US Pharm. 2012;37(3):37-42.
Autism spectrum disorders (ASDs) are a group of developmental disabilities in which patients have delayed development in skills associated with socialization, communication, and overall behavior. The broader term of ASD includes Pervasive Developmental Disorder-Not Otherwise Specified (PDD-NOS), Asperger’s Disorder, and Autistic Disorder (AD). Currently all three of these disorders have their own separate diagnostic criteria. Patients with PDD-NOS will meet some, but not all, of the diagnostic criteria for AD and will generally have milder symptoms compared to AD patients.1 Patients with Asperger’s Disorder have unusual interests and behaviors and social challenges, but do not have intellectual deficits.1 Patients with AD, however, have both social as well as intellectual impairments. According to the American Psychiatric Association’s Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition, Text Revision (DSM-IV-TR), the diagnosis of AD requires1:
• Qualitative impairment in social function and communication.
• Restricted, repetitive, and stereotyped patterns of behavior, interests, and activities.
• Delays in or abnormal functioning.
• Exclusion of Rett’s Disorder or Childhood Disintegrative Disorder diagnosis.
Despite these structured characteristics needed for diagnosis, patients can have a wide variety of symptoms. They can be highly functioning students who successfully complete a college degree and possess mildly inadequate social skills, or they can be nonverbal individuals with severe mental retardation.2 TABLE 1 lists the DSM-IV-TR diagnostic criteria for AD.1

The prevalence of patients with ASDs has risen over the past few decades. As of 2006, it has been estimated that an average of one in 110 children in the United States has an ASD, with approximately 730,000 individuals under 21 years of age diagnosed with an ASD.3 Instead of being a true increase in the number of patients with ASDs, this rise could be due to policy and practice changes, which improves awareness on the spectrum of the disorders and diagnoses of ASDs.4
The DSM-V is scheduled to be released in 2013. One of the major revisions includes the diagnostic criteria for AD. Under the proposed revisions, the three diagnoses of AD, Asperger’s Disorder, and PDD-NOS will be consolidated under one category of ASD. Several experts are concerned that this may potentially diagnose fewer patients with AD because of more stringent diagnostic criteria. Physicians involved in the DSM taskforce who are proposing the revisions state that the new diagnostic tool will provide clarity and not decrease the number of patients being diagnosed. The process will be carefully monitored.5
Risk Factors And Pathophysiology
Over the past several decades, there has been a considerable amount of research to determine the risk factors and underlying pathophysiology of ASDs, including AD. While still somewhat controversial, current evidence supports a major genetic component and environmental factors playing an important role in the etiology of autism.6
Genetic Links: The ratio of males to females diagnosed with AD is approximately three to one.2 Since the diagnosis is more common in males, this has led to discussion that AD could be an X-linked disorder. Data to support this hypothesis are inconsistent with cases of male-to-male transmission of AD within families, which would argue against this theory.2 But it is also known that when assessing patients with AD who have other comorbid genetic diseases, one of the more frequently shared diseases is Fragile X Syndrome (FXS). It is an X-linked disorder and the most common cause for inherited intellectual disability.7 Just like those with AD, patients with FXS include a wide spectrum of symptoms, with the most consistent one being developmental delay. Approximately 30% of patients with FXS are on the autism spectrum; however, this population still only comprises a small percentage of patients who are diagnosed with an ASD.2
Further potential genetic links have been identified when looking at the association of AD between siblings. The rate of recurrence is 2% to 8% among children with siblings who have been diagnosed with AD. When comparing monozygotic to dizygotic twins, there is a concordance of greater than 60% in monozygotic twins, with almost no concordance in dizygotic twins.8,9 However, the fact that the concordance is not 100% suggests that there may be some aspect of environmental influence to the disorder.2
Environmental Factors: Similar to the ambiguity of the genetic links discussed above, there are no definitive environmental factors known to be linked to the development of an ASD. Studies since 1979 have investigated the link of ASD with environmental factors showing inconclusive results. Potential risk factors in the studies that were identified include advanced maternal age, low birth weight, breech presentation, and respiratory distress syndrome, but no single factor was consistently recognized in all of the studies.10-17
A recent study conducted out of Atlanta, Georgia, focused on estimating the birth weight- and gestational age-specific risks of autism.18 The authors found that when compared to control patients who were not receiving special education services, there was a 2.3-fold increased risk of autism in children who were born weighing <2.5 kg. Furthermore, after stratifying according to gestational age, the risk of autism in preterm infants (<37 weeks’ gestation) weighing <2 kg was elevated compared to control patients, but was not statistically significant.18
Another study from Utah looked more generally at the prenatal, perinatal, and neonatal risk factors associated with the development of an ASD.19 By using a population-based, nested case-control design, they were able to find a statistically significant association between increased maternal age (>35 years old) and a risk of ASDs. They also found a significant link between the risk for ASDs and the baby being in the breeched position prior to birth. However, when analyzing neonatal risk factors such as birth weight, 5-minute Apgar scores (a scoring system used to rapidly assess the status of a newborn),20 congenital anomalies, and assisted ventilation for greater than 30 minutes, they did not find an association with an increased risk of AD.19
Despite the results of these trials, there is still no consensus on what risk factors truly have a link to the development of an ASD.
