US Pharm. 2014;39(4):23-26.
ABSTRACT: Pneumococcal disease caused by Streptococcus pneumoniae
is a common medical concern in young children, older adults, and the
immunocompromised. Two vaccines, PPSV23 (Pneumovax 23) and PCV13
(Prevnar 13), are available for the prevention of conditions caused by
this bacterium and are recommended for routine use and for use in those
with high-risk comorbidities. While these vaccines do not cover all
serotypes, they have produced a significant reduction in both invasive
and noninvasive pneumococcal diseases.
Streptococcus pneumoniae (pneumococcus) is a gram-positive bacterium with more than 90 known serotypes. S pneumoniae, which is spread by airborne droplets, colonizes the nasopharynx in as many as 5% to 70% of healthy adults and 20% to 60% of healthy children.1 S pneumoniae causes a myriad of infections, categorized as either invasive (e.g., meningitis, bacteremia, pneumonia) or noninvasive (e.g., bronchitis, sinusitis, otitis media), and is a leading cause of serious illness among children and adults.2 Invasive disease is seen in both healthy and immunocompromised hosts, but it is most common in adults ≥65 years of age and children <2 years old and in certain immunocompromising conditions, such as HIV infection.2
S pneumoniae is the most common bacterial pathogen in hematologic malignancies, neutropenia, and prior respiratory viral infection, especially influenza. In the United States, pneumococcal disease is also more common in certain ethnic groups such as African Americans and Navajo Indians. This prevalence is thought to reflect genetic and environmental factors.3
Prior to 1990, S pneumoniae was almost uniformly susceptible to penicillin. However, during the 1990s, resistance to penicillin and other classes of antimicrobial agents spread throughout the U.S. Following the introduction of the 7-valent pneumococcal conjugate vaccine (PCV7) into the routine childhood immunization program in 2000, the incidence of invasive pneumococcal disease declined substantially among both young children and older persons.4
Reducing the burden of pneumococcal infections has been a significant public health priority. The Healthy People 2010 initiative aimed to reduce invasive pneumococcal disease (IPD) cases to 46 per 100,000 in children and 42 per 100,000 in adults.4 These goals were achieved with the introduction of PCV7, with 22 new cases and 41 new cases, respectively. Healthy People 2020 seeks to further reduce IPD rates to 12 and 31 new cases per 100,000 in children and adults, respectively.4 The use of vaccination strategies to decrease the burden of pneumococcal disease will be reviewed here.
Vaccine Development
In the early 1890s, Felix and Georg Klemperer showed that immunization with killed pneumococci protected animals against subsequent pneumococcal infection.3 In the early 1900s, studies of humoral immunity began in humans, showing a significant reduction in incidence of pneumonia. Lloyd D. Felton prepared the first purified pneumococcal capsular polysaccharides for immunization of human subjects, and a preparation of type 1 polysaccharide was used in an epidemic of pneumonia at a hospital in the winter of 1937-1938. These studies and others showed that a specific bacterial polysaccharide antigen could be used to stimulate humoral antibodies and confer protection against infection. During World War II, Colin M. MacLeod and colleagues found that vaccinating military recruits with material from four serotypes of S pneumoniae greatly reduced the incidence of pneumonia caused by serotypes in the vaccine, but not by other pneumococcal serotypes.3
Pneumococcal Vaccines
There are two commercially available vaccines for the prevention of pneumococcal disease. The pneumococcal polysaccharide vaccine is Pneumovax 23 (PPSV23),5 and Prevnar 13 (PCV13) is the conjugate version.6
Pneumococcal Polysaccharide Vaccine (PPSV): The pneumococcal vaccine was the first immunization developed from a bacterial capsular polysaccharide.1 While the introduction of the vaccine was a monumental medical milestone, it coincided with and was overshadowed by the discovery of penicillin, and was not widely utilized because of the efficacy of antibiotics in treating pneumococcal disease.1 Increasing resistance has largely shifted public health efforts to prevention rather than treatment, making immunization with PPSV part of the routine immunization schedule for those at high risk.
