US Pharm. 2011;36(10):73-76.
It seems as though almost every patient admitted to the hospital in the United States is prescribed either a proton pump inhibitor (PPI) or a histamine-2 antagonist (H2A) as stress ulcer prophylaxis (SUP). Stress ulcers are gastric mucosal erosions that can develop in patients with a serious illness or severe injury. Unlike peptic ulcers, which tend to develop in the antrum or duodenum, stress-related mucosal injuries typically occur in the acid-producing areas of the stomach (i.e., corpus and fundus).1 Stress ulcers, unlike peptic ulcers, tend to cause gastrointestinal (GI) bleeding and are not associated with abdominal pain. The mucosal changes of stress-related mucosal injury mainly involve small erosions that do not lead to bleeding but may develop into deeper ulcers, which cause bleeding.
Several factors contribute to stress ulcer formation, including acid hypersecretion; alteration of normal protective mechanisms such as mucus and bicarbonate secretions; release of mediators such as arachidonic acid metabolites, cytokines, and oxygen free radicals; and ischemia to the GI system.2 These erosions may occur quickly (£24 hours of admission) or take longer to develop (>10-14 days).2 Stress ulcers and related bleeding cause significant morbidity and mortality in critically ill patients. One study reported a mortality rate of 46% in critically ill patients with GI bleeding compared with 21% in those without bleeding (P <.001).3 Other studies have confirmed this high mortality rate.4,5
Large studies have indicated that the strongest risk factors for stress-related GI bleeding are prolonged mechanical ventilation and coagulopathy.4 The risk increases with increasing number of days of mechanical ventilation and length of ICU stay.6 Other risk factors include recent major surgery, major trauma, severe burns, head trauma, hepatic or renal disease at admission, sepsis, and hypotension.1 TABLE 1 provides a complete list of risk factors.

Prophylactic Therapy
Although proven to be highly effective in reducing the risk of both overt and clinically important stress-related mucosal bleeding, prophylactic therapy alone does not decrease the overall mortality rate.7 Still, the prescribing of PPIs or H2As as SUP is supported by the literature and by guidelines for the critical care population. In recent years, the use of SUP in non-ICU patients—often with no indication—has increased. Multiple studies have demonstrated the overuse of acid-suppressive drugs (ASDs); as many as 71% of admitted patients are prescribed ASDs, with many being erroneously discharged with these agents.8 Many patients are inappropriately prescribed ASDs in the community, and little is done to discontinue these medications during hospitalization. Additionally, patients on twice-daily PPI dosing to treat gastroesophageal reflux disease (GERD) symptoms often are not stepped down to single-daily-dose therapy once they are asymptomatic.9
The overuse and misuse of ASDs may result in patient harm and increased costs for the health care system. The number needed to treat (NNT) to prevent a single episode of clinically significant GI bleeding in the ICU exceeds 900; the NNT is unknown in the non-ICU setting, but is expected to be much higher.9 In a retrospective cohort study in an academic hospital, inpatient costs for SUP in the non-ICU setting were $44,000 annually and outpatient pharmacy costs were nearly $68,000 when the drugs were continued postdischarge, for a combined yearly estimated expenditure of nearly $112,000.10 This could easily have been prevented.
In light of these statistics, SUP continues to be overprescribed. In a cross-sectional Web-based survey assessing physicians’ knowledge, beliefs, and behavior regarding the prescribing of SUP in non-ICU patients, fears about GI bleeding and the legal repercussions of not prescribing SUP were associated with a higher prescribing level, whereas knowledge of SUP indications and concern about side effects were associated with low prescribing behavior.11 Fewer than half of physicians could identify a single side effect of PPI therapy.11 Many practitioners view these drugs as harmless, but studies have suggested that PPI and H2A therapy is strongly associated with a greater likelihood of Clostridium difficile infection (CDI) and increased recurrence rates.12,13 Other often-overlooked risks associated with this therapy include hypomagnesemia; vitamin B12 deficiency; upper respiratory infections, including community-acquired pneumonia (CAP) and hospital-acquired pneumonia (HAP); fractures of the hip, spine, and wrists; and altered absorption of pH-dependent medications.
