US Pharm. 2009;34(12):Epub.
Irritable bowel syndrome (IBS) is a common condition that affects patients physically, psychologically, socially, and economically. Based on strict criteria, it is estimated that 7% to 10% of people worldwide have IBS.1 The prevalence of IBS in North America is estimated to be between 3% and 20%, with most studies indicating a range of 10% to 15%.2 IBS appears to peak in the third and fourth decades of life and decline in the sixth and seventh decades.3 Women are approximately 1.5 times more likely than men to have IBS.4-7 Race does not seem to be a factor in the prevalence of the disorder.8Patients with IBS consume a significant amount of health care resources. In 2001, it was estimated that those with IBS made 3.6 million physician visits annually and incurred up to $20 billion in both direct and indirect health care costs.9 Moreover, patients with IBS consume 50% more health care resources that those without it.10,11 Clearly, IBS places a significant burden on society; it also presents an excellent opportunity for pharmacists to participate in the care of patients with the disorder. This article reviews the pathophysiology, diagnosis, signs and symptoms, and treatment of IBS and discusses how the pharmacist can assist patients seeking care for this condition.
Pathophysiology
Although the pathophysiology of IBS remains unclear, theories abound regarding possible etiologies. Genetic predisposition, disturbances of gastrointestinal (GI) motility, enhanced gut pain sensation (visceral hypersensitivity), mucosal inflammatory processes, and emotional or psychological disturbances have all been described as possible mechanisms of the disease.3,12 Owing to the variety of complaints seen in patients with IBS, the actual pathophysiology is likely a combination of these mechanisms, as well as some processes not currently recognized.3
The digestive nervous system contains a sizable percentage of the body's serotonin (5-HT).13 Two types of 5-HT are present in the gut: 5-HT3 and 5-HT4.14 These monoamine neurotransmitters are responsible for secretion, sensitization, and motility in the digestive tract.14 Studies have indicated that there is a postprandial rise of 5-HT levels in people with diarrhea-predominant IBS that does not occur in nonsufferers.13 As a result, stimulation or antagonism of these receptors has been a focus of research on IBS.
Diagnosis
IBS is defined by abdominal pain or discomfort that occurs in association with altered bowel habits over a period of at least 3 months.1 It is noteworthy that IBS is distinguished by the absence of biochemical markers or structural abnormalities that are often present in other diseases. Individual symptoms are of limited utility in diagnosing IBS; therefore, the illness should be considered as a syndrome complex.1 Diagnostic criteria have been developed that identify a combination of symptoms. These include the Kruis, Manning, and Rome I criteria (TABLE 1).1,15-17 Newer versions of the Rome criteria have been developed, but they have not been evaluated.1
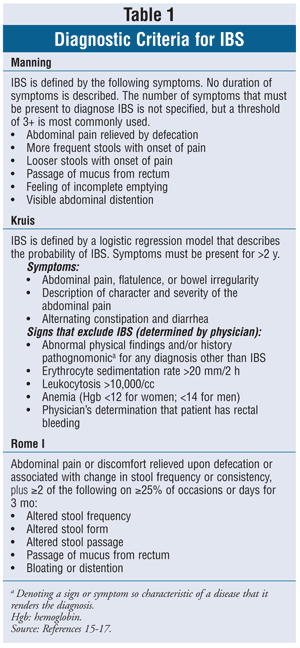
Signs and Symptoms
Most IBS patients complain of intermittent abdominal pain, bloating, abnormal stool frequency, and abnormal defecation (diarrhea or constipation).18 In general, IBS may be divided into two subcategories depending on stool consistency. One subcategory is IBS with constipation (IBS-C), in which patients have hard stools more than 25% of the time and loose stools less than 25% of the time; the other is IBS with diarrhea (IBS-D), in which patients have loose stools more than 25% of the time and hard stools less than 25% of the time.18
Non-GI symptoms such as lethargy, backache, headache, urinary symptoms, and dyspareunia also may occur in patients with IBS.19 Psychological distress is common; approximately 50% of IBS patients who seek medical care are depressed or anxious.19 Taken in total, health-related quality of life in IBS patients may be similar to that in patients with diabetes, hypertension, or kidney disease.1
Treatment
IBS is a chronic disease with no known cure. Treatment is often directed at specific symptoms. However, the main goal of treatment is to provide relief from symptoms and improve overall quality of life.3 This practice is often referred to as global symptom management. For the purposes of this article, a description of most therapies for IBS is provided, followed by their respective roles in IBS-C and IBS-D.