Upon immunization, type-specific antibodies are produced, which activate the immune system response. Only a few pneumococcal serotypes are responsible for the majority of IPD; however, the current PPSV provides expanded coverage to target 23 subgroups of the most prevalent serotypes responsible for invasive disease in the U.S.7 Although IPD is most common in children <2 years of age, polysaccharide vaccines are not recommended in this population because of their inability to adequately elicit a T cell–dependent immune response in young children.3
Pneumovax 23 is the only 23-valent pneumococcal polysaccharide vaccine available.5 The 23-valent vaccine replaced the 14-valent vaccine in 1983. PPSV23 is administered as an intramuscular (IM) or subcutaneous (SC) injection of one 0.5-mL dose.5 PPSV23 can be administered concurrently with other immunizations at separate sites of injection, although children completing PCV and PPSV in series may have a decreased immune response to further PCV immuni-zations.8 Additionally, a decreased immune response to the zoster vaccine may be seen when PPSV23 is administered concurrently. Although concurrent administration is not contraindicated and is recommended when accessibility is a concern, consideration could be given to separate administration of PPSV23 and zoster at least 4 weeks apart.5
PPSV23 is indicated in children at high risk of IPD, including immunocompromised children, those with functional or anatomical asplenia, and immunocompetent children with chronic heart or lung disease, diabetes mellitus, or cerebrospinal fluid leaks (TABLE 1).9,10 Children undergoing cochlear implants should also receive PPSV23. In children over the age of 2 years, the first dose of PPSV23 should follow the last indicated dose of PCV13 by 8 weeks. A booster dose of PPSV23 is indicated 5 years after the first dose in children who are immunocompromised, have sickle cell disease, or who have asplenia. A second dose is not recommended for immunocompetent children.9
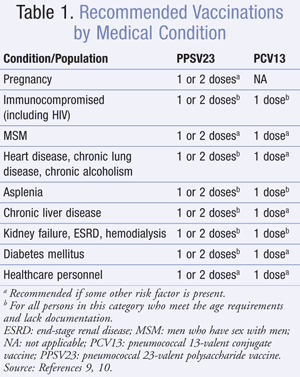
PPSV23 is recommended for all patients ≥65 years of age, regardless of immune status (TABLE 2).10 For those who received their first dose of PPSV23 prior to age 65, a second dose of PPSV23 is recommended 5 years after the first dose.10
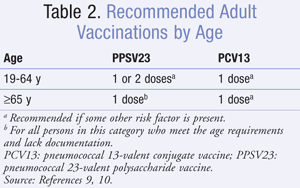
Persons aged 19 to 64 years who are at increased risk of morbidity and mortality associated with pneumococcal disease should also receive PPSV23. Cigarette smoking; chronic heart (excluding hypertension), lung, or liver disease; diabetes mellitus; alcoholism; cerebrospinal fluid leaks; and cochlear implants are all considered risk factors in immunocompetent adults.10
Patients who have functional or anatomical asplenia or who are immunocompromised should also be vaccinated, particularly those with hematologic malignancies, HIV infection, or solid tumors. The Advisory Council on Immunization Practices (ACIP) recommends vaccination with PCV13 in addition to PPSV23 in this population.10 When indicated, individuals not previously receiving PCV13 or PPSV23 should receive a single dose of PCV13 followed by a dose of PPSV23 at least 8 weeks later. If one or more doses of PPSV23 have been received, a single dose of PCV13 should be given 1 or more years after the last PPSV23 dose. In those who require additional doses of PPSV23, vaccination should occur at least 8 weeks after the last dose of PCV13, and at least 5 years after the last dose of PPSV23.10
Patients who experience a severe allergic reaction to any component of the PPSV23 formulation should not receive the vaccine. Injection site reactions are frequent and include pain, soreness, tenderness, swelling, and erythema.5 Adverse events outside of injection site reactions such as headache, fatigue, and myalgia have been reported. Although they are much less common than injection site reactions, these adverse effects tend to be more frequent in those who undergo revaccination.6 For any vaccination, suspected adverse events should be reported to the Vaccine Adverse Event Reporting System (VAERS).5
Overall, more than 80% of adults who receive PPSV23 develop antibodies, usually within 2 to 3 weeks.1 Older adults and those with chronic conditions may not respond as well, if at all. In studies of clinical effectiveness, PPSV23 is about 60% to 70% effective in preventing invasive disease; however, it does not provide reliable protection against pneumococcal pneumonia.1 Referring to PPSV23 as the “pneumonia vaccine” is therefore a misnomer and should be avoided.
