US Pharm. 2012;37(1):36-39.
Gastroesophageal reflux disease (GERD) is one of the most common gastrointestinal (GI) disorders.1 More than 60 million Americans have heartburn or regurgitation at least once a month, while more than 15 million experience daily symptoms.2 Additionally, many patients with GERD are asymptomatic.1 According to the National Institutes of Health, the total cost associated with GERD in 2004 was $12.6 billion, comprising $12.1 billion in direct costs and $0.5 billion in indirect costs.3
Symptoms
GERD symptoms are caused by a reflux of acid and gastric contents into the esophagus.1 Incompetent barriers at the gastroesophageal junction cause this backflow. Symptoms of GERD include heartburn and the regurgitation of sour material into the mouth. Some patients may also experience chest pain and dysphagia. Extraesophageal symptoms of GERD may occur when the gastric contents reflux into the pharynx, larynx, tracheobronchial tree, nose, and mouth. These manifestations may include chronic cough, laryngitis, pharyngitis, chronic sinusitis, and pulmonary aspiration. Serious complications may develop when GERD is left untreated. These complications include severe chest pain, esophageal stricture, Barrett’s esophagus, and esophageal cancer.1
Awakenings Due to Nocturnal Heartburn and GERD
Nocturnal heartburn has been reported in 10% of the U.S. population.4,5 Sleep disturbance is a common complaint in patients suffering from GERD.6 According to a Gallup poll conducted for the American Gastroenterology Association, 75% of patients with GERD reported that their symptoms affected sleep, and 40% stated that nighttime heartburn impaired daily functioning the following day. Patients with nighttime heartburn also reported reduced health-related quality of life. Approximately 30% of patients reporting nocturnal heartburn denied taking any kind of medication.6
Fass and colleagues reported clinical predictors of heartburn during sleep in a large, prospective, cohort study.7 The following were all strong predictors of nocturnal heartburn: increased body mass index, consumption of carbonated soft drinks, snoring and daytime sleepiness, difficulty sleeping, high blood pressure, asthma, and the use of benzodiazepines.7 In a different study that examined risk factors for nocturnal reflux in GERD patients, subjects with frequent and severe daytime symptoms were significantly more likely to report nighttime symptoms.8
Some medications utilized as sleep aids may worsen GI symptoms.7,9,10 Benzodiazepines are associated with heartburn during sleep and have been shown to decrease basal lower esophageal sphincter pressure, thereby increasing the number of reflux events.7 Additionally, zolpidem reduced the arousal response to nighttime reflux and increased the duration of acid reflux incidents in patients with and without GERD.9 Sleep-aid medications should be used with caution in patients with GERD.
The consequences secondary to nocturnal GERD and heartburn may be greater than those secondary to daytime symptoms.8 Although nocturnal symptoms may be less frequent than daytime symptoms, these episodes may result in more serious outcomes because of decreased esophageal motility, resulting in a longer episode duration and increased contact time. Thus, these patients may be at greater risk for esophageal lesions, respiratory conditions, esophageal adenocarcinoma, extraesophageal symptoms (e.g., cough, choking, snoring), and reduced quality of life.5,11,12 Patients often do not report nighttime GERD-related symptoms to primary care providers unless specifically asked.
Treatment
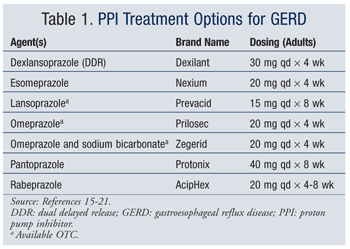
The goal of GERD treatment is acid suppression. Proton pump inhibitors (PPIs) suppress gastric acid secretion via the inhibition of the hydrogen-potassium–activated adenosine triphosphatase proton pump; thus, the gastric acid pH is increased.13 PPIs, the most widely used class of drugs for the treatment of GERD, provide rapid symptom relief and heal esophagitis in many patients.14 Currently, seven drugs are available in this class (TABLE 1).15-21 Histamine-2 (H2) receptor blockers are also used to treat GERD; however, they are typically less effective than PPIs.14 Nonpharmacologic strategies for reducing symptoms of GERD are given in TABLE 2.2,22