Diet: Many IBS patients (60%-70%) report that particular foods exacerbate their symptoms.1,20,21 As a result, most patients attempt to exclude these foods from their diet.22 Dietary intervention in the form of exclusion diets (foods that aggravate symptoms are omitted) has been explored as a means of controlling IBS symptoms. Unfortunately, evidence is inconclusive as to whether exclusion diets are efficacious in the treatment of IBS, and their routine use is not recommended.1Dietary Fiber, Bulking Agents, and Laxatives: A common recommendation made to IBS sufferers is to increase the amount of fiber in the diet. However, the quality of evidence supporting this recommendation is poor.1 The majority of studies have examined the effect of wheat bran or psyllium hydrophilic mucilloid (e.g., Metamucil) on IBS symptoms. While wheat bran provided no global benefit in patients with IBS, psyllium hydrophilic mucilloid was moderately effective.1 The use of corn fiber has not been found to confer a substantial benefit in IBS.22 A study comparing calcium polycarbophil (FiberCon) with placebo concluded that IBS patients with constipation or alternating diarrhea and constipation preferred calcium polycarbophil over placebo; the researchers also suggested that this product might be particularly useful in individuals who suffer from bloating.23 Laxatives have not been studied in adults with IBS, but they have been evaluated in chronic constipation.1 A study of polyethylene glycol (PEG) laxative in adolescents with IBS-C showed improved stool frequency, but no effect on pain intensity.1
Antispasmodics: Pain associated with IBS often is due to colonic muscle spasm.1 As a result, numerous agents have been developed to reduce this pain by acting directly on the smooth muscle or by affecting cholinergic receptors in the gut. Specific antispasmodic agents--hyoscine, cimetropium, pinaverium (cimetropium and pinaverium are not available in the U.S.), and peppermint oil--may provide short-term relief of abdominal pain or discomfort in IBS.1 Hyoscine appears to show the most evidence of providing relief; trimebutine, although studied in IBS, has no effect on symptoms versus placebo.1 The most common adverse events are dry mouth, dizziness, and blurred vision.1 Evidence of long-term efficacy is not available.Antidiarrheals: Patients with IBS-D often have a more rapid intestinal transit than healthy individuals. For this reason, it has been suggested that delaying intestinal transit may aid in reducing IBS symptoms. Loperamide is the only antidiarrheal agent sufficiently investigated for use in IBS that results in a reduction in stool frequency and improvement in stool consistency.1 Loperamide has not been shown to reduce pain, bloating, or global symptoms of IBS.1
Antibiotics: Nonabsorbable antibiotics may have a role in the treatment of IBS. Rifaximin (Xifaxan), a structural analog of rifampin indicated for traveler's diarrhea, has demonstrated activity in IBS.1,24 Overall, rifaximin demonstrates improvement in global IBS symptoms and bloating in patients who have predominantly IBS-D.1 The optimal dose is not known; however, based on available data, 400 mg po three times daily for a course of 10 to 14 days seems to be effective.1 No significant adverse effects were seen with higher doses. IBS relief appears to last 10 to 12 weeks, but symptoms may recur over 3 to 9 months.1 No data exist supporting the long-term safety and effectiveness of rifaximin for the management of IBS symptoms.1
Neomycin, metronidazole, and clarithromycin have been evaluated for the management of IBS. In a single trial, only neomycin demonstrated some efficacy in the treatment of IBS.1
Probiotics: Probiotics have been used empirically for many years in the treatment of IBS.1 In theory, probiotics may possess properties that may prove beneficial for patients with this disease. Unfortunately, evaluation of probiotic use for IBS is hampered by the wide variation in species, strains, preparation methods, and doses used.1 In single-organism studies, lactobacilli do not appear to confer a benefit in patients with IBS. Bifidobacteria appear to have some efficacy in the treatment of IBS, as do selected combinations of probiotics.1 Notably, when bifidobacteria and lactobacilli have been used in combination, lactobacilli did not hinder the reduction of IBS symptoms.1 It is possible, therefore, that bifidobacteria are the active agent in probiotic combinations, or that different species of probiotics are synergistic in promoting a therapeutic effect on IBS.1
