US Pharm. 2012;37(9):Epub.
Interstitial cystitis (IC) is a bladder pain syndrome whose exact cause remains unknown. About 1.2 million people in the United States have IC, most of them female (197 women vs. 41 men in 100,000).1-4 Typically, IC onset occurs in an individual’s mid-40s, but one study noted that 25% of patients were younger than 30 years.5 Because the clinical hallmarks of IC (urinary urgency and frequency, bladder pain, nocturia, dysuria) are not specific, misdiagnosis or delayed diagnosis of this syndrome is common. Most patients have symptoms for 2 to 7 years and visit several health care providers before receiving an accurate diagnosis.6 Outpatient-clinic visits for management of IC incurred about $66 million in health care expenditures in 2000.4,5
The impact of IC on quality of life (QOL) is significant, considering the associated pain and urinary urgency and frequency, which may curtail daily activities. Patients may experience interrupted sleep, leading to fatigue and depression. Additionally, IC may cause pain during sexual intercourse, thereby affecting sexual intimacy.
Pathophysiology
Various theories have been developed to explain IC pathophysiology, but none of them can entirely explain why patients get IC. Most clinicians agree that IC is caused by multiple factors, such as defective bladder lining, histamines released by mast cells, autoimmune conditions, and other situations (e.g., bladder trauma, bladder overdistention, pelvic-floor dysfunction, spinal cord injury).4-6 The following are descriptions of theories about IC pathophysiology.
Defective Bladder Lining: The most favored theory holds that a defect in the bladder lining could allow waste in the urine (e.g., potassium ions) to infiltrate the nerves, causing pain and stimulating frequent voiding. One study found that the urine of IC patients had a lower potassium:creatinine ratio, which supports the theory of permeability resulting in increased potassium diffusion.7 However, this theory cannot explain every case of IC, because only 50% of IC patients had bladder-lining damage from petechial hemorrhage (glomerulation, or pinpoint area of bleeding in the bladder wall) and only 10% had Hunner’s lesion (reddened mucosal area with vessels radiating toward a central scar on the bladder surface).3,5
Mast Cell Theory: IC patients have an increased number of mast cells in the bladder wall.8 Recent studies have found that mast cells containing histamine are involved in a variety of neural inflammatory diseases.6 The presence of a bladder insult or stimulation by acetylcholine, stress, and hormone fluctuation activates mast cells, and histamine is released. This histamine release triggers an inflammation response, causing smooth-muscle contraction, local tissue damage, and nerve stimulation. Over time, repetition of these events may further damage the bladder lining, leading to increased urgency, frequency, and pain.5 Again, this theory cannot explain all cases of IC, and the use of antihistamines to treat IC produces suboptimal results.
Autoimmune Theory: Some clinicians believe that immune-system dysregulation contributes to IC development because some clinical features of IC resemble those seen in the dysregulated immune system. Additionally, IC has unexplained associations with autoimmune diseases and the pain syndrome involved in vulvar vestibulitis, fibromyalgia, Hashimoto’s thyroiditis, and irritable bowel syndrome.1,6 A retrospective study revealed that 97% of IC patients and 93% of endometriosis patients had chronic pelvic pain.5 The similarly high percentages of chronic pain among IC patients and endometriosis patients reinforce the belief that immune-system dysregulation is a contributing factor to IC.
