US Pharm. 2013;38(11):23-26.
ABSTRACT: When used as
monotherapy, warfarin is associated with an increased risk of bleeding.
When warfarin is used concomitantly with other drugs that may also
increase the risk of bleeding or affect the concentration of warfarin,
the bleeding risk may be further increased. The combined use of warfarin
and antidepressants, particularly the selective serotonin reuptake
inhibitors, may lead to drug-drug interactions via pharmacodynamic and
pharmacokinetic mechanisms. Limited data exist concerning the potential
for drug-drug interactions between warfarin and antidepressants. When it
is necessary to use both warfarin and an antidepressant, it is
recommended to select an agent with minimal effect on the CYP450 system
and a lower degree of inhibition of serotonin reuptake, as well as to
increase the frequency of monitoring of the international normalized
ratio and signs and symptoms of bleeding.
Warfarin is a vitamin K antagonist, more commonly referred to as an oral coumarin anticoagulant. It is indicated for the treatment and prophylaxis of venous thrombosis, pulmonary embolism, thromboembolic complications associated with atrial fibrillation and/or cardiac valve replacement, and reduction in the risk of death, recurrent myocardial infarction, and thromboembolic events.1 Although warfarin’s anticoagulant effect proves to be beneficial when utilized for the above indications, an expected adverse consequence of warfarin use is an increased risk of bleeding. In fact, bleeding complications, such as gastrointestinal (GI) tract hemorrhage, epistaxis, and rectal hemorrhage, are listed among the top-ranked adverse events for warfarin in the FDA Adverse Event Reporting System (FAERS).2
Warfarin is a narrow therapeutic index (NTI) agent and therefore must be maintained within a narrow therapeutic range in order to achieve maximal efficacy with minimal risk of adverse events. Consequently, when administered with other medications, drug interactions associated with even small changes in warfarin concentration and/or effect have the potential to increase the risk of warfarin’s adverse events, most notably an increased risk of bleeding and bleeding complications.
Antidepressants are categorized into five different groups: selective serotonin reuptake inhibitors (SSRIs), serotonin-norepinephrine reuptake inhibitors (SNRIs), tricyclic antidepressants (TCAs), monoamine oxidase inhibitors (MAOIs), and “other” or atypical antidepressants. The SSRIs continue to be used as first-line therapy for FDA-approved indications such as major depressive disorder and generalized anxiety disorder because of their favorable side-effect profile and safety. It has been suggested that antidepressants, particularly SSRIs and SNRIs, may themselves lead to an increased risk of bleeding through several different mechanisms, such as impairment of platelet aggregation, depletion of platelet serotonin levels, and reduction in platelet count.3 The bleeding risk associated with antidepressants is not dose-dependent for all classes, but the TCAs, in particular, may be associated with dose-dependent increases in bleeding risk.
This increased risk of bleeding associated with both warfarin and certain antidepressants may lead to an additive effect when these therapies are used in combination. Furthermore, warfarin use in patients on antidepressant therapy can also lead to drug interactions via pharmacodynamic and pharmacokinetic mechanisms. Due to the possible increased risk of bleeding associated with warfarin monotherapy and the concomitant use of warfarin and antidepressants, this review focuses on the potential drug interactions between warfarin and antidepressants so that healthcare professionals may be better equipped when caring for patients requiring both of these commonly prescribed therapies.