Vaccines: In addition to the potential risk factors stated above, more commonly discussed is the link between vaccines and ASDs. Of significant concern were vaccines that included a mercury-containing compound called thimerosal that was added in order to protect multiple-dose vials from bacterial contamination.21 Although there was no evidence that the low doses of thimerosal in vaccines caused harm in pediatrics, in 1999 the American Academy of Pediatrics (AAP) and the Public Health Service agencies, along with the vaccine manufacturers, agreed to eliminate or reduce the content of thimerosal in vaccines as a precautionary measure.22
In 2010, Lewis et al compared children ages 6 to 13 receiving mercury exposure from thimerosal-containing products, including vaccines, with any of the three ASD outcomes, including ASD, AD, and ASD with regression.23 The study included a total of 256 case-children and 752 matched controls and found no evidence that there was an increased risk of ASD, AD, or ASD with regression. To further support the argument that thimerosal does not contribute to the development of an ASD, it is now known that the rate of diagnosis of ASDs has continued to climb despite the removal of thimerosal from vaccines more than a decade ago.24
More specifically, the measles, mumps, and rubella (MMR) vaccine has been most commonly selected as having a link with increasing rates of autism. In 1998, Wakefield et al published an article in The Lancet that suggested a link between the MMR vaccine and autism.25 However, in 2004, 10 out of the original 12 authors printed a retraction that acknowledged that there was insufficient evidence to establish the previously stated causal link,26 and in 2010 the editors of the journal officially published a retraction of the original article, again emphasizing that several elements presented in the article were incorrect.27
In 2002, Madsen et al published the results of their retrospective cohort study that was designed to evaluate the relationship between autism and the MMR vaccine in a population of children who were born in Denmark.28 A total of 537,303 children were included in the cohort. Eighty-two percent of the included patients were vaccinated with the MMR vaccine and a total of 316 children were diagnosed with AD, with an additional 422 children being diagnosed with other ASDs. After adjusting for age, calendar period, sex, birth weight, gestational age, mother’s education, and socioeconomic status, they found that when compared to unvaccinated children, there was no increase in the risk of AD or other ASDs among those children who were vaccinated.28
Goals of Treatment
Goals of therapy focus on maximizing the patient’s independence and quality of life and alleviating family stress.29 Furthermore, core features of the disorder, including deficits in communication and social skills, are minimized. The education and support provided to families, in addition to facilitating learning and development of the patient, can help accomplish these goals.