Signs and Symptoms
IC patients typically experience urinary urgency, urinary frequency, and bladder pain.3-6 With urinary urgency, the patient senses a strong need to void immediately; pain or spasms may or may not accompany this sensation.3-6 Urinary urgency leads to urinary frequency, with IC patients typically voiding many times per day. On average, IC patients void up to 16 times per day, but the urinary volume is less than that of non-IC patients. Urinary urgency often creates the need to get up at night to urinate (nocturia), resulting in insomnia and fatigue.3-6
Typically (in about 70% of patients), early-stage IC involves the presence of only one symptom: either urinary urgency or urinary frequency.9 Over time, however, bladder pain may develop and become a dominant symptom. Bladder pain may manifest in different locations, such as the suprapubic or perineal area, vagina, lower back, or medial aspect of the thigh.5 It is difficult for the patient to pinpoint exactly where the pain originates, which could lead to the mistaken belief that the pain indicates a urinary tract infection (UTI). One characteristic of IC is that the pain is relieved by voiding, but often recurs once the bladder fills with urine.5
Diagnosis
For initial screening and simple cases of IC, diagnosis is based on signs and symptoms and the exclusion of other bladder disorders, such as bladder cancer, overactive bladder (OAB), and UTI. Since IC symptoms are not specific, it is extremely important for the clinician to differentiate IC symptoms from those of other bladder disorders (TABLE 1).1,4,5 Bladder cancer shares the typical three IC symptoms, but unlike IC, it is also associated with hematuria and dysuria. OAB patients have urinary frequency and urgency, but no bladder pain. UTI involves the three typical IC symptoms, but bacteriuria, hematuria, and dysuria are also present. In acute uncomplicated cystitis, patients may experience urinary frequency and urgency and pain or pressure around the bladder, but the urine contains bacteria and/or pus.10-13

Basic assessments include a careful review of medical history, a thorough physical examination, and a laboratory workup to identify IC symptoms and rule out other bladder-related pathology. The medical history should include chief complaints; onset, duration, characteristics, and location of symptoms; and relieving and/or aggravating factors. In women, the physical examination should include a pelvic examination, as they may have tenderness at the bladder neck and anterior vaginal wall; pelvic-floor muscle strength should also be evaluated because 81% of IC patients have high-tone pelvic-floor muscle dysfunction.14,15
Baseline voiding symptoms and pain levels should be obtained for future reference and treatment monitoring. The patient should keep a voiding diary for 3 days to accurately record fluid intake and urine output; establish severity of frequency and urgency; determine number of daily voids and voided volumes; and identify which foods may worsen symptoms. Acidic and potassium-containing foods trigger symptoms in 50% of IC patients. Some foods that may worsen symptoms are alcohol, caffeine, carbonated drinks, spicy foods, chocolate, tomatoes, yogurt, bananas, apples, citrus fruits and juices, dried beans, spinach, and artificial sweeteners. Patients should test these foods to determine which ones trigger IC symptoms.6,15
Clinicians typically use symptom and problem questionnaires to screen for IC. The O’Leary-Sant (OLS) questionnaire includes a symptom index to measure urgency and pain and a problem index to measure the degree to which the patient experiences each symptom.16 OLS is also an effective tool for following up and monitoring response to treatment.5,17 The pelvic pain and urgency/frequency (PUF) scale is another tool used to evaluate the presence and severity of IC symptoms; it focuses on urinary urgency, urinary frequency, and pelvic pain and quantifies symptoms related to sexual intercourse.18 For patients in whom IC is suspected, the intravesical potassium sensitivity test (PST; discussed below) is used to confirm the diagnosis.19 In one study, PST was positive in 74% of patients with a PUF score of 10–14, 76% of those with a score of 15–19, and 91% of those with a score ≥20; if the total score exceeded 5 over 35, then 55% of cases had IC.5,18 One study found that OLS was more efficient than PUF for detecting IC.17
Differential laboratory tests include urinalysis, urine culture to exclude UTI, urine cytology to rule out carcinoma, and vaginal culture to exclude Chlamydia, herpes, or other sexually transmitted diseases.1,5,6
For more complicated IC cases in which initial screening cannot confirm IC, urodynamic testing, PST, and cystoscopy with hydrodistention should be performed.