Mechanism of Drug Interactions With Warfarin
Interactions between warfarin and antidepressant therapies most often occur through two distinct mechanisms, namely alteration in the CYP450 system responsible for warfarin metabolism and/or alteration in warfarin protein binding. Warfarin consists of a racemic mixture of S-warfarin and R-warfarin, both of which inhibit the synthesis of vitamin K–dependent clotting factors leading to an anticoagulant effect. Both the S and R isomers differ in their elimination half-life, metabolism, and potency. The S isomer is three to five times more potent as compared to the R isomer and is therefore most associated with drug interactions linked to warfarin.4 The more potent S isomer is metabolized by CYP2C9, while the R isomer is primarily metabolized by CYP1A2 and to a lesser extent by CYP3A4 and CYP2C19.5
Warfarin is also highly protein-bound; therefore when used concomitantly with other medications that are highly protein-bound, it has the potential to be displaced from serum protein, leading to increased free warfarin concentration and an increased risk of bleeding. Although warfarin is approximately 99% protein-bound, there is little clinical evidence that this pharmacodynamic interaction produces a meaningful drug-drug interaction, even among antidepressants that are noted to be highly protein-bound (i.e., sertraline, duloxetine).4,6
Each of the antidepressants has varying effects on the CYP450 system as it relates to warfarin (TABLE 1).4,6-8
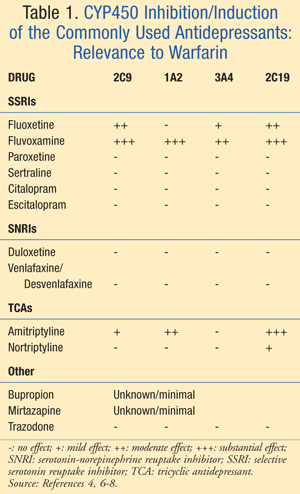
SSRIs: Among the SSRIs, fluoxetine and fluvoxamine are thought to have the highest potential for inhibiting warfarin metabolism and therefore enhancing warfarin’s anticoagulant effect. Fluoxetine is a mild inhibitor of 3A4 and a moderate inhibitor of 2C9, while fluvoxamine is the only SSRI that is a potent inhibitor of 1A2 and is also an inhibitor of 2C19, 3A4, and 2C9.9 Although paroxetine does not affect the CYP450 enzymes relevant to warfarin, it has been shown to increase bleeding time when coadministered with warfarin; however, its mechanism is undetermined.4,6,9 Sertraline, citalopram, and escitalopram, the S isomer of citalopram, do not affect the CYP450 enzymes relevant to warfarin and appear to have a low potential for interactions with warfarin when compared to the other SSRIs.8
Vortioxetine is the newest FDA-approved SSRI. Like the other SSRIs, it may potentially increase the risk of bleeding when used as monotherapy. This risk is noted to be increased when used in combination with other medications that may increase the risk of bleeding themselves, such as nonsteroidal anti-inflammatory drugs (NSAIDs), warfarin, and other anticoagulants. Vortioxetine is primarily metabolized by CYP2D6 and has not been shown to affect the INR of patients using warfarin concomitantly.10
SNRIs: Duloxetine, an SNRI, does not appear to affect the CYP450 enzymes relevant to warfarin, although there are case reports of duloxetine increasing and decreasing the effects of warfarin.4 Venlafaxine, another SNRI, and its active metabolite desvenlafaxine, do not appear to have any relevant CYP450 interactions as they relate to warfarin. Therefore, the SNRIs are considered to have a low risk for drug interactions when coadmin-istered with warfarin.4
Levomilnacipran, a newer SNRI, does not appear to affect pharmacokinetic properties associated with warfarin. Like other SNRIs, the package insert for levomilnacipran contains a warning for increased risk of bleeding when used concomitantly with other drugs that interfere with hemostasis (i.e., NSAIDs, warfarin, aspirin).11
TCAs: Cardiotoxic effects and lethality in overdose limit the use of TCAs. In addition to their FDA-approved indications in clinical practice, they are commonly prescribed for off-label indications (e.g., headache, insomnia). The TCAs have the potential to increase the effects of warfarin but usually not at the normally prescribed dose.6 The TCAs, particularly nortriptyline and amitriptyline, have been shown to increase prothrombin time (PT) in a dose-dependent manner. The effect of the TCAs (nortriptyline and amitriptyline) on the metabolism of warfarin was greatest with nortriptyline. When used as monotherapy, the TCAs do not appear to increase the risk of bleeding; however, when they are used with warfarin there may be an increased risk of bleeding secondary to their effect on the metabolism of warfarin.3,4
