US Pharm. 2012;37(7):27-30.
The annual cost of sinusitis in the United States is approximately $5.8 billion,1 which includes over 500,000 surgical procedures performed on the paranasal sinuses.2 Over 30 million Americans are affected by rhinosinusitis annually,3 with 73 million days of restricted activity or lost work per year.1 Bacteria cause sinusitis in only 2% to 10% of cases, yet a recent national survey showed that 81% of adults presenting with acute sinusitis were prescribed antibiotics.4-7 Acute sinusitis is now the fifth leading indication for antimicrobial prescriptions by primary care physicians.1 Primary care clinicians and pharmacists are challenged to differentiate between viral and bacterial sinusitis, which have nearly identical symptoms, and recommend appropriate therapy for patients. Due to these concerns, in March 2012 the Infectious Diseases Society of America (IDSA) published its first clinical practice guidelines on the treatment of acute bacterial sinusitis.8 A community pharmacist is poised to help treat this public health concern through education and proper recognition of when to treat symptoms or an underlying bacterial infection.
Definition and Etiology
Sinusitis, or rhinosinusitis, is defined as inflammation of the paranasal sinuses and nasal cavity.9 The term rhinosinusitis is preferred because sinusitis is typically associated with inflammation of the nasal mucosa. Rhinosinusitis can be categorized based on the duration of infection, and then further subdivided by the causative pathogen (FIGURE 1). Regarding duration of symptoms, rhinosinusitis can be classified as acute rhinosinusitis (<4 weeks), subacute rhinosinusitis (4-12 weeks), chronic rhinosinusitis (>12 weeks), or recurrent acute rhinosinusitis (four or more cases per year). When categorizing based on causative organism, the majority of cases are viral in origin, but a small minority are caused by bacterial and/or fungal pathogens. Acute rhinosinusitis is typically subdivided into acute bacterial rhinosinusitis (ABRS) or acute viral rhinosinusitis (AVRS).3
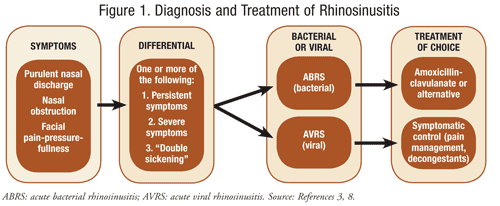
The most common etiology for rhinosinusitis is the rhinovirus, also known as the common cold. AVRS is the most common type of rhinosinusitis, accompanying upper respiratory tract infections in about 0.5% of patients when viral inoculation occurs via contact with nasal mucosa or conjunctiva.4 The virus causes inflammation in the nasal passages and sinus cavities, impairing mucociliary clearance and leading to the obstruction of the sinuses. ABRS most often occurs when the inflamed mucosa is secondarily infected by bacteria. Regardless of the causative organism, this mucosal inflammation causes the typical symptoms of acute rhinosinusitis.10
Symptoms
Rhinosinusitis has three cardinal symptoms: purulent nasal discharge, nasal obstruction, and facial pain-pressure-fullness. Purulent nasal discharge is cloudy or colored in appearance; nasal obstruction is defined by the patient as obstruction, congestion, blockage, or stuffiness; and facial pain-pressure-fullness involves the anterior face and periorbital region or manifests with headache that can be localized or diffuse.3 Secondary symptoms can include fever, cough, fatigue, hyposmia, anosmia, maxillary dental pain, and ear fullness or pressure.5,9 The 2012 IDSA guidelines help distinguish between viral and bacterial rhinosinusitis by defining three clinical presentations that may be present in bacterial but not viral infections: persistent and nonimproving symptoms, severe symptoms, or a worsening (i.e., “double sickening”).8
Some risk factors that predispose patients to rhinosinusitis include anatomical abnormalities, nasal allergic reactions, dental infections, mucosal abnormalities (i.e., cystic fibrosis), chemical irritants, and immunodeficiency.9 Some conditions that present similarly to rhinosinusitis warrant urgent referral due to possible complications, including intracranial and orbital infections. Symptoms that indicate urgent referral are diplopia, blindness, change in mental status, and periorbital edema.11
Categorization of Illnesses
Acute: As mentioned previously, acute rhinosinusitis can be divided into two categories depending on the causative organism—AVRS and ABRS. Symptoms of AVRS and ABRS can be identical, so distinguishing the two can be difficult. AVRS is normally self-limiting and typically resolves without treatment in 7 to 10 days. Bacterial complications occur in 0.5% to 2% of AVRS cases when the virally inflamed sinus cavity becomes secondarily infected.12 AVRS can promote bacterial infection by obstructing sinus drainage, promoting growth of bacteria, and depositing bacteria into the sinus cavity during nose blowing.12,13 Typical pathogens include Streptococcus pneumoniae, Haemophilus influenzae, Moraxella catarrhalis, and Staphylococcus aureus, with 74% percent of cases in adult patients caused by the first two bacteria.3 Resistance patterns have changed due to beta-lactamase–producing H influenzae and the increasing use of pneumococcal vaccines; for example, the amoxicillin resistance in H influenzae varies depending on geographical location. The southeastern U.S. has a 35% resistance while the southwestern U.S. has only a 25% resistance pattern.8
The 2012 IDSA guidelines help differentiate AVRS from ABRS by defining three clinical presentations that identify bacterial infections: symptoms for ≥10 days without improvement, severe symptoms, or worsening symptoms after initial improvement.8 Severe symptoms are defined as high fever (≥ 39°C [102°F]) and purulent nasal discharge or facial pain lasting for at least 3 to 4 consecutive days. Worsening symptoms, also called “double sickening,” are defined as the new onset of fever, headache, or nasal discharge following an upper respiratory tract infection that lasted 5 to 6 days, when initially symptoms were improving.8
Subacute: Subacute rhinosinusitis is a designation used to define the time period between acute rhinosinusitis (<4 weeks) and chronic rhinosinusitis (>12 weeks). No clinical data exist for the evaluation or treatment of subacute rhinosinusitis.3 As such, no specific recommendations can be made on the treatment of subacute rhinosinusitis.