Urodynamic Testing: A series of tests is performed to evaluate urinary function during bladder filling, storage, and emptying. Of these tests, the cystometrogram, or filling study, is the most important because it quantifies maximum bladder capacity, sensory urgency, and pain.6 IC patients generally have reduced bladder capacity (<350 mL) compared with non-IC patients, except in the early phase.5,6
PST: This test compares pain and urgency levels before and after instillation of two solutions into the bladder. First, 40 mL of sterile water is instilled over 2 to 3 minutes; then the water is drained and replaced with a solution of potassium chloride 0.4 M.19 Patient urgency and pain are graded from 0–5 before and after the instillations. If the total score is greater than 2 over 5, then the test is positive for IC. About 81% of IC patients have a positive test, versus fewer than 3% of non-IC patients.6,14 PST is 100% false-positive in cases of acute bacterial cystitis, radiation cystitis, and UTIs.5 The test is useful in early-phase IC because such patients may not yet be experiencing pain. PST may reveal urgency in 40% of IC patients and pain in 28%. A negative PST means that the patient may not have IC; may have IC, but does not have epithelial permeability problems; or has IC plus an epithelial permeability problem, but the test is not positive because of recent intravesical treatment involving dimethyl sulfoxide (DMSO) hydrodistention.
Cystoscopy With Hydrodistention: This procedure is performed only in patients with hematuria and/or abnormal cytology, to differentiate IC from other bladder-related diseases. Water is instilled into the bladder to a pressure of 80 to 100 cm H2O. When the water is drained from the bladder, a patient with IC often shows glomerulations. For a diagnosis of IC, the glomerulations must be diffused and located in at least three bladder quadrants.6
Treatment
To optimize IC treatment, clinicians should adhere to certain principles. First, the goal is to alleviate or minimize symptoms. The most conservative treatment should be implemented first. Initial treatment type and level of treatment should correspond to symptom severity, clinical judgment, and patient preferences. For multiple treatments, baseline symptom measurement and regular assessment are critical to monitor and document the efficacy of combined versus single treatment. Ineffective treatments should be stopped once a clinically meaningful interval has elapsed. Pain management and its effect on QOL should be considered and regularly assessed. Finally, if no improvement in symptoms is observed after various treatments have been tried, the diagnosis of IC should be reevaluated.15
Treatment should be initiated with the most conservative regimen (i.e., least risk of side effects) and progress sequentially to the least conservative (i.e., greatest risk). Optimally, risks for adverse drug reactions (ADRs) should be minimized, and benefits maximized. TABLE 2 lists treatment regimens ranked from most conservative to least conservative based on the 2011 American Urological Association guidelines.15
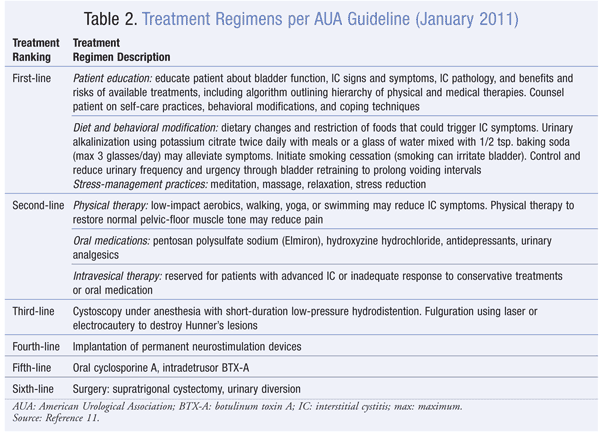
First-Line: For initial-phase IC, first-line treatment should be patient education, diet and behavioral modification, and stress management. As the symptoms worsen over time, oral medication should be added.4-6
Second-Line: The only FDA-approved medication for IC treatment is pentosan polysulfate sodium (Elmiron). This drug, a light-molecular heparinoid, works by adhering to the bladder-wall mucosa to protect tissues from irritating substances in the urine. The dosage is 100 mg orally three times daily, taken 2 hours before or after meals. The most common ADRs are headache, dizziness, alopecia, rash, rectal hemorrhage, diarrhea, nausea, abdominal pain, and dyspepsia. The drug typically requires at least 6 months to achieve some symptom relief. However, early-phase IC patients may experience pain relief in 4 weeks.20 The risks and benefits of use beyond 6 months in unresponsive patients are unknown.