Although PPIs are standard treatment for GERD, acid suppression naturally declines overnight, and patients may experience nocturnal heartburn. Sleep quality and nighttime heartburn improve in patients receiving standard PPI therapy.23-28 A reduction in reflux-associated awakenings and nighttime heartburn has been documented in patients experiencing these symptoms at study onset (TABLE 3).24-28 However, none of the PPIs has been shown to entirely eliminate heartburn or GERD symptoms in all patients.11,25,29 Prevalence of nighttime heartburn in patients with GERD may be high even in patients receiving continuous PPI therapy.11 In studies involving patients with erosive esophagitis, only 64% to 85% had resolution of nocturnal heartburn after 8 weeks of therapy.30-33 Additional studies in patients with erosive esophagitis reported rates of heartburn-free nights of 80% to 90% following treatment with standard-dose PPIs.34-39
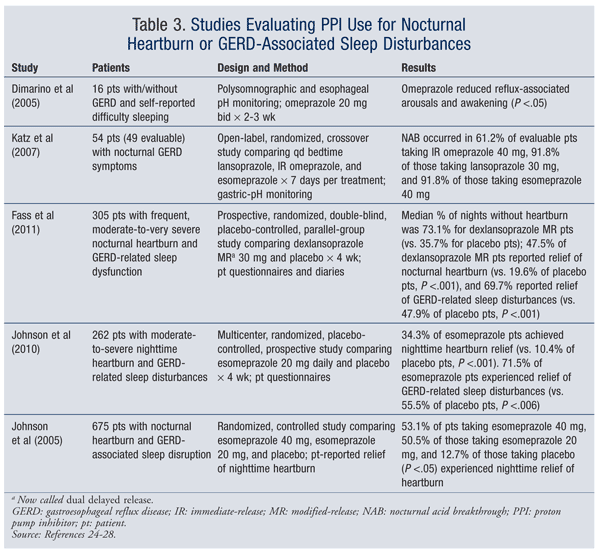
Sleep quality, work productivity, and regular daily activities have been assessed in clinical trials.26 In a placebo-controlled study by Fass and colleagues, patients receiving dexlansoprazole MR (now called dual delayed release) 30 mg daily had significantly less impairment of daily activities (child care, housework, shopping, studying) (P = .001) and fewer impaired work hours (P = .036).26 In a study conducted by Johnson and colleagues, work productivity (P = .002) and regular daily activities (P = .003) significantly improved in patients treated with esomeprazole 20 mg versus placebo.27
Higher doses of PPIs provide a longer duration of acid suppression; however, studies have not documented a greater reduction of nighttime symptoms.11 Twice-daily dosing of PPIs for the reduction of nocturnal heartburn has been evaluated.11,40-43 Nocturnal acid breakthrough occurs despite the administration of a PPI prior to dinner or bedtime.11,40,42,43 For this reason, bedtime dosing of an additional agent (e.g., an H2-receptor antagonist [H2RA]) was evaluated in several studies.43-45 The addition of an H2RA improved nighttime acid suppression, but concern exists that regular use of such agents may result in tachyphylaxis and, therefore, a limited duration of symptom relief.11,43
Although PPIs are well tolerated overall, there may be some risks associated with their use. The increase in fracture risk in both men and women is a concern.15-21,46 The risk of fractures appears to be highest in patients receiving high doses of PPIs (not available OTC) and in patients using a PPI for 1 year or more.46 Other safety concerns when PPIs are used for more than a year include reduced absorption of magnesium and vitamin B12.15-21,47 Patients taking PPIs may also be at increased risk for pneumonia or Clostridium difficile infection.
Conclusion
Nighttime heartburn occurs in a large number of patients with GERD, and nocturnal heartburn and GERD impair sleep and impact daily functioning. Acid suppression is the goal of GERD treatment. PPIs are currently the most effective treatment for increasing gastric pH, and PPI treatment has been shown to reduce nocturnal symptoms and awakenings from heartburn and GERD. However, none of the PPIs hs been shown to entirely eliminate heartburn or GERD symptoms in all patients. Greater education of patients and providers is needed. It is imperative that providers discuss nocturnal GERD-related symptoms with patients since the complications secondary to nocturnal GERD and heartburn may be more serious owing to the increased contact time of the acid content.
REFERENCES
1. Goyal RK. Diseases of the esophagus. In: Fauci AS, Kasper DL, Longo DL, et al, eds. Harrison’s Principles of Internal Medicine. 17th ed. New York, NY: McGraw-Hill Medical; 2008:1847-1855.
2. American College of Gastroenterology. The word on GERD.
http://67.192.160.122/
3. Ruhl CE, Sayer B, Byrd-Holt DD, Brown DM. Costs of digestive diseases. In: Everhart JE, ed. The Burden of Digestive Disease in the United States. U.S. Department of Health and Human Services, Public Health Service, National Institutes of Health, National Institute of Diabetes and Digestive and Kidney Diseases. Washington, DC: U.S. Government Printing Office, 2008; NIH Publication No. 09-6443:137-147.
4. Farup C, Kleinman L, Sloan S, et al. The impact of nocturnal symptoms associated with gastroesophageal reflux disease on health-related quality of life. Arch Intern Med. 2001;161:45-52.
5. Shaker R. Nighttime GERD: clinical implications and therapeutic challenges. Best Pract Res Clin Gastroenterol. 2004;18:31-38.
