated 38 men with NAION and compared them to 38 men without a history of the disease.17 There was not a marked overall difference in PDE-5 inhibitor use in either group; however, there was a significant association observed for those patients with a history of myocardial infarction and PDE-5 inhibitor use and the occurrence of NAION (P = .04). Similarly, the incidence of NAION was greater in those patients with a history of hypertension and PDE-5 inhibitor use, although this was not significant (P = .07).17
The mechanism of action of this adverse effect of PDE-5 inhibitors is not quite understood, and this is partly due to the fact that the mechanism of action for the development of NAION is not fully understood. It has been suggested that nocturnal hypotension may play a role in NAION development, and PDE-5 inhibitors may enhance this effect, resulting ultimately in ischemia of the optic nerve.12,13,18
The incidence of NAION cases of men using PDE-5 inhibitors worldwide is consistent with that of the general U.S. population.14 The World Health Organization (WHO) and the FDA have identified the association of NAION and PDE-5 inhibitors as “possible” causality. A “possible” adverse event “occurs within a reasonable time after administration of the drug, but could also be explained by concurrent disease or other drugs or chemicals. Positive dechallenge data are lacking or unclear in this category.”2 In 2005, the FDA recommended that the product labeling be updated to contain a statement describing this temporal association with the use of PDE-5 inhibitors. To date, there is no compelling evidence to suggest that PDE-5 inhibitor use is associated with a higher incidence of NAION.
Patients who have experienced an episode of NAION should avoid using PDE-5 inhibitors, as such individuals may be susceptible to developing NAION in the contralateral eye.13,19,20 Some conservative ophthalmologists may also recommend avoiding the use of these agents in those patients who have a small disc at risk, a criterion that may require all patients to have an ophthalmologic examination prior to initiation of treatment.13 Patients who are using PDE-5 inhibitors should be counseled on the risk of NAION development. If they notice any sudden vision loss they should stop the use of the PDE-5 inhibitor and seek medical attention immediately. All incidences should also be reported to the FDA, including information on drug duration and dose, time from PDE-5 inhibitor ingestion to onset of findings, and any ophthalmologic examination findings.19
Amiodarone: This drug, a class III antiarrhythmic agent used primarily for ventricular arrhythmias, has been implicated in various types of vision changes, including corneal abnormalities, conjunctival effects, and optic neuropathy, but the exact mechanism of optic neuropathy is unknown.21 Visual changes have been reported with varying prevalence of 0% to 11.4% in patients taking this drug; optic neuropathy was found in one study to occur in 1.79% of patients.20 The condition is typically insidious in onset, takes months to resolve, and presents bilaterally.4,21,22 It has been noted that amiodarone-induced optic neuropathy is very similar to NAION, but differs from it in that NAION typically exhibits rapid onset, rapid resolution, and unilateral presentation.4,21 Distinguishing NAION from amiodarone-induced optic neuropathy is often difficult.20
The prescribing information for amiodarone recommends regular ophthalmic examinations, including at baseline and then every 6 months, for patients receiving the drug.4,23 Some authors have suggested that more frequent eye examinations, every 4 months for up to a year after treatment initiation, may be warranted.21 As an antiarrhythmic agent, amiodarone is often not interchangeable with other drugs, and the decision to change therapy should be done in consultation with the patient’s ophthalmologist and cardiologist.
Linezolid: This oxazolidinone antibiotic is used for its efficacy against gram-positive organisms, specifically methicillin-resistant Staphylococcus aureus (MRSA). Its overall safety has been demonstrated in clinical trials of up to 28 days of treatment. Optic neuropathy has been associated with the long-term use of linezolid, with reported duration of treatment ranging from 5 to 11 months in doses of 600 to 1,200 mg/day, well outside of the studied clinical trial durations of 28 days or less.4,24 However, newer literature reports optic neuropathy after a shorter duration of treatment of just 16 days.25,26
Symptoms are gradual, and there is a painless onset of bilateral vision loss. Discontinuation of linezolid has led to improvement in visual symptoms in all reported cases. The mechanism of linezolid-induced optic neuropathy is not known, but may be due to mitochondrial toxicity from binding to mitochondrial 16S rRNA.27 Linezolid penetrates the central nervous system and eye well, which may lead to enhanced neurotoxicity.24,28 Withdrawal of linezolid and switching antibacterial therapy to another agent may aid recovery, although in some cases continuation of linezolid may be necessary to adequately treat certain infections.2 Baseline and periodic ophthalmologic examinations are recommended for patients receiving long-term linezolid treatment.4
Ethambutol: This drug, a first-line agent in the treatment and prevention of tuberculosis, causes numerous ocular adverse effects. It has been well documented to cause optic neuropathy in up to 5% of patients taking the drug.2 The mechanism of this adverse effect is not completely clear, but has been speculated to be related to chelation of copper in retinal cells.4
Most patients with this adverse effect had taken doses of 60 to 100 mg/kg/day with onset of symptoms after 2 to 12 months of treatment.4,29 The FDA-approved daily dose of ethambutol in the treatment of tuberculosis is 15 to 25 mg/kg/day. Ethambutol-induced optic neuropathy is thought to be dose related; it occurs at rates of 18% of patients at a dose of 35 mg/kg/day, 5% to 6% at 25 mg/kg/day, and less than 1% at 15 mg/kg/day.30 Dosage adjustment is recommended for patients with renal insufficiency, as inadequate clearance of the drug can lead to additional ocular toxicity.