For patients with moderate-to-severe IC, a therapeutic regimen of Elmiron combined with antihistamines, antidepressants, and analgesics may be used.21 Hydroxyzine hydrochloride is another drug used to control IC symptoms, and a combination of pentosan and hydroxyzine has achieved symptom relief in 40% of IC patients.5 Hydroxyzine acts like an antihistamine, but its use is limited by many ADRs (e.g., central nervous system depression, pruritus, rash, urticaria, xerostomia, paresthesia, tremor, blurred vision); therefore, it may be inappropriate for use in geriatric patients.
Since IC patients are three to four times more likely to have depression and suicidal thoughts, antidepressants such as amitriptyline have been used to control depression and pain. Amitriptyline relieves pain by inhibiting histamine secretion. Its reduction of norepinephrine and serotonin reuptake in the peripheral nervous system alleviates frequency and urgency.5 In a randomized, large-scale trial, amitriptyline at a daily dose of 50 mg orally achieved symptom relief in more than 50% of IC patients.4,18,20 The most common side effects are sedation, constipation, and dry mouth.
Common pain medications used for short-term pain relief during an IC symptom flare are phenazopyridine (Pyridium), nonsteroidal anti-inflammatory drugs, and narcotics.6
Intravesical therapy is used alone or in conjunction with oral medications in advanced IC or in cases when a patient has an inadequate response to conservative treatments or oral medications. An agent—usually DMSO—is instilled into the bladder to reduce urgency, frequency, and pain through its anti-inflammatory, analgesic, and antispasmodic activity.1,4,5 At a dosage of 50 mL of 50% DMSO every 1 to 2 weeks for 4 to 8 weeks, this method provides symptom relief, but the pain frequently returns.6 In a randomized, controlled trial, intravesical DMSO had 70% efficacy in reducing symptoms for several months to 1 year. Heparin, also used off label in intravesical therapy, works as a mucosal surface protector with anti-inflammatory and inhibitory effects on bladder scars. One study found that more than 50% of IC patients who received 25,000 U of heparin twice a week intravesically for 3 months showed significant improvement in symptom scores.6
Third-Line: If first- and second-line treatments do not provide desired symptom control and improve QOL, then cystoscopy with hydrodistention may be attempted. The stretch stimulus from hydrodistention increases heparin-binding epidermal growth factor and reduces antiproliferative-factor activity in the urine. As a result, this treatment provides short-term benefit of about 6 months in up to 50% of IC patients.4-6 If Hunner’s lesions are present, fulguration—a procedure using laser or electrocautery—may be performed to burn the lesions. In one study, 100% of patients achieved complete pain relief and 70% achieved reduced or normalized frequency for 2 to 42 months.15
Fourth-Line: InterStim is a surgically implanted neuromodulation device used to control urinary frequency and urgency in IC patients who fail conventional therapy.4,6 InterStim therapy, approved by the FDA in 1997, has proven effective for up to 5 years. The device sends mild electrical impulses via a thin wire lead to the sacral nerves that control the bladder, sphincter, and pelvic-floor muscles. The device may be discontinued at any time, with the condition reverting to that originally present. Patients receiving InterStim therapy experienced more significant QOL improvement than those receiving standard treatments.22,23
Fifth-Line: Cyclosporine A (CyA) may be used orally if previous treatments fail to provide adequate symptom control. In one randomized trial, oral CyA dosed at 3 mg/kg/day resulted in significant clinical improvement after 6 months in 75% of patients versus 19% of patients taking Elmiron. Serious ADRs include immunosuppression and nephrotoxicity.15
Sixth-Line: Cystectomy is a surgical procedure that removes all or part of the bladder. This procedure is rarely used because of its complications and unpredictable results; therefore, it is reserved for severe cases in which all other therapies have failed.6 Supratrigonal cystectomy or urinary diversion may be used.