Other Antidepressants: Other commonly used antidepressants include bupropion, trazodone, and mirtazapine. They may be referred to as atypical antidepressants because their mechanism of action does not fit into a well-defined antidepressant class. Mirtazapine is metabolized by CYP2D6, 1A2, and 3A4, but it is thought to have minimal inhibitory effects on the CYP450 enzymes. Norton and Quarles describe a case in which a patient was stabilized on warfarin 10 mg (international normalized ratio [INR] varying between 2 and 3) for 7 months, and sertraline 200 mg and topiramate 100 mg twice daily for 4 months.12 After the addition of mirtazapine 15 mg for increased depressive symptoms, a repeat INR 2 weeks later was 5.9. Mirtazapine is considered to have a low risk of drug-drug interactions when used with warfarin; however, caution and monitoring should still occur with their concomitant use.4
Trazodone’s interaction potential with warfarin is considered low, although it has been shown to decrease PT and partial thromboplastin time (PTT). The mechanism for this property of trazodone is unknown and is not considered significant.6 Bupropion’s inhibitory effect on enzymes relevant to warfarin is considered minimal or unknown.13,14
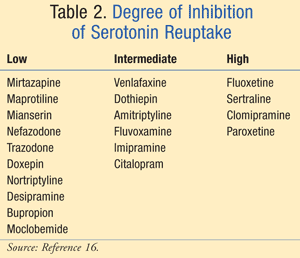
The antidepressants, particularly those affecting serotonin, may contribute to increased bleeding risk because of their effects on platelet aggregation. The mechanism of action of the SSRIs as a class is reduced uptake of serotonin into platelets. Serotonin promotes platelet aggregation. By inhibiting the entry of serotonin into platelet cells, the risk of abnormal bleeding is increased due to low concentrations of serotonin.15
Studies reporting adverse effects of bleeding with
antidepressant use have been mostly observational in design. Meijer et
al conducted a case-control study of first-time antidepressant users
investigating the relationship between serotonin reuptake inhibition and
bleeding risks.16 A greater degree of serotonin reuptake
inhibition, which was categorized as high, intermediate, or low, was
associated with a higher risk of abnormal bleeding (TABLE 2).16
Monitoring Parameters Associated With Warfarin
The use of warfarin is noted for its NTI, which necessitates close monitoring of the patient’s INR. Patients on long-term anticoagulation maintained on warfarin with stable therapeutic INRs may be most affected by the addition of newer agents such as an SSRI or other antidepressants.
The updated CHEST guidelines from the American College of Chest Physicians provide new recommendations on the frequency of INR monitoring.17 If patients consistently maintain a stable, therapeutic INR, the guidelines recommend checking the INR every 12 weeks instead of the previously recommended monthly testing. However, if the INR is not within a stable therapeutic range, it is still recommended to monitor the INR every 4 weeks until stable. Patients who present with a single INR out of range of ≤0.5, who normally present with stable INR values, are no longer recommended to receive a warfarin dosage change. Instead, the guidelines now recommend continuing the current dose and rechecking the INR within 1 to 2 weeks. Additionally, bridging with enoxaparin is no longer recommended for single out-of-range INR values of ≤0.5.17
In addition to monitoring of the INR, patients should be continuously questioned at each visit about dietary choices, medication adherence, and use of prescription and OTC drugs. Pharmacists should continue to update patient medication profiles to include prescription and OTC drugs.17
Because of warfarin’s NTI, INR monitoring, adverse effects, and numerous drug interactions, several new agents have been developed as therapeutic options in anticoagulant management. Oral anticoagulants such as dabigatran, rivaroxaban, and apixaban are associated with bleeding as a major adverse effect but do not require INR monitoring.13,14 As newer agents, further studies are warranted to evaluate drug-drug interactions and risks of bleeding and hospitalization with concomitant antidepressant use.