5-HT3 Receptor Antagonists: As stated previously, expression of 5-HT3 receptors can affect normal gastric physiology, with an increased level of 5-HT3 seen in patients with IBS-D.13,14 The 5-HT3 receptor antagonist alosetron (Lotronex) has been shown to be effective at relieving global IBS symptoms in male and female patients with IBS-D.1 Potential serious side effects include constipation, obstructed bowel, and colonic ischemia.1 These side effects caused the manufacturer to voluntarily withdraw the drug from the U.S. market in November 2000, but since April 2002 alosetron has been remarketed under restricted conditions of use.25-29
Alosetron currently is to be used only for the management of severe IBS-D in women with chronic symptoms (generally lasting ≥6 months) in whom anatomical or biochemical GI abnormalities have been excluded and who fail to respond to conventional therapy.29,30 IBS-D is considered severe if the diarrhea is accompanied by one or more of the following: frequent, severe abdominal pain or discomfort; frequent bowel urgency or fecal incontinence; or disability or restriction of daily activities.29,30 Unfortunately, the drug's safety has not been evaluated in men.29
Alosetron should be discontinued if a patient develops symptoms or signs suggestive of severe constipation or ischemic colitis.1 The medication also should be stopped if the patient has no clinical response to the maximum dose of 1 mg twice daily after 4 weeks.1
Chloride Channel 2 Activators: Lubiprostone (Amitiza) is a bicyclic fatty acid that selectively activates intestinal type 2 chloride channels, with a net effect of increasing salt and water movement into the intestinal lumen.1,31 Lubiprostone seems to be effective for relieving global symptoms in women with IBS-C. Although lubiprostone is derived from prostaglandin, it does not work entirely through prostaglandin receptors.32,33 It is poorly absorbed into the systemic circulation and appears to work topically in the small intestine.1
Because lubiprostone is a pregnancy category C drug, women should have a negative pregnancy test before initiating this medication, and they should not become pregnant while taking the drug.31 Patients with a GI mechanical obstruction should not use lubiprostone; patients should be assessed for symptoms suggesting obstruction before starting the medication.34 The most common adverse reactions (incidence >4%) in IBS-C are nausea, diarrhea, and abdominal pain.34
5-HT4 Receptor Agonists: Tegaserod (Zelnorm) is the only 5-HT4 receptor agonist that has been brought to market. It was withdrawn in March 2007 owing to potential cardiovascular events (myocardial infarction, unstable angina, stroke).1 Tegaserod has demonstrated efficacy for global symptoms and individual IBS symptoms in women with IBS-C.1 The agent is available only through the FDA for emergency situations (i.e., a state that is immediately life-threatening or serious enough to warrant hospitalization).35 The FDA may deny authorization even in a life-threatening situation if available evidence fails to provide a reasonable basis for concluding that tegaserod may be effective for the intended use, or if exposure to the agent would pose an unreasonable or significant additional risk to the patient.35
Antidepressants: Tricyclic antidepressants (TCAs) and selective serotonin reuptake inhibitors (SSRIs) effectively relieve global IBS symptoms and reduce abdominal pain.1 Reasons suggested for TCAs' effectiveness include antihyperalgesia, improvement in sleep, and normalization of GI transit.36 In theory, TCAs should be more effective in patients with IBS-D and SSRIs should be more effective in those with IBS-C owing to these classes' differing effects on intestinal transit time, but this supposition has not been investigated.1
Psychological Therapies: Often, IBS patients exhibit concomitant psychiatric disorders such as anxiety, depression, or somatization (persistent, chronic complaints of diverse physical symptoms with no identifiable physical basis).1 Cognitive therapy, dynamic psychotherapy, and hypnotherapy are more effective than usual care for relieving global symptoms; however, relaxation therapy does not seem to provide any benefit.1