Pneumococcal Conjugate Vaccine (PCV): PCV13 was released in 2010 and replaced the former 7-valent version (PCV7), which had been released in 2000.1 These 13 serotypes are most commonly implicated in disease of children. PCV was developed after discovering that children <2 years of age do not respond well to polysaccharide antigens. However, when those polysaccharides are conjugated to carrier proteins, they stimulate good antibody responses in young children.3
In late 2011, PCV13 was approved for the prevention of pneumonia and invasive disease in people aged ≥50 years; however, the effectiveness of the vaccine in this age group has not been demonstrated in controlled clinical studies and therefore it is not recommended in the routine vaccination schedule for adults.1 Children vaccinated with PCV13 were shown to have a reduction in IPD caused by vaccine serotypes of 97% and a reduction of all serotypes by 89%. However, the vaccine is less effective against pneumonia and acute otitis media.1
The ACIP recommends that PCV13 be used for all children <5 years old.1 PCV13 is given IM as an 0.5-mL dose. In order to improve vaccine series completion, it may be administered on the same day as other vaccinations at separate injection sites.6
For routine immunization of infants, PCV13 is recommended as a four-dose series at ages 2, 4, 6, and 12 to 15 months (TABLE 3).9 Infants and children who have received at least one dose of PCV7 should complete the immunization series with PCV13. A single supplemental dose of PCV13 is recommended for all children 14 to 59 months who have received four doses of PCV7 or another age-appropriate complete PCV7 schedule.9
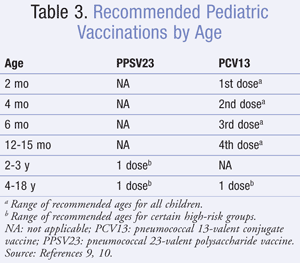
For children who have underlying immunocompromising conditions or anatomical or functional asplenia, or for immunocompetent children with chronic heart or lung disease, a supplemental PCV13 dose is recommended through age 71 months. 8 Children aged 2 to 18 years with underlying medical conditions should also receive PPSV23 after completing all recommended doses of PCV13. Additionally, a single dose of PCV13 may be administered to children aged 6 to 18 years who are at increased risk of IPD due to sickle cell disease, HIV, or other immunocompromising conditions (TABLE 3).9 Routine use of PCV13 is not recommended for healthy children >5 years old.9
In children vaccinated at the recommended intervals, the most common adverse reactions were irritability; injection site tenderness, redness, or swelling; decreased appetite; decreased sleep; and fever. It is recommended that patients be observed for at least 15 minutes after immunization to watch for adverse reactions.6
Pharmacist’s Role
The burden of pneumococcal disease has been significantly reduced through immunization strategies targeting both children and adults. Pharmacists should help play a role in identifying patients who are candidates for vaccination, particularly adults who may have indications because of chronic health conditions or who are immunocompromised. Where licensed, pharmacist immunizers are also in a prime position to improve immunization rates for populations that could benefit from protection from pneumococcal disease.
REFERENCES
1. CDC. Pneumococcal disease. In: Epidemiology and Prevention of Vaccine-Preventable Diseases.
12th ed. Washington, DC: Public Health Foundation; 2012.
http://www.cdc.gov/vaccines/pubs/pinkbook/pneumo.html Accessed January
6, 2014.
2. World Health Organization (WHO). Introduction of Pneumococcal Vaccine PCV13, A Handbook for District and Health Facility Staff.
Geneva, Switzerland: WHO; October 2013.
http://apps.who.int/iris/bitstream/10665/90380/1/WHO_IVB_13.10_eng.pdf.
Accessed January 14, 2014.
3. Musher D. Streptococcus pneumonia. In: Mandell GL, Bennett JE, Dolin R, eds. Principles and Practice of Infectious Disease. 7th ed. Philadelphia, PA: Elsevier;2010:2623-2642.
4. Centers for Disease Control and Prevention (CDC). Pneumococcal disease. In: Manual for the Surveillance of Vaccine-Preventable Diseases. 5th ed. Atlanta, GA: CDC; 2012. www.cdc.gov/vaccines/pubs/surv-manual/chpt11-pneumo.html. Accessed January 10, 2014.
5. Pneumovax 23 (pneumococcal vaccine polyvalent) package
insert. Whitehouse Station, NJ: Merck & Co., Inc; March 2013.
www.merck.com/product/usa/pi_circulars/p/pneumovax_23/pneumovax_pi.pdf.
Accessed January 6, 2014.
6. Prevnar 13 (pneumococcal 13-valent conjugate vaccine) package
insert. Philadelphia, PA: Wyeth Pharmaceuticals (Pfizer); January 2014.
http://labeling.pfizer.com/showlabeling.aspx?id=501. Accessed January
6, 2014.
7. Nuorti JP, Whitney CG; CDC. Prevention of pneumococcal
disease among infants and children—use of 13-valent pneumococcal
conjugate vaccine and 23-valent pneumococcal polysaccharide
vaccine—recommendations of the Advisory Committee on Immunization
Practices (ACIP). MMWR Recomm Rep. 2010;59:1.
8. Sigurdardottir ST, Center KJ, Davidsdottir K, et al.
Decreased immune response to pneumococcal conjugate vaccine after
23-valent pneumococcal polysaccharide vaccine in children. Vaccine. 2014;32:417-424.
9. CDC. Recommended immunization schedule for persons age 0
through 18 years.
www.cdc.gov/vaccines/schedules/hcp/imz/child-adolescent.html. Accessed
January 6, 2014.
10. CDC. Recommended adult immunization schedule, by
vaccine and age groups.
www.cdc.gov/vaccines/schedules/hcp/imz/adult.html. Accessed January 6,
2014.
To comment on this article, contact rdavidson@uspharmacist.com.