Risks of Therapy
CDI: The toxin produced by C difficile, an anaerobic, spore-forming, gram-positive bacterium, is a major cause of diarrhea and colitis in hospitalized patients that has increased in incidence, severity, and mortality in recent years. Approximately 3 million cases occur annually in the U.S., and health care costs exceed $1.1 billion per year.14,15 C difficile–induced pseudomembranous colitis has a mortality rate of 6% to 30%.16 CDI has been implicated in 20% to 30% of all cases of antibiotic-associated diarrhea, 50% to 75% of cases of antibiotic-associated colitis, and more than 90% of cases of antibiotic-associated pseudomembranous colitis in hospitalized patients.17
Despite better control of antibiotic prescribing and improvements in hospital hygiene, the number of CDI cases continues to rise.18 Improved diagnosis, hypervirulent strains, and an aging population may have contributed to this, but these factors do not fully explain the extent of recent increases.18 Common risk factors for CDI include age over 65 years, current and recurrent hospitalization, recent GI surgery, immunosuppression, previous CDI, residing in a nursing home or long-term care facility, and antibiotic use. Antibiotic use is the biggest risk factor for the development of CDI, but it often cannot be avoided in patients with infections. A less common but more modifiable risk factor is the use of ASDs in hospitalized patients in whom these drugs are not indicated.
Gastric acid is a nonspecific protective mechanism against ingested pathogens. The stomach is normally free of bacteria, with the exception of Helicobacter pylori, which is uniquely equipped to deal with gastric acidity.19 C difficile is transmitted by ingestion, and its relatively acid-stable spores go on to germinate in the small intestine and colon. The vegetative cells of C difficile, unlike the spores, are usually killed in the low-pH environment of the stomach; however, they have been found to remain viable in the gastric contents of patients taking ASDs. Research in animal models has shown that C difficile spores transform into vegetative cells within 1 hour of ingestion. It is plausible that this transformation may occur in the stomach if the pH is higher or in the case of slower gastric transit times.
Several studies have found an association between PPI or H2A use and CDI. A recent retrospective cohort study found that recurrent CDI was more common in patients exposed to PPIs than in unexposed patients (25.2% vs. 18.5%) and that PPI use during CDI treatment was associated with a 42% increased risk of recurrence.12 Exposed patients aged older than 80 years had the highest risk.12 A recent analysis of data from patient discharges from a tertiary medical center over a 5-year period found that the risk of nosocomial CDI was positively correlated with the level of acid suppression and was highest in patients receiving PPI therapy more than once daily, suggesting a dose-response effect on the risk of nosocomial CDI.13
Pneumonia: Although results of investigations have been inconsistent, several studies have found an association between the use of ASDs and an increased risk of respiratory tract infections and CAP. A recent systematic review and meta-analysis suggested that the use of PPIs and H2As is associated with a greater risk of both CAP and HAP, estimating an occurrence of one case of pneumonia for every 200 inpatients treated with ASDs.20 Considering that 40% to 70% of inpatients receive ASDs, the magnitude of the morbidity and mortality of HAP can be fully appreciated. The largest increase in the risk of pneumonia associated with PPIs was observed in the first week of use.20
As with enteric infections, ASDs are thought to increase the risk of pneumonia by inhibiting the secretion of gastric acid, thus allowing bacterial overgrowth and colonization. Bacteria may then be translocated to the lungs by aspiration.20 In mechanically ventilated patients, oropharyngeal colonization with gram-negative organisms may lead to gastric colonization.19 Another theory is that hydrogen potassium adenosine triphosphatase is present not only in the parietal cells of the stomach, but also in the respiratory tract.20 Inhibition of this enzyme by a PPI could encourage bacterial growth in the respiratory tract, thus leading to an increased risk of pneumonia.20 Lastly, in vitro studies have shown that ASDs may impair the function of neutrophils and the activity of natural killer cells.20
Patients over 65 years of age are at increased risk for pneumonia due to ASDs. Not only are elderly patients generally hypochloric because of the aging process, but PPI or H2A use is common in this group; up to 5% take these medications regularly, and a high percentage of hospitalized elderly patients receive SUP.21 Additionally, many PPIs and H2As do not require a prescription, resulting in increased and unmonitored use, often for long periods of time without medical supervision.21
Other Risks: In addition to CDI, other enteric infections—in particular, from Salmonella and Campylobacter species—have been associated with ASDs. Both Campylobacter and Salmonella are acid-sensitive microbes. Parasitic infections have been linked to acid suppression as well, especially giardiasis and strongyloidiasis.22 As previously mentioned, gastric acid is important in the killing of ingested organisms, but an elevated gastric pH may have other negative effects on GI host defenses, such as delayed gastric emptying, increased bacterial translocation, decreased gastric-mucus viscosity, and changes in the normal microbial flora.23