Nonpharmacologic Treatment Options
Due to the incurable nature of AD, effective chronic management options are essential to effective treatment of the disorder.29 Currently, the cornerstone of therapy focuses on nonpharmacologic treatments, including behavioral therapy, with the addition of pharmacologic agents if needed. Due to the importance of early intervention in this patient population, the Education of All Handicapped Children Act of 1975 made the education of patients with AD a public responsibility.30 The National Research Council formed the Committee on Educational Interventions for Children with Autism in order to evaluate the current literature regarding treatment options and provide appropriate recommendations for public policy.31
While there are several different methodologies on effective behavioral therapy for patients with AD currently available, two of the primary resources are Applied Behavior Analysis (ABA) and the Treatment and Education of Autistic and Related Communication Handicapped Children (TEACCH) program. ABA uses psychological conditioning to systematically teach skills associated with social, communication, and behavioral management. It utilizes positive reinforcement to teach new behaviors, and addresses destructive behavior that can be harmful to the patient as well as others by analyzing the underlying factors to make an effective behavioral modification.21,29,32 ABA also longitudinally evaluates the effectiveness of the therapy so that the interventions can be modified according to the progress being made. Ideally, treatment is started as soon as possible after diagnosis and requires 20 hours of intervention per week for patients to achieve the maximum results.21
On the other hand, the TEACCH program focuses on a specific strength of patients with AD. For example, the processing of visual information is emphasized in contrast to verbal communication, which is more likely to be lacking in this patient population.21,32 The program utilizes controlled and organized classrooms that have structured learning activities that are broken down into visually organized steps.
One of the landmark studies carried out regarding behavioral therapy for patients with AD was conducted by Lovaas in 1987.33 The study looked at the results of behavioral modification treatment for two groups of similar preschool-aged children. Of the 19 children who were treated with intensive behavioral therapy, 47% achieved normal intellectual and educational functioning compared to the control group of 40 children, where only 2% achieved the same result.33
A follow-up study was conducted in 1993 by McEachin et al following the same 19 patients at a mean age of 11.5 years.34 Their study showed that the intensive behavioral therapy group retained its gains over the control group, signifying that behavioral treatment can produce long-lasting improvements for many children who have AD.34
While younger children are usually the focus of targeted behavioral therapy, it is important to keep in mind that as these patients grow into adolescence and adulthood, they will generally need continual support. Patients with AD may need assistance in finding meaningful employment, and they often have the opportunity to live in group homes when their families can no longer provide support or shelter.35 If the network of support is continued, it will help decrease the number of patients with AD requiring long-term facilities in the future.
Pharmacologic Treatment Options
The use of pharmacologic agents in patients who have ASDs generally focuses on alleviating certain behavioral symptoms such as irritability, hyperactivity, aggression, and other self-injurious behaviors that can be detrimental to the child’s ability to learn, as well as to their family environment. The percentage of patients with AD who exhibit aggressive behaviors has not yet been evaluated; however, one study found that approximately 20% of patients with PDD experience moderate-to-severe irritability.36 Currently, there are only two FDA-approved medications for treating irritability associated with autism. These drugs, risperidone and aripiprazole, are both classified as atypical antipsychotics, which work by blocking postsynaptic dopamine and serotonin receptors. TABLE 2 provides a summary of the dosing and adverse effects for both agents.37,38
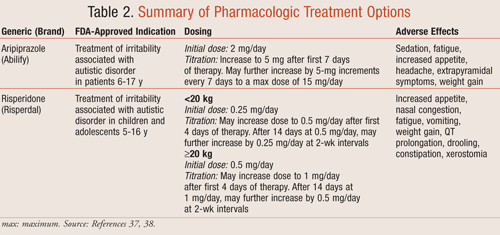
Risperidone: This drug was approved in October 2006 by the FDA for the treatment of irritability associated with autism in children and adolescents 5 to 16 years of age.37 Risperidone, an atypical antipsychotic, antagonizes both serotonin as well as dopaminergic receptors, with a greater affinity for the serotonin receptors. The decision to approve risperidone for the treatment of autism came after the publication of a few key studies. One of the main trials included an 8-week randomized treatment period followed by a 4-month open-label treatment and a 2-month discontinuation phase.39 The study was designed to look for a decrease in the Aberrant Behavior Checklist (ABC) irritability subscale score that asked questions related to aggression, self-injury, tantrums, agitations, and unstable mood. The first treatment period included 101 children 5 to 17 years old who were randomized to receive either placebo or risperidone. Risperidone was initiated at a dose of 0.25 mg at bedtime for patients that weighed <20 kg and 0.5 mg for patients who weighed ≥20 kg. The dose could be titrated appropriately until day 29 of the study. The results showed a 56.9% decrease in the mean irritability score for patients receiving risperidone compared to a 14.1% decrease in the placebo group, a difference which was statistically significant (P <.001). Adverse events reported were mostly mild, and included increased appetite, nasal congestion, fatigue, vomiting, weight gain, drooling, constipation, and xerostomia.39 While not seen in this study, QT prolongation can also be a concern when prescribing risperidone to patients.37