Conclusion
Limited data exist concerning the potential for drug-drug interactions between warfarin and antidepressants. In patients on warfarin therapy who also require antidepressant therapy, it may be prudent to initiate anti-depressant therapy with an agent with minimal to no effect on CYP2C9, 2C19, 3A4, and 1A2 enzymes. It also may be prudent to initiate antidepressant therapy with an agent with a lower degree of inhibition of serotonin reuptake, although the clinical relevance of this interaction is likely minimal.
In addition, more frequent monitoring of INRs and close monitoring of signs and symptoms of bleeding are recommended upon initiation, dosage alterations, and discontinuation of antidepressant therapy in patients also on warfarin. In patients already receiving antidepressant therapy who are being initiated on warfarin therapy, the potential drug-drug interactions would likely be accounted for during initial warfarin dose titration and INR monitoring. Pharmacists can play a large role in preventing adverse events due to warfarin drug interactions, including those associated with antidepressant therapy.
REFERENCES
1. Coumadin (warfarin) package insert. Princeton, NJ: Bristol-Meyers Squibb Co; 2011.
2. Wysowski D, Nourjah P, Swartz L. Bleeding complications
with warfarin use: a prevalent adverse effect resulting in regulatory
action. Arch Intern Med. 2007;167:1414-1419.
3. Cochran K, Cavallari L, Shapiro N, Bishop J. Bleeding incidence with concomitant use of antidepressants and warfarin. Ther Drug Monit. 2011;33:433-438.
4. Sansone R, Sansone L. Warfarin and antidepressants: happiness without hemorrhaging. Psychiatry (Edgemont). 2009;6:24-29.
5. Hauta-Aho M, Tirkkonen T, Vahlberg T, et al. The effect
of drug interactions on bleeding risk associated with warfarin therapy
in hospitalized patients. Ann Med. 2009;41:619-628.
6. Bachawati M. Anticoagulation and psychotropic medications. Curr Psychiatry Rep. 2010;12:265-271.
7. Preskorn S, Borges-Gonzalez, S, Flockhart D. Clinically
relevant pharmacology of neuropsychiatric drugs approved over the last
three years: part II. J Psych Pract. 2006;12:312-316.
8. Sayal KS, Duncan-McConnell DA, McConnell HW, Taylor DM. Psychotropic interactions with warfarin. Acta Psychiatr Scand. 2000;102;250-255.
9. Hiemke C, Hartter S. Pharmacokinetics of selective serotonin reuptake inhibitors. Pharmacol Ther. 2000;85:11-28.
10. Brintellix (vortioxetine) package insert. Deerfield, IL: Takeda Pharmaceuticals America, Inc; September 2013.
11. Fetzima (levomilnacipran) package insert. St. Louis, MO: Forest Laboratories, Inc; July 2013.
12. Norton J, Quarles E. Mirtazapine-induced warfarin toxicity. Prim Psychiatry. 2002;9:30-31.
13. Lexi-Comp Online. Hudson, OH: Lexi-Comp, Inc; 2013. http://online.lexi.com. Accessed October 25, 2013.
14. Micromedex Healthcare Series. Micromedex 2.0. Greenwood Village, CO: Truven Health Analytics; 2013. www.micromedex.com. Accessed October 25, 2013.
15. Hergovich N, Aigner M, Hans-Georg E. Paroxetine decreases platelet serotonin storage and platelet function in human beings. Clin Pharmacol Ther. 2000;68:435-442.
16. Meijer W, Heerdink E, Nolen W, et al. Association of
risk of abnormal bleeding with degree of serotonin reuptake inhibition
by antidepressants. Arch Intern Med. 2004;164:2367-2370.
17. Guyatt GH, Akl EA, Crowther M, et al; American College
of Chest Physicians (ACCP) Antithrombotic Therapy and Prevention of
Thrombosis Panel. Executive summary: Antithrombotic Therapy and
Prevention of Thrombosis, 9th ed: ACCP Evidence-Based Clinical Practice
Guidelines. Chest. 2012;141(suppl):7S-47S.
To comment on this article, contact rdavidson@uspharmacist.com.