Drug interactions are another concern with acid suppression. Medications with pH-dependent absorption (i.e., digoxin, nifedipine, indinavir, midazolam, didanosine, alendronate, methadone, aspirin) may be affected by both PPIs and H2As, resulting in increased rates of absorption.9,24 Likewise, several drugs have decreased rates of absorption (i.e., ketoconazole, cefpodoxime, dipyridamole, atazanavir, itraconazole).24 There are many CYP450-related interactions between PPIs and other drugs. The potential interaction between PPIs and clopidogrel has attracted attention because of possible serious adverse effects. An FDA safety alert in November 2009 recommended avoiding the use of omeprazole or esomeprazole with clopidogrel, warning that the CYP-mediated interaction could reduce clopidogrel’s effectiveness.22 Because of insufficient data, the FDA made no specific recommendations concerning other PPIs.22
Several cases of patients with hypomagnesemia and subsequent hypokalemia and hypocalcemia who presented with life-threatening symptoms requiring hospitalization have been reported. Most of these cases were associated with long-term PPI use. The mechanism of this adverse effect is unknown. In patients with PPI-induced hypomagnesemia, no apparent magnesium malabsorption or renal magnesium wasting has occurred.22 Although hypomagnesemia is rare, because it is so life-threatening providers are encouraged to watch carefully for unexplained hypomagnesemia, hypokalemia, or hypocalcemia in PPI users.22
The use of PPIs for 12 months or longer has been associated with vitamin B12 deficiency, particularly in elderly patients.9 Gastric acidity and pepsin enable the release of ingested vitamin B12 from its protein-bound state; therefore, reduction in gastric acid may lead to malabsorption and vitamin B12 deficiency.22 Study results have been inconsistent, and larger studies are needed to define the association between PPI use and vitamin B12 malabsorption and subsequent deficiency. Poor absorption of dietary iron has also been linked to PPI therapy, but current data are weak. Gastric acid facilitates the absorption of nonheme iron by reducing ferric iron to the more soluble ferrous form, and also enhances iron-salt dissociation from ingested food.22 Several case reports exist, but no prospective studies have been performed in this area.
Long-term PPI use has been associated with an increased incidence of fractures. Possible mechanisms include decreased absorption of calcium in a more alkaline gastric environment and impairment of bone remodeling due to decreased osteoclast activity, leading to inhibited microfracture repair.22 Once again, studies are inconsistent, but existing evidence was enough to prompt the FDA to revise the PPI labeling in May 2010 to include new safety information about the possible increased risk of fractures of the hip, wrist, and spine.22 The risk of fracture may be more clinically significant in patients with osteoporosis or other risk factors for fracture, such as steroid use. Physicians are encouraged to weigh the risks and benefits of PPI use in vulnerable patients. If calcium supplements are prescribed to PPI users, they should be in a soluble form, such as citrate, rather than carbonate.22
Appropriate Use
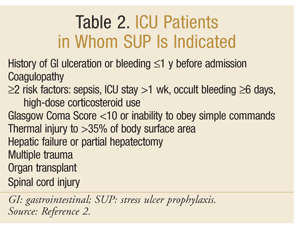
The American Society of Health-System Pharmacists has developed one of the most comprehensive evidence-based guidelines for SUP. The current therapeutic guidelines were published in 1999, with an update in progress at this writing.25 SUP is not recommended for non-ICU patients; prophylaxis is recommended only for ICU patients who require mechanical ventilation for more than 48 hours, have a history of GI ulceration or bleeding within 1 year before admission, or who have coagulopathy. Prophylaxis is also advised for ICU patients with at least two of the following risk factors: sepsis, ICU stay exceeding 1 week, occult bleeding lasting 6 days or more, and the use of high-dose corticosteroids (hydrocortisone >250 mg/day or equivalent). Additionally, SUP is recommended for special ICU populations, such as patients with a Glasgow Coma Score <10 (or inability to obey simple commands), thermal injury to >35% of body surface area, hepatic failure or partial hepatectomy, multiple trauma, transplants, or spinal cord injury.2 The recommendations are summarized in TABLE 2.
Conclusion: Pharmacist’s Role
Efforts by clinicians to curb the use of ASDs in non-ICU patients can prevent significant patient morbidity and reduce both hospital costs and costs to the overall health care system. It is important to carefully weigh the risks and benefits of ASDs before using these medications in the inpatient setting. Special consideration should be given to patients already at risk for pneumonia, such as the elderly, those with chronic lung disease, and those taking immunosuppressants. Every effort should be made to critically assess acid-suppression therapy in inpatients and to discontinue therapy whenever possible. When SUP is indicated, the patient should receive the lowest dose possible.
Pharmacists can help stem the use of ASDs by identifying patients who are inappropriately prescribed these medications, and they can be instrumental in educating physicians about which patients should receive SUP. Pharmacists involved in processing discharge medications can help ensure that patients are not sent home on acid-suppressive therapy without an appropriate indication. Unfortunately, the measures taken to prevent stress ulcers in hospitalized patients have created a new problem, and by working together the health care team can help prevent further unnecessary morbidity by enforcing guideline recommendations at their institution.