The extension trial was an open-label study and included 63 patients who showed a positive response to risperidone or who were nonresponders to placebo.40 Treatment with risperidone was continued for an additional 4 months, after which 38 patients enrolled in the 2-month discontinuation phase, where they were randomized to either continue treatment with risperidone or be transitioned over to placebo. Results from the 4-month treatment period continued to show a significant improvement in symptoms for 80% of patients, with continued tolerability as well. The discontinuation phase demonstrated that as patients came off the risperidone, they started to show a return of aggressive behavior, signifying that continuation of medication can help maintain the control of symptoms. While this was an extension trial, patients were still only treated for a maximum of 8 months, so long-term efficacy and tolerability are unknown.40 Additional trials involving risperidone have also shown promising data in patients who suffer from aggressive behavior associated with ASDs.41,42
Aripiprazole: In November 2009, after two randomized controlled trials, the FDA approved aripiprazole for the treatment of irritability associated with ASDs in patients 6 to 17 years of age.38 Aripiprazole is also an atypical antipsychotic that is a functional antagonist of specific serotonin receptors. In addition, it has partial agonist activity at other serotonin and dopaminergic receptors. Similar to risperidone, these randomized, placebo-controlled trials were designed to measure the mean change from baseline to endpoint in the caregiver-rated ABC irritability subscale score.
The first trial was a fixed-dose study that included 218 patients, aged 6 to 17 years, who were randomized to receive either placebo or aripiprazole 5, 10, or 15 mg/day for the total treatment duration of 8 weeks.43 The results of this study showed that for the primary outcome, patients who received placebo had a mean decrease on the irritability subscale score of 8.4 compared to a decrease of 12.4 for patients who received aripiprazole 5 mg/day, 13.2 for patients who received 10 mg/day, and 14.4 for patients who received 15 mg/day. All of these values were statistically significant (P <.05). Eighty-four percent of patients reported one adverse event during the study; however, most of them were considered mild to moderate and included sedation, fatigue, increased appetite, headache, extrapyramidal symptoms, and weight gain.43 While this short-term study showed the benefit of aripiprazole in this unique patient population, the long-term effects, favorable and unfavorable, are still widely unknown.
The second trial was a flexible-dose study that randomized 98 patients, aged 6 to 17 years, to receive either placebo or aripiprazole for the total treatment duration of 8 weeks.44 For patients who received aripiprazole, their dose was initiated at 2 mg/day and was titrated to a target dose of 5, 10, or 15 mg/day, as deemed appropriate by the investigator, making sure that the dose remained stable for the final 2 weeks of treatment. Similar to the first study, the results showed a statistically significant difference with a mean decrease from baseline on the irritability subscale score of 12.9 for patients who received aripiprazole compared to 5 for patients who received placebo. This study also demonstrated that a majority of patients experienced at least one adverse event, generally mild in nature and similar to those listed above. This trial had the advantage of administering only 5 or 10 mg/day of aripiprazole to the majority of patients during the last 2 weeks of treatment, suggesting that these lower doses might be sufficient to achieve the desired results.44 Information regarding long-term efficacy and safety is still lacking; however, aripiprazole does have a black box warning regarding the increased risk of suicidal thinking and behavior in children, adolescents, and young adults with major depressive disorder and other psychiatric disorders.38
Off-label Use: Other medications have also been studied for the treatment of repetitive and aggressive-type behaviors in ASDs. These drugs include the selective serotonin reuptake inhibitors (SSRIs) fluoxetine and fluvoxamine as well as atomoxetine, a selective norepinephrine reuptake inhibitor (SNRI).29,45 Furthermore, the alpha2-adrenergic agonists guanfacine and clonidine have also been investigated for the treatment of patients with ASDs.29,45 While these drugs have shown promise in a limited number of trials, the data are more restricted, and these agents are not FDA approved for these indications in patients with ASDs.