6. Shaker R, Castell DO, Schoenfeld PS, Spechler SJ. Nighttime heartburn is an under-appreciated clinical problem that impacts sleep and daytime function: the results of a Gallup survey conducted on behalf of the American Gastroenterological Association. Am J Gastroenterol. 2003;98:1487-1493.
7. Fass R, Quan SF, O’Connor GT, et al. Predictors of heartburn during sleep in a large prospective cohort study. Chest. 2005;127:1658-1666.
8. Gaddam S, Maddur H, Wani S, et al. Risk factors for nocturnal reflux in a large GERD cohort. J Clin Gastroenterol. 2011;45:764-768.
9. Gagliardi GS, Shah AP, Goldstein M, et al. Effect of zolpidem on the sleep arousal response to nocturnal esophageal acid exposure. Clin Gastroenterol Hepatol. 2009;7:948-952.
10. Hall AW, Moossa AR, Clark J, et al. The effects of premedication drugs on the lower oesophageal high pressure zone and reflux status of rhesus monkeys and man. Gut. 1975;16:347-352.
11. Johnson DA, Katz PO. Nocturnal gastroesophageal reflux disease: issues, implications, and management strategies. Rev Gastroenterol Disord. 2008;8:98-108.
12. Orr WC, Allen ML, Robinson M. The pattern of nocturnal and diurnal esophageal acid exposure in the pathogenesis of erosive mucosal damage. Am J Gastroenterol. 1994;89:509-512.
13. Hoogerwerf WA, Pasricha PJ. Pharmacotherapy of gastric acidity, peptic ulcers, and gastroesophageal reflux disease. In: Brunton LL, Lazo JS, Parker KL, eds. Goodman & Gilman’s The Pharmacological Basis of Therapeutics. 11th ed. New York, NY: McGraw-Hill Medical; 2006:967-981.
14. DeVault KR, Castell DO. Updated guidelines for the diagnosis and treatment of gastroesophageal reflux disease. Am J Gastroenterol. 2005;100:190-200.
15. Dexilant (dexlansoprazole) package insert. Deerfield, IL: Takeda Pharmaceuticals America, Inc; October 2011.
16. Nexium (esomeprazole) package insert. Wilmington, DE: AstraZeneca Pharmaceuticals LP; July 2011.
17. Prevacid (lansoprazole) package insert. Deerfield, IL: Takeda Pharmaceuticals America, Inc; October 2011.
18. Prilosec (omeprazole) package insert. Wilmington, DE: AstraZeneca Pharmaceuticals LP; June 2011.
19. Zegerid (omeprazole/sodium bicarbonate) package insert. San Diego, CA: Santarus, Inc; May 2011.
20. Protonix (pantoprazole) package insert. Philadelphia, PA: Wyeth Pharmaceuticals Inc; October 2011.
21. AcipHex (rabeprazole) package insert. Woodcliff Lake, NJ: Eisai Inc; May 2011.
22. Zweber A, Berardi RR. Heartburn and dyspepsia. In: Berardi RR, Ferreri SP, Hume AL, et al, eds. Handbook of Nonprescription Drugs. 16th ed. Washington, DC: American Pharmacists Association; 2009:231-246.
23. Chand N, Johnson DA, Tabangin M, Ware JC. Sleep dysfunction in patients with gastro-oesophageal reflux disease: prevalence and response to GERD therapy, a pilot study. Aliment Pharmacol Ther. 2004;20:969-974.
24. Dimarino AJ Jr, Banwait KS, Eschinger E, et al. The effect of gastro-oesophageal reflux and omeprazole on key sleep parameters. Aliment Pharmacol Ther. 2005;22:325-329.
25. Katz PO, Koch FK, Ballard ED, et al. Comparison of the effects of immediate-release omeprazole oral suspension, delayed-release lansoprazole capsules and delayed-release esomeprazole capsules on nocturnal gastric acidity after bedtime dosing in patients with night-time GERD symptoms. Aliment Pharmacol Ther. 2007;25:197-205.
26. Fass R, Johnson DA, Orr WC, et al. The effect of dexlansoprazole MR on nocturnal heartburn and GERD-related sleep disturbances in patients with symptomatic GERD. Am J Gastroenterol. 2011;106:421-431.
27. Johnson DA, Crawley JA, Hwang C, Brown K. Clinical trial: esomeprazole for moderate-to-severe nighttime heartburn and gastro-oesophageal reflux disease-related sleep disturbances. Aliment Pharmacol Ther. 2010;32:182-190.
28. Johnson DA, Orr WC, Crawley JA, et al. Effect of esomeprazole on nighttime heartburn and sleep quality in patients with GERD: a randomized, placebo-controlled trial. Am J Gastroenterol. 2005;100:1914-1922.