REFERENCES
1. Spirt MJ. Stress-related mucosal disease: risk factors and prophylactic therapy. Clin Ther. 2004;26:197-213.
2. ASHP therapeutic guidelines on stress ulcer prophylaxis. ASHP Commission on Therapeutics and approved by the ASHP Board of Directors on November 14, 1998. Am J Health Syst Pharm. 1999;56:347-379.
3. Cook DJ, Griffith LE, Walter SD, et al. The attributable mortality and length of intensive care unit stay of clinically important gastrointestinal bleeding in critically ill patients. Crit Care. 2001;5:368-375.
4. Cook DJ, Fuller HD, Guyatt GH, et al. Risk factors for gastrointestinal bleeding in critically ill patients. Canadian Critical Care Trials Group. N Engl J Med. 1994;330:377-381.
5. Zuckerman GR, Shuman R. Therapeutic goals and treatment options for prevention of stress ulcer syndrome. Am J Med. 1987;83:29-35.
6. Harris SK, Bone RC, Ruth WE. Gastrointestinal hemorrhage in patients in a respiratory intensive care unit. Chest. 1977;72:301-304.
7. Sesler JM. Stress-related mucosal disease in the intensive care unit: an update on prophylaxis. AACN Adv Crit Care. 2007;18:119-128.
8. Grube RR, May DB. Stress ulcer prophylaxis in hospitalized patients not in intensive care units. Am J Health Syst Pharm. 2007;64:1396-1400.
9. Heidelbaugh JJ, Goldberg KL, Inadomi JM. Overutilization of proton pump inhibitors: a review of cost-effectiveness and risk in PPI. Am J Gastroenterol. 2009;104(suppl 2):S27-S32.
10. Heidelbaugh JJ, Inadomi JM. Magnitude and economic impact of inappropriate use of stress ulcer prophylaxis in non-ICU hospitalized patients. Am J Gastroenterol. 2006;101:2200-2205.
11. Hussain S, Stefan M, Visintainer P, Rothberg M. Why do physicians prescribe stress ulcer prophylaxis to general medicine patients? South Med J. 2010;103:1103-1110.
12. Linsky A, Gupta K, Lawler EV, et al. Proton pump inhibitors and risk for recurrent Clostridium difficile infection. Arch Intern Med. 2010;170:772-778.
13. Howell MD, Novack V, Grgurich P, et al. Iatrogenic gastric acid suppression and the risk of nosocomial Clostridium difficile infection. Arch Intern Med. 2010;170:784-790.
14. Kyne L, Hamel MB, Polavaram R, Kelly CP. Health care costs and mortality associated with nosocomial diarrhea due to Clostridum difficile. Clin Infect Dis. 2002;34:346-353.
15. Schroeder MS. Clostridium difficile–associated diarrhea. Am Fam Physician. 2005;71:921-928.
16. McMaster-Baxter NL, Musher DM. Clostridium difficile: recent epidemiologic findings and advances in therapy. Pharmacotherapy. 2007;27:1029-1039.
17. Kelly CP, Pothoulakis C, LaMont JT. Clostridium difficile colitis. N Engl J Med. 1994;330:257-262.
18. Cunningham R, Dial S. Is over-use of proton pump inhibitors fuelling the current epidemic of Clostridium difficile-associated diarrhoea? J Hosp Infect. 2008;70:1-6.
19. Vakil N. Acid inhibition and infections outside the gastrointestinal tract. Am J Gastroenterol. 2009;104:S17-S20.
20. Eom CS, Jeon CY, Lim JW, et al. Use of acid-suppressive drugs and risk of pneumonia: a systematic review and meta-analysis. CMAJ. 2011;183:310-319.
21. Eurich DT, Sadowski CA, Simpson SH, et al. Recurrent community-acquired pneumonia in patients starting acid-suppressing drugs. Am J Med. 2010;123:47-53.
22. Sheen E, Triadafilopoulos G. Adverse effects on long-term proton pump inhibitor therapy. Dig Dis Sci. 2011;56:931-950.
23. Dial MS. Proton pump inhibitor use and enteric infections. Am J Gastroenterol. 2009;104(suppl 2):S10-S16.
24. Lahner E, Annibale B, Della Fave G. Systematic review: impaired drug absorption related to the co-administration of antisecretory therapy. Aliment Pharmacol Ther. 2009;29:1219-1229.
25. American Society of Health-System Pharmacists. Therapeutic guidelines.
www.ashp.org/Import/
To comment on this article, contact rdavidson@uspharmacist.com.