General Guidelines
Patients with IBS may use insoluble fiber products such as psyllium to help regulate bowel movements and reduce pain.1 Psychotherapy may help improve symptoms by reducing stress.1 Specific agents used to treat IBS are given in TABLE 2.1,31,36-40
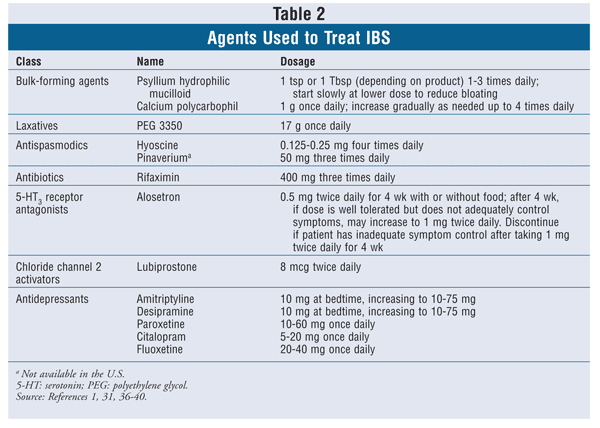
For patients with IBS-C, increasing dietary fiber and fluid intake may help regulate the bowels.1 Unfortunately, bloating can occur secondary to the use of bulking agents. Osmotic laxatives (PEG) may be tried when stool frequency needs to be increased.1 Lubiprostone can be used in women with IBS-C who have not responded to other therapies and who do not have contraindications to the medication. Pregnancy must be ruled out before lubiprostone is initiated, and the patient should avoid becoming pregnant while taking the drug. If pain is associated with IBS-C, an SSRI may improve global symptoms, reduce the pain, and improve the constipation.1
For patients with IBS-D, loperamide can reduce the frequency of bowel movements, but it will not improve abdominal cramping.1 If bloating accompanies the diarrhea, a short trial of rifaximin may be considered.1A TCA may help reduce the pain associated with IBS-D and improve the diarrhea.1 Alosetron may be considered, but only in women with severe, chronic IBS-D in whom conventional therapy has failed.1
When pain is the predominant symptom associated with IBS, antispasmodics or peppermint oil may be tried.1 TCAs and SSRIs are useful for reducing pain and improving overall symptoms.1
The Role of the Pharmacist
Pharmacists can play an integral role in assisting patients diagnosed with IBS. First, the pharmacist can establish an effective therapeutic relationship, educate the patient, and provide reassurance. The pharmacist can also help the patient identify the most predominant symptoms associated with the IBS and help develop a treatment plan to ameliorate these symptoms. Finally, the pharmacist can refer the patient to other health care providers, such as psychologists and dieticians, for advice and potential adjuvant therapies.
REFERENCES
1. Brandt LJ, Chey WD, Foxx-Orenstein AE, et al. An evidence-based systematic review on the management of irritable bowel syndrome. Am J Gastroenterol. 2009;104(suppl 1):S1-S35.
2. Saito YA, Schoenfeld P, Locke GR. The epidemiology of irritable bowel syndrome in North America: a systematic review. Am J Gastroenterol. 2002;97:1910-1915.
3. Bassett JT, Cash BD. A review of irritable bowel syndrome and an update on therapeutic approaches. Expert Opin Pharmacother. 2008;9:1129-1143.
4. Saito YA, Talley NJ, Melton LJ III, et al. The effect of new diagnostic criteria for irritable bowel syndrome on community prevalence estimates. Neurogastroenterol Motil. 2003;15:687-694.
5. Andrews EB, Eaton SC, Hollis KA, et al. Prevalence and demographics of irritable bowel syndrome: results from a large web-based survey. Aliment Pharmacol Ther. 2005;22:935-942.
6. Hungin AP, Chang L, Locke GR, et al. Irritable bowel syndrome in the United States: prevalence, symptom patterns and impact. Aliment Pharmacol Ther. 2005;21:1365-1375.
7. Minocha A, Johnson WD, Abell TL, Wigington WC. Prevalence, sociodemography, and quality of life of older versus younger patients with irritable bowel syndrome: a population-based study. Dig Dis Sci. 2006;51:446-453.
8. Podovei M, Kuo B. Irritable bowel syndrome: a practical review. South Med J. 2006;99:1235-1242.
9. American Gastroenterological Association (AGA). The Burden of Gastrointestinal Diseases. Bethesda, MD: AGA Press; 2001.
10. Talley NJ , Gabriel SE , Harmsen WS, et al. Medical costs in community subjects with irritable bowel syndrome. Gastroenterology. 1995;109:1736-1741.
11. Longstreth GF, Wilson A , Knight K, et al. Irritable bowel syndrome, healthcare use, and costs: a U.S. managed care perspective. Am J Gastroenterol. 2003;98:600-607.
12. Spiller R, Aziz Q, Creed F, et al. Guidelines on the irritable bowel syndrome: mechanisms and practical management. Gut. 2007;56:1770-1798.
13. Bearcroft CP, Perrett D, Farthing MJ. Postprandial plasma 5-hydroxytryptamine in diarrhea predominant irritable bowel syndrome: a pilot study. Gut. 1998;56:42-46.