Conclusion
While there are several theories concerning the main causes of ASDs, the data remain somewhat controversial but support a pathway that is most likely a combination of both genetic links and environmental factors. Treatment for patients with ASDs should focus on early intervention with behavioral modification therapy, with the addition of pharmacologic agents if necessary to help manage aggressive-type behaviors. Pharmacists can play a key role in educating families on the different medications available and how to take them appropriately, if prescribed.
REFERENCES
1. American Psychiatric Association. Pervasive developmental disorders. In: Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition, Text Revision (DSM-IV-TR). Washington, DC: American Psychiatric Association; 2000:69-70.
2. Muhle R, Trentacoste SV, Rapin I. The genetics of autism. Pediatrics. 2004;113:e472-e486.
3. Autism and Developmental Disabilities Monitoring
Network Surveillance Year 2006 Principal Investigators. Prevalence of
autism spectrum disorders—Autism and Developmental Disabilities
Monitoring Network, United States, 2006. MMWR Surveill Summ. 2009;18:1-20.
4. Levy SE, Mandell DS, Schultz RT. Autism. Lancet. 2009;374:1627-1638.
5. Carey B. New definition of autism will exclude many, study suggests. NY Times.
January 19, 2012.
www.nytimes.com/2012/01/20/health/research/new-autism-definition-would-exclude-many-study-suggests.html?pagewanted=all.
Accessed February 15, 2012.
6. Johnson CP, Myers SM. Identification and evaluation of children with autism spectrum disorders. Pediatrics. 2007;120:1183-1215.
7. De Rubeis S, Bagni C. Regulation of molecular pathways in the Fragile X Syndrome: insights into autism spectrum disorders. J Neurodev Disord. 2011;3:257-269.
8. Bailey A, Le Couteur A, Gottesman I, et al. Autism as a strongly genetic disorder: evidence from a British twin study. Psychol Med. 1995;25:63-77.
9. Steffenburg S, Gillberg C, Hellgren L, et al. A twin study of autism in Denmark, Finland, Iceland, Norway and Sweden. J Child Psychol Psychiatry. 1989;30:405-416.
10. Croen LA, Najjar DV, Fireman B, et al. Maternal and paternal age and risk of autism spectrum disorders. Arch Pediatr Adolesc Med. 2007;161:334-340.
11. Larsson HJ, Eaton WW, Madsen KM, et al. Risk factors
for autism: perinatal factors, parental psychiatric history, and
socioeconomic status. Am J Epidemiol. 2004;161:916-925.
12. Mason-Brothers A, Ritvo ER, Pingree C, et al. The
UCLA-University of Utah epidemiologic survey of autism: prenatal,
perinatal, and postnatal factors. Pediatrics. 1990;86:514-519.
13. Hultman CM, Sparen P, Cnattingius S. Perinatal risk factors for infantile autism. Epidemiology. 2002;13:417-423.
14. Burd L, Severud R, Kerbeshian J, et al. Prenatal and perinatal risk factors for autism. J Perinat Med. 1999;27:441-450.
15. Bryson SE, Smith IM, Eastwood D. Obstetrical suboptimality in autistic children. J Am Acad Child Adolesc Psychiatry. 1988;27:418-422.
16. Finegan J, Quarrington B. Pre-, peri-, and neonatal factors and infantile autism. J Child Psychol Psychiatry. 1979;20:119-128.
17. Gillberg C, Gillberg IC. Infantile autism: a total
population study of reduced optimality in the pre-, peri-, and neonatal
period. J Autism Dev Disord. 1983;13:153-166.
18. Schendel D, Bhasin TK. Birth weight and gestational
age characteristics of children with autism, including a comparison with
other developmental disabilities. Pediatrics. 2008;121:1155-1164.
19. Bilder D, Pinborough-Zimmerman J, Miller J, et al.
Prenatal, perinatal, and neonatal factors associated with autism
spectrum disorders. Pediatrics. 2009;123:1293-1300.
20. Committee on Obstetric Practice and Committee on Fetus and Newborn. The Apgar Score. Pediatrics. 2006;117:1444-1447.
21. Barbaresi WJ, Katusic SK, Voigt RG. Autism: a review
of the state of the science for pediatric primary health care
clinicians. Arch Pediatr Adolesc Med. 2006;160:1167-1175.