Chronic: Chronic rhinosinusitis (CRS) is characterized by greater than 12 weeks of sinus inflammation regardless of appropriate medical management.14 The four cardinal signs of CRS include anterior and/or posterior mucopurulent drainage, nasal obstruction, facial pain, and decreased sense of smell. CRS may involve nasal polyps or allergic fungal infection, and many different patient-specific factors can contribute to the disease. CRS needs to be evaluated by a physician via endoscopy and/or CT imaging, and some patients may need endoscopic surgery; therefore, treatments for patients with rhinosinusitis symptoms lasting longer than 12 weeks are outside the scope of the community pharmacy practice.3
Treatment
The goals of therapy for both acute and chronic rhinosinusitis are to control infection, reduce tissue edema, facilitate drainage, maintain patency of the sinus ostia, and break the pathologic cycle that leads to CRS.15 Acute viral rhinosinusitis is a self-limiting condition. Since no pharmacotherapeutic interventions are proven to reduce the duration of illness, the goals of medical management are to relieve the symptoms of nasal obstruction and rhinorrhea.
Treatment of ABRS may include antibiotics to eliminate the infection, but, as stated before, the majority of bacterial infections will clear spontaneously. Since ABRS cannot generally be differentiated from its viral counterpart in the first 10 days, antibiotic therapy should be reserved for patients with severe symptoms for at least 3 to 4 consecutive days, signs of double sickening, and illness persisting for longer than 10 days without evidence of clinical improvement.8
Patients with facial swelling, edema around the eyes, abnormal vision, or mental status alterations may be experiencing intracranial or intraorbital extension of sinusitis, and should be referred immediately for urgent medical attention.11
Acute Viral Rhinosinusitis: Supportive therapy for AVRS should be tailored to an individual’s symptom profile. Nonsteroidal anti-inflammatory drugs (NSAIDs) and acetaminophen are recommended for the pain and discomfort associated with mucosal swelling. Oral and topical decongestants reduce symptoms of rhinosinusitis by constricting the blood vessels of the nasal mucosa, thereby reducing edema and inflammation. Topical decongestants, such as oxymetazoline, can provide more relief than systemic decongestants due to their increased potency and local administration, but they should be used for no more than 3 days to avoid rebound congestion.3 Systemic decongestants should be used with caution in patients with cardiovascular disease or uncontrolled hypertension.
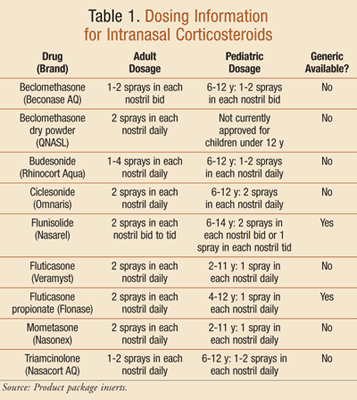
Intranasal corticosteroids reduce edema and inflammation and have been shown to reduce symptoms of acute rhinosinusitis (TABLE 1). In clinical trials, intranasal mometasone furoate has been shown to effectively reduce symptoms of congestion associated with rhinosinusitis both as monotherapy and in combination with amoxicillin-clavulanate.16 Similar results have been achieved with intranasal fluticasone, flunisolide, and budesonide. Intranasal corticosteroids are unlikely to cause systemic adverse effects because of low systemic bioavailability. The bioavailability of fluticasone propionate is <1%, and the bioavailability of mometasone furoate is ≤0.1%.16 Systemic therapy with corticosteroids should be avoided in most patients due to limited evidence and an increased risk of adverse events. Potential effects of short-term systemic corticosteroid use include hyperglycemia, hypertension, increased appetite, and insomnia.3
Antihistamines are often prescribed for symptom relief due to their drying effect, but there is no evidence to support their use in infectious rhinosinusitis. Overdrying the mucous membrane can impair mucus clearance and cause additional discomfort. Antihistamines should only be recommended to patients with symptoms suggesting a significant allergic component.3
Saline nasal rinses are used to soften viscous secretions and improve mucous clearance. A 2010 Cochrane literature review found three small trials showing limited evidence to support the use of sinus rinses in acute rhinosinusitis.17 In one study, 389 medium-risk patients using daily nasal saline irrigation saw a decrease in antibiotic use (relative effect 0.44, 95% confidence interval [CI], 0.18-1.09) and time away from work or school (relative effect 0.29, 95% CI, 0.16-0.53).18 Better evidence exists to support the role of sinus rinses in treating recurrent rhinosinusitis. As saline rinse product have few medication-related side effects and drug interactions, pharmacists can feel comfortable recommending their use to patients in the community setting.