In supratrigonal cystectomy, a small portion of the bladder is left intact and bowel tissue is used to form a new bladder. This procedure increases the size of the bladder, but it can impair the bladder’s ability to empty properly. Consequently, the patient must insert a catheter once or twice a day to remove the remaining urine, which increases the risk of bladder infection. Additionally, bowel tissue naturally produces mucus, which may block the flow of urine or make self-catheterization more difficult.24
In urinary diversion, a section of the bowel is used to redirect the flow of the urine to a stoma (hole in abdominal wall). The bladder may be left in place or removed entirely, and urine in the stoma is collected in an external bag. This procedure is associated with a lower risk of complications, but an external bag may be inconvenient for the patient. Alternatively, an internal pouch (ileal reservoir) may be created next to the abdominal wall. The pouch is made of bowel tissue and has a small stoma where a catheter can be inserted for draining the pouch. Urinary diversion has a high rate of complications and mucus blockage.25,26
Another surgical procedure for IC uses segments of the small intestine to make a neobladder (storage pouch). The neobladder is connected to the urethra, allowing urine to pass out of the body normally. It may take a few weeks to months for the neobladder to function properly. In a case study, this procedure was performed in 35 female patients with IC for at least 2 years who had failed conservative treatments. All patients showed significant improvement in QOL, and only two (5.7%) were placed on intermittent catheterization.25
Treatments to Avoid: According to the recent guidelines for interstitial cystitis/bladder pain syndrome, the following treatments should be avoided because of lack of efficacy and/or unacceptable side effects: long-term oral antibiotics; intravesical instillation of bacillus Calmette-Guérin outside of investigational study settings; high-pressure, long-duration hydrodistention; and long-term systemic oral glucocorticoids.15
Conclusion
IC is caused by multiple factors and occurs mostly in women. Typical symptoms include urgency, frequency, and bladder pain without signs or symptoms of infection. IC is diagnosed by a thorough medical history and physical examination, along with laboratory tests to rule out other bladder disorders. For optimal treatment, clinicians should implement the most conservative therapies to prevent unnecessary additional risks for patients. Multiple simultaneous treatments should be considered. Additionally, pain management must be applied throughout the course of therapy. Pharmacists can play an important role in optimizing treatment by educating patients about IC and available therapies and collaborating and intervening with the medical team or prescribers to develop the best treatment regimen for the IC patient.
REFERENCES
1. Pontari MA. Seeking a rational approach to the diagnosis and treatment of interstitial cystitis. Medscape Womens Health. 1996;1:2.
2. Clemens JQ, Meenan RT, O’Keeffe Rosetti MC, et al. Prevalence of
interstitial cystitis symptoms in a managed care population. J Urol. 2005;174:576-580.
3. Homma Y, Ueda T, Tomoe H, et al. Clinical guidelines for interstitial cystitis and hypertensive bladder syndrome. Int J Urol. 2009;16:597-615.
4. Association of Reproductive Health Professionals. Screening,
treatment, and management of IC/PBS.
www.arhp.org/publications-and-resources/clinical-proceedings/screening-treatment-and-management-of-icpbs.
Accessed August 17, 2012.
5. Heck BN. Interstitial cystitis: enhancing early identification in primary care settings. J Nurse Pract. 2007;3:509-519.
6. Panzera AK. Interstitial cystitis/painful bladder syndrome. Urol Nurs. 2007;27(1):13-19.
7. Parsons CL, Greene RA, Chung M, et al. Abnormal urinary potassium metabolism in patients with interstitial cystitis. J Urol. 2005;173(4):1182-1185.
8. Theoharides TC, Kempuraj D, Sant GR. Mast cell involvement in
interstitial cystitis: a review of human and experimental evidence. Urology. 2001;57(6 suppl 1):47-55.