22. CDC. Thimerosal. www.cdc.gov/vaccinesafety/Concerns/thimerosal. Accessed October 8, 2011.
23. Lewis E, Bernal P, Shay D, et al. Prenatal and infant
exposure to thimerosal from vaccines and immunoglobulins and risk of
autism. Pediatrics. 2010;126:656-664.
24. Madsen KM, Lauritsen MB, Pedersen CB, et al.
Thimerosal and the occurrence of autism: negative ecological evidence
from Danish population-based data. Pediatrics. 2003;112:603-606.
25. Wakefield AJ, Murch SH, Anthony A, et al.
Ileal-lymphoid-nodular hyperplasia, non-specific colitis, and pervasive
developmental disorder in children. Lancet. 1998;351:637-641.
26. Murch SH, Anthony A, Casson DH, et al. Retraction of an interpretation. Lancet. 2004;363:750.
27. Retraction—Ileal-lymphoid-nodular hyperplasia, non-specific colitis, and pervasive developmental disorder in children. Lancet. 2010;375:445.
28. Madsen KM, Hviid A, Vestergaard M, et al. A population-based study of measles, mumps, and rubella vaccination and autism. N Engl J Med. 2002;347:1477-1482.
29. Myers SM, Johnson CP. Management of children with autism spectrum disorders. Pediatrics. 2007;120:1162-1182.
30. Campbell M, Schopler E, Cueva J, et al. Treatment of autistic disorder. J Am Acad Child Adolesc Psychiatry. 1996;32:134-143.
31. National Research Council. Educating Children with Autism. Washington, DC: National Academy Press; 2001.
32. Rogers SJ, Vismara LA. Evidence-based comprehensive treatments for early autism. J Clin Child Adolesc Psychol. 2008;37:8-38.
33. Lovaas OI. Behavioral treatment and normal educational and intellectual functioning in young autistic children. J Consult Clin Psychol. 1987;55:3-9.
34. McEachin JJ, Smith T, Lovaas OI. Long-term outcome for
children with autism who received early intensive behavioral treatment.
Am J Ment Retard. 1993;97:359-372.
35. Rapin I. Autism. N Engl J Med. 1997;337:97-104.
36. Lecavalier L. Behavioral and emotional problems in
young people with pervasive developmental disorders: relative
prevalence, effects of subject characteristics, and empirical
classification. J Autism Dev Disord. 2006;36:1101-1114.
37. Risperdal (risperidone) package insert. Titusville, NJ: Ortho-McNeil-Janssen Pharmaceuticals, Inc; 2011.
38. Abilify (aripiprazole) package insert. Tokyo, Japan: Otsuka Pharmaceutical Co, Ltd; 2011.
39. Research Units on Pediatric Psychopharmacology Autism
Network. Risperidone in children with autism and serious behavioral
problems. N Engl J Med. 2002;347:314-321.
40. Research Units on Pediatric Psychopharmacology Autism
Network. Risperidone treatment of autistic disorder: longer-term
benefits and blinded discontinuation after 6 months. Am J Psychiatry. 2005;162:1361-1369.
41. Shea S, Turgay A, Carroll A, et al. Risperidone in the
treatment of disruptive behavioral symptoms in children with autistic
and other pervasive developmental disorders. Pediatrics. 2004;114:e634-e641.
42. Lemmon ME, Gregas M, Jeste SS. Risperidone use in
autism spectrum disorders: a retrospective review of a clinic-referred
patient population. J Child Neurol. 2011;26:428-432.
43. Marcus RN, Owen R, Kamen L, et al. A
placebo-controlled, fixed-dose study of aripiprazole in children and
adolescents with irritability associated with autistic disorder. J Am Acad Child Adolesc Psychiatry. 2009;48:1110-1119.
44. Owen R, Sikich L, Marcus RN, et al. Aripiprazole in
the treatment of irritability in children and adolescents with autistic
disorder. Pediatrics. 2009;124:1533-1540.
45. Wink LK, Erickson CA, McDougle CJ. Pharmacologic
treatment of behavioral symptoms associated with autism and other
pervasive developmental disorders. Curr Treat Options Neurol. 2010;12:529-538.
To comment on this article, contact rdavidson@uspharmacist.com.