14. Chey WD. Tegaserod and other serotonergic agents: what is the evidence? Rev Gastroenterol Disord. 2003;3(suppl 2):S35-S40.
15. Manning AP, Thompson WG, Heaton KW, Morris AF. Towards positive diagnosis of the irritable bowel. Br Med J. 1978;2:653-654.
16. Kruis W, Thieme C, Weinzierl M, et al. A diagnostic score for the irritable bowel syndrome. Its value in the exclusion of organic disease. Gastroenterology. 1984;87:1-7.
17. Thompson WG, Dotewall G, Drossman DA, et al. Irritable bowel syndrome: guidelines for the diagnosis. Gastroenterol Int. 1989;2:92-95.
18. Spiller R. Clinical update: irritable bowel syndrome. Lancet. 2007;369:1586-1588.
19. Whitehead WE, Palsson O, Jones KR. Systematic review of the comorbidity of irritable bowel syndrome with other disorders: what are the causes and implications? Gastroenterology.
20. Monsbakken KW, Vandvik PO, Farup PG. Perceived food intolerance in subjects with irritable bowel syndrome--etiology, prevalence and consequences. Eur J Clin Nutr. 2006;60:667-672.
21. Simrén M, Mansson A, Langkilde AM, et al. Food-related gastrointestinal symptoms in the irritable bowel syndrome. Digestion. 2001;63:108-115.
22. Cook IJ, Irvine EJ, Campbell D, et al. Effect of dietary fiber on symptoms and rectosigmoid motility in patients with irritable bowel syndrome. A controlled, crossover study. Gastroenterology. 1990;98:66-72.
23. Toskes PP, Connery KL, Ritchey TW. Calcium polycarbophil compared with placebo in irritable bowel syndrome. Aliment Pharmacol Ther. 1993;7:87-92.
24. Xifaxan (rifaximin) product information. Morrisville, NC: Salix Pharmaceuticals, Inc; March 2008.
25. Woodcock J. Letter regarding Lotronex. Rockville, MD: FDA; March 8, 2001.
26. Lotronex® tablets to be re-introduced for women with severe diarrhea-predominant IBS [press release]. Research Triangle Park, NC: GlaxoSmithKline; June 7, 2002.
27. Dear irritable bowel syndrome (IBS) patient: Lotronex information. Rockville, MD: FDA; January 24, 2002 [letter].
28. Young D. Lotronex returns to market. June 12, 2002. American Society of Health-System Pharmacists. www.ashp.org. Accessed November 11, 2009.
29. Alosetron (Lotronex) revisited. Med Lett Drugs Ther. 2002; 44:67-68.
30. Lotronex (alosetron) product information. Research Triangle Park, NC: GlaxoSmithKline; 2002.
31. Amitiza (lubiprostone) product information. Bethesda, MD: Sucampo Pharma Americas, Inc, and Deerfield, IL: Takeda Pharmaceuticals America, Inc; May 2009.
32. Bassil AK, Borman RA, Jarvie EM, et al. Activation of prostaglandin EP receptors by lubiprostone in rat and human stomach and colon. Br J Pharmacol. 2008;154:126-135.
33. Cuppoletti J, Malinowska DH, Chakrabarti J, Ueno R. Effects of lubiprostone on human uterine smooth muscle cells. Prostaglandins Other Lipid Mediat. 2008;86:56-60.
34. MedWatch. Detailed view: safety and labeling changes approved by FDA Center for Drug Evaluation and Research (CDER)--April 2008. www.fda.gov/Safety/MedWatch/ 2002;122:1140-1156.
35. FDA. Zelnorm (tegaserod maleate) information. www.fda.gov/Drugs/DrugSafety/
36. Mayer EA. Clinical practice. Irritable bowel syndrome. N Engl J Med. 2008;358:1692-1699.
37. Metamucil (psyllium hydrophilic mucilloid) product information. Cincinnati, OH: Procter & Gamble; 1999.
38. FiberCon (calcium polycarbophil) product information. Wayne, NJ: Lederle; 1990.
39. MiraLAX (polyethylene glycol 3350) product information. Kenilworth, NJ: Schering-Plough; 2007.
40. Lotronex (alosetron) product information. San Diego, CA: Prometheus Laboratories Inc; April 2008.
To comment on this article, contact rdavidson@jobson.com.