29. Nocon M, Labenz J, Jaspersen D, et al. Nighttime heartburn in patients with gastroesophageal reflux disease under routine care. Digestion. 2008;77:69-72.
30. Dekkers CP, Beker JA, Thjodleifsson B, et al. Double-blind, placebo-controlled comparison of rabeprazole 20 mg vs. omeprazole 20 mg in the treatment of erosive or ulcerative gastro-oesophageal reflux disease. Aliment Pharmacol Ther. 1999;13:49-57.
31. Bochenek WJ, Mack ME, Fraga PD, Metz DC. Pantoprazole provides rapid and sustained symptomatic relief in patients treated for erosive oesophagitis. Aliment Pharmacol Ther. 2004;20:1105-1114.
32. Richter JE, Bochenek W. Oral pantoprazole for erosive esophagitis: a placebo-controlled, randomized clinical trial. Pantoprazole US GERD Study Group. Am J Gastroenterol. 2000;95:3071-3080.
33. Sontag SJ, Hirschowitz BI, Holt S, et al. Two doses of omeprazole versus placebo in symptomatic erosive esophagitis: the U.S. Multicenter Study. Gastroenterology. 1992;102:109-118.
34. Kahrilas PJ, Falk GW, Johnson DA, et al. Esomeprazole improves healing and symptom resolution as compared with omeprazole in reflux oesophagitis patients: a randomized controlled trial. The Esomeprazole Study Investigators. Aliment Pharmacol Ther. 2000;14:1249-1258.
35. Richter JE, Kahrilas PJ, Johanson J, et al. Efficacy and safety of esomeprazole compared with omeprazole in GERD patients with erosive esophagitis: a randomized controlled trial. Am J Gastroenterol. 2001;96:656-665.
36. Castell DO, Kahrilas PJ, Richter JE, et al. Esomeprazole (40 mg) compared with lansoprazole (30 mg) in the treatment of erosive esophagitis. Am J Gastroenterol. 2002;97:575-583.
37. Fennerty MB, Johanson JF, Hwang C, Sostek M. Efficacy of esomeprazole 40 mg vs. lansoprazole 30 mg for healing moderate to severe erosive oesophagitis. Aliment Pharmacol Ther. 2005;21:455-463.
38. Lightdale CJ, Schmitt C, Hwang C, Hamelin B. A multicenter, randomized, double-blind, 8-week comparative trial of low-dose esomeprazole (20 mg) and standard-dose omeprazole (20 mg) in patients with erosive esophagitis. Dig Dis Sci. 2006;51:852-857.
39. Schmitt C, Lightdale CJ, Hwang C, Hamelin B. A multicenter, randomized, double-blind, 8-week comparative trial of standard doses of esomeprazole (40 mg) and omeprazole (20 mg) for the treatment of erosive esophagitis. Dig Dis Sci. 2006;41:844-850.
40. Tutuian R, Katz PO, Castell DO. Nocturnal acid breakthrough: pH, drugs and bugs. Eur J Gastroenterol Hepatol. 2004;16:441-443.
41. Katz PO, Koch FK, Ballard ED, et al. Comparison of the effects of immediate-release omeprazole oral suspension, delayed-release lansoprazole capsules and delayed-release esomeprazole capsules on nocturnal gastric acidity after bedtime dosing in patients with night-time GERD symptoms. Aliment Pharmacol Ther. 2007;15:197-205.
42. Katz PO, Hatlebakk JG, Castell DO. Gastric acidity and acid breakthrough with twice-daily omeprazole or lansoprazole. Aliment Pharmacol Ther. 2000;14:709-714.
43. Fackler WK, Ours TM, Vaezi MF, Richter JE. Long-term effect of H2RA therapy on nocturnal gastric acid breakthrough. Gastroenterology. 2002;122:625-632.
44. Rackoff A, Agrawal A, Hila A, et al. Histamine-2 receptor antagonists at night improve gastroesophageal reflux disease symptoms for patients on proton pump inhibitor therapy. Dis Esophagus. 2005;18:370-373.
45. Robinson M, Rodriguez-Stanley S, Ciociola AA, et al. Control of nocturnal gastric acidity: a role for low dose bedtime ranitidine to supplement daily omeprazole. Dig Dis Sci. 2002;47:265-273.
46. FDA drug safety communication: possible increased risk of fractures of the hip, wrist, and spine with the use of proton pump inhibitors.
www.fda.gov/Drugs/DrugSafety/
47. FDA drug safety communication: low magnesium levels can be associated with long-term use of proton pump inhibitor drugs (PPIs).
www.fda.gov/Drugs/DrugSafety/
To comment on this article, contact rdavidson@uspharmacist.com.