The vision loss associated with ethambutol has been commonly reported to be reversible; however, it may be irreversible in certain populations, including geriatric patients and those who received the drug chronically.30
Ethambutol is contraindicated in patients with a history of optic neuritis unless clinical judgment warrants its use.31 Baseline and periodic eye examinations should be conducted; the prescribing information for ethambutol recommends monthly examinations for patients taking >15 mg/kg/day.31 Some authors have suggested that annual testing may be appropriate for those patients taking <15 mg/kg/day, with more frequent monitoring for those at high risk of developing optic neuropathy. This would include patients over 60 years or under 16 years; those with renal disease, alcoholism, or peripheral neuropathy; those taking more than 15 mg/kg/day; or treatment for longer than 6 months.2 Patients exhibiting symptoms of optic neuropathy should be evaluated, and the drug should be discontinued.
Isoniazid: This antibiotic is another drug used in the treatment and prevention of tuberculosis. It has been reported to cause optic neuropathy; however, since isoniazid is used in conjunction with ethambutol, which is also implicated in optic neuropathy, determining the cause of vision problems is often difficult.20 Isoniazid-induced optic neuropathy is thought to be less frequent and reversible.2
Treatment
Prompt ophthalmic evaluation is paramount, and that, along with discontinuation of the offending drug when possible, constitutes the basis of treatment of drug-induced optic neuropathy.4 While discontinuation of the drug is often recommended, this may not always be possible, as medications like amiodarone, linezolid, and ethambutol may be necessary to treat the patient optimally. In these circumstances, careful monitoring and ongoing care by an ophthalmologist is necessary.
Implications
Many medications can cause ocular adverse effects, and the potential risks should be discussed with patients prior to initiating therapy. Pharmacists are in the unique position of being the most accessible health care provider; they may be the ones patients will more likely consult when experiencing adverse effects due to their medications. It may not be possible to know all drug-induced ocular adverse effects, but it is important to recognize that many systemic medications can affect eye health. Early detection can potentially prevent or minimize serious harm. Pharmacists should be aware that most patients experiencing drug-induced optic neuropathy will present with decreased vision.2 Any patient complaining of a sudden and painless decline in vision should be advised to report these changes immediately.
In addition to contacting the FDA and the drug manufacturer, pharmacists can report suspected cases of drug-induced optic neuropathy to the National Registry of Drug-Induced Ocular Side Effects (www.eyedrugregistry.com). Information that will be helpful when reporting ocular adverse reactions includes drug name, duration, and dose; time from drug ingestion to onset of findings; and any ophthalmologic examination findings.
REFERENCES
1. O’Neill EC, Danesh-Meyer HV, Connell PP, et al. The optic nerve head in acquired optic neuropathies. Nat Rev Neurol. 2010;6:221-236.
2. Lloyd MJ, Fraunfelder FW. Drug-induced optic neuropathies. Drugs Today. 2007;43:827-836.
3. Spiteri MA, James DG. Adverse ocular reactions to drugs. Postgrad Med J. 1983;59:343-349.
4. Li J, Tripathi RC, Tripathi BJ. Drug-induced ocular disorders. Drug Saf. 2008;21:127-141.
5. Kaakeh Y, Abel SR. Visual disturbances. In: Tisdale JE, Miller DA, eds. Drug-Induced Diseases: Prevention, Detection, and Management. 2nd ed. Bethesda, MD: American Society of Health-Systems Pharmacists; 2010:
250-274.
6. Hatzimouratidis K, Hatzichristou D. Phosphodiesterase type 5 inhibitors: the day after. Eur Urol. 2007;51:75-89.
7. Lee M. Chapter 86. Erectile dysfunction. In: DiPiro JT, Talbert RL, Yee GC, et al, eds. Pharmacotherapy: A Pathophysiologic Approach. 7th ed. New York, NY: McGraw-Hill; 2010. www.accesspharmacy.com/
8. Kerr NM, Danesh-Meyer HV. Phosphodiesterase inhibitors and the eye. Clin Experiment Ophthalmol. 2009;37:514-523.
9. Bischoff E. Potency, selectivity, and consequences of nonselectivity of PDE inhibition. Int J Impot Res. 2004;16(suppl 1):S11-S14.