9. Porru D, Politanò R, Gerardini M, et al. Different clinical presentation of interstitial cystitis syndrome. Int Urogynecol J Pelvic Floor Dysfunct. 2004;15:198-202.
10. Sexton WJ, Wiegand LR, Correa JJ, et al. Bladder cancer: a review of non-muscle invasive disease. Cancer Control. 2010;17;256-268.
11. Kuo YC, Kuo HC. Potential factors that can be used to
differentiate between interstitial cystitis/painful bladder syndrome and
bladder oversensitivity in women. Int J Clin Pract. 2012;66;146-151.
12. Baerheim A, Digranes A, Jureen R, Malterud K. Generalized
symptoms in adult women with acute uncomplicated lower urinary tract
infection: an observational study. MedGenMed. 2003;5:1.
13. Giesen LG, Cousins G, Dimitrov BD, et al. Predicting acute
uncomplicated urinary tract infection in women: a systematic review of
the diagnostic accuracy of symptoms and signs. BMC Fam Pract. 2010;11:78.
14. Parsons CL, Dell J, Stanford EJ, et al. The prevalence of
interstitial cystitis in gynecologic patients with pelvic pain, as
detected by intravesical potassium sensitivity. Am J Obstet Gynecol. 2002;187:1395-1400.
15. Hanno PM, Burks DA, Clemens JQ, et al. American Urological
Association (AUA) Guideline. Diagnosis and treatment of interstitial
cystitis/bladder pain syndrome.
www.auanet.org/content/guidelines-and-quality-care/clinical-guidelines/main-reports/ic-bps/diagnosis_and_treatment_ic-bps.pdf.
Accessed August 17, 2012.
16. O’Leary MP, Sant GR, Fowler FJ Jr, et al. The interstitial cystitis symptom index and problem index. Urology. 1997;49(suppl 5A):58-63.
17. Kushner L, Moldwin RM. Efficiency of questionnaires used to screen for interstitial cystitis. J Urol. 2006;176(2):587-592.
18. Parsons CL, Dell J, Stanford EJ, et al. Increased prevalence of
interstitial cystitis: previously unrecognized urologic and gynecologic
cases identified using a new symptom questionnaire and intravesical
potassium sensitivity. Urology. 2002;60:573-578.
19. Teichman JM. Potassium sensitivity testing in the diagnosis and treatment of interstitial cystitis. Infect Urol. 2003;16:87-94.
20. Foster HE Jr, Hanno PM, Nickel JC, et al. Effect of amitriptyline
on symptoms in treatment naïve patients with interstitial
cystitis/painful bladder syndrome. J Urol. 2010;183:1853-1858.
21. Dell JR, Parsons CL. Multimodal therapy for interstitial cystitis. J Reprod Med. 2004;49(suppl 3):243-252.
22. Stein J. Sacral neuromodulation effective for pain of interstitial cystitis. Medscape Medical News. www.medscape.com/viewarticle/720824. Accessed August 17, 2012.
23. Rittenmeyer H. Sacral nerve neuromodulation (InterStim). Part I: review of the InterStim system. Urol Nurs. 2008;28:15-20.
24. Linn JF, Hohenfellner M, Roth S, et al. Treatment of interstitial
cystitis: comparison of subtrigonal and supratrigonal cystectomy
combined with orthotopic bladder substitution. J Urol. 1998;159:774-778.
25. Hewitt A. Urinary diversion—formation of a urostomy, Indiana
pouch and ileal neobladder.
http://urobop.co.nz/urology/wp-content/uploads/2012/03/8838_UrinaryDiversion.pdf.
Accessed August 17, 2012.
26. Kochakarn W, Lertsithichai P, Pummangura W. Bladder substitution by ileal neobladder for women with interstitial cystitis. Int Braz J Urol. 2007;33:486-492.
To comment on this article, contact rdavidson@uspharmacist.com.