10. Barbehenn E, Lurie P, Wolfe SM, et al. Petition to the FDA to require a black box warning for erectile dysfunction drugs sildenafil (Viagra), tadalafil (Cialis), vardenafil (Levitra) to warn of the potential for irreversible vision loss. Public Citizen. October 20, 2005. www.citizen.org/hrg1753. Accessed February 24, 2011.
11. Laties AM. Vision disorders and phosphodiesterase type 5 inhibitors: a review of the evidence to date. Drug Safety. 2009;32:1-18.
12. Danesh-Meyer HV, Levin HA. Erectile dysfunction drugs and risk of anterior ischaemic optic neuropathy: casual or causal association? Br J Ophthalmol. 2007;91:1551-1555.
13. Carter JE. Anterior ischemic optic neuropathy and stroke with use of PDE-5 inhibitors for erectile dysfunction: cause or coincidence? J Neurol Sci. 2007;262:89-97.
14. Gorkin L, Hvidsten, Sobel RE, et al. Sildenafil citrate use and the incidence of nonarteritic anterior ischemic optic neuropathy. Int J Clin Pract. 2006;60:500-503.
15. Pomeranz HD, Bhavsar AR. Nonarteritic ischemic optic neuropathy developing soon after use of sildenafil (Viagra): a report of seven new cases. J Neurol Ophthalmol. 2005;25:9-13.
16. Margo CE, French DD. Ischemic optic neuropathy in male veterans prescribed phosphodiesterase-5 inhibitors. Am J Ophthalmol. 2007;143:538-539.
17. McGwin G, Vaphiades MS, Hall TA, et al. Non-arteritic anterior ischaemic optic neuropathy and the treatment of erectile dysfunction. Br J Ophthalmol. 2006;90:154-157.
18. Fraunfelder FW. Visual side effects associated with erectile dysfunction agents. Am J Ophthalmol. 2005;140:723-724.
19. McKoy JM, Bolden CR, Samaras A, et al. Sildenafil- and tadalafil-associated optic neuropathy: implications for men after prostate cancer treatment. Community Oncol. 2009;6:78-79.
20. Santaella RM, Fraunfelder FW. Ocular adverse effects associated with systemic medications. Drugs. 2007;67:75-93.
21. Johnson LN, Krohel GB, Thomas ER. The clinical spectrum of amiodarone-associated optic neuropathy. J Nat Med Assoc. 2004;96:1477-1491.
22. Mantyjarvi M, Tuppurainen K, Ikaheimo K. Ocular side effects of amiodarone. Surv Ophthalmol. 1998;42:360-366.
23. Cordarone (amiodarone HCl) package insert. Philadelphia, PA: Wyeth Pharmaceuticals, Inc; December 2010.
24. Jahaveri M, Khurana RN, O’Hearn TM, et al. Linezolid-induced optic neuropathy: a mitochondrial disorder? Br J Ophthalmol. 2007;91:111-115.
25. Azamfirei L, Copotoiu SM, Branzaniuc K, et al. Complete blindness after optic neuropathy induced by short-term linezolid treatment in a patient suffering from muscle dystrophy. Pharmacoepidemiol Drug Saf. 2007;16:402-404.
26. Joshi L, Taylor SR, Large O, et al. A case of optic neuropathy after short-term linezolid use in a patient with acute lymphocytic leukemia. Clin Infect Dis. 2009;48:e73-e74.
27. Narita M, Tsuji BT, Yu VL. Linezolid-associated peripheral and optic neuropathy, lactic acidosis, and serotonin syndrome. Pharmacotherapy. 2007;27:1189-1197.
28. Rucker JC, Hamilton SR, Bardenstein D, et al. Linezolid-associated toxic optic neuropathy. Neurology. 2006;66:595-598.
29. Melamud A, Kosmorsky GS, Lee MS. Ocular ethambutol toxicity. Mayo Clin Proc. 2003;78:1409-1411.
30. Kokkada SB, Barthakur R, Natarajan M, et al. Ocular side effects of antitubercular drugs—a focus on prevention, early detection and management. Kathmandu Univ Med J. 2005;3:438-441.
31. Myambutol (ethambutol) package insert. Northport, NY: X-Gen Pharmaceuticals, Inc; May 2007.
To comment on this article, contact rdavidson@uspharmacist.com.