Zinc has frequently been used to reduce the severity and duration of colds and sinus infections, but recent reports of long-term or permanent anosmia associated with zinc use has led the FDA to advise against the use of zinc-containing products.19
Acute Bacterial Rhinosinusitis: The symptoms of bacterial and viral rhinosinusitis are almost indistinguishable within the first 10 days. As such, patients presenting with fewer than 10 days of nonsevere symptoms, including mild pain and fever ≤101˚ F (38.3˚ C), should be managed symptomatically as previously discussed. Antibiotics should be initiated when signs and symptoms of acute rhinosinusitis do not improve within 10 days, in patients who experience a secondary worsening after initial improvement of symptoms (double sickening), or in patients with severe symptoms lasting for 3 to 4 days.8
Initial antibiotic choice should be based on a number of factors including safety, cost, and efficacy against microorganisms likely to cause ABRS. A review of aspiration studies in adult ABRS shows that the most commonly isolated organisms include S pneumoniae (20%-43%), H influenzae (22%-35%), and M catarrhalis (2%-10%).3 To prevent bacterial resistance, antibiotics with a narrow spectrum of activity are preferred. The 2012 IDSA guidelines for the treatment of ABRS suggest amoxicillin-clavulanate as first-line therapy for patients requiring antibiotics. Previously, amoxicillin alone had been recommended for initial therapy. For patients with a penicillin allergy, doxycycline or a respiratory fluoroquinolone (levofloxacin, moxifloxacin) should be used. Macrolide antibiotics (erythromycin, azithromycin) are no longer recommended for the treatment of ABRS.8
The recommended duration of therapy is 5 to 7 days for adults and 10 to 14 days for pediatric patients, based on the typical therapy used in randomized, controlled trials of antibiotics in ABRS10; however, no significant differences in cure rates are obtained with a shorter, 3- to 4-day course of therapy.3
Some other factors may dictate the use of alternative therapy. A history of antibiotic use in the previous 4 to 6 weeks increases the risk of antibiotic-resistant microorganisms. Guidelines suggest a fluoroquinolone or high-dose amoxicillin-clavulanate (2,000 mg/125 mg by mouth twice a day) for such patients.8
Treatment failure is defined as progression of symptoms during antibiotic therapy or no improvement after 7 days of therapy. These patients should be reevaluated for a nonbacterial cause or infection with drug-resistant bacteria. If treatment with narrow-spectrum antibiotics is insufficient, a more broad-spectrum fluoroquinolone or high-dose amoxicillin-clavulanate should be considered. Refractory cases should be referred to an otolaryngologist, who may obtain endoscopic cultures to guide therapy.3
Chronic Rhinosinusitis: The symptoms of CRS, regardless of origin, can be managed similarly to those of acute rhinosinusitis. Better evidence for the use of daily nasal saline rinse exists for CRS sufferers. One randomized, controlled trial of 76 subjects, mostly in the family practice setting, found that CRS sufferers using once-daily saline nasal rinses for 6 months used less nasal spray, required fewer antibiotics, and experienced fewer 2-week periods with sinus-related symptoms.20
As previously discussed, patients with symptoms of rhinosinusitis lasting more than 12 weeks require medical management, including imaging studies, endoscopy, and potentially surgery. Therefore, these patients should be referred to a physician.
Conclusion
Each year, 1 in 7 noninstitutionalized Americans over the age of 18 years is diagnosed with acute rhinosinusitis.21 Despite the fact that 70% of patients improve without antibiotics, rhinosinusitis is still the fifth leading indication for practitioners to prescribe antimicrobials.1 Community pharmacists are poised to play a significant role in the appropriate treatment of rhinosinusitis through proper recognition of cardinal symptoms and clinical manifestations, patient education, and evidence-based pharmacotherapy.
REFERENCES
1. Anand VK. Epidemiology and economic impact of rhinosinusitis. Ann Otol Rhinol Laryngol Suppl. 2004;193:3-5.
2. Owings MF, Kozak LJ. Ambulatory and inpatient procedures in the United States, 1996. National Center for Health Statistics. Vital Health Stat. 1998;13:1-119.
3. Rosenfeld RM, Andes D, Bhattacharyya N, et al. Clinical practice guideline: adult sinusitis. Otolaryngol Head Neck Surg. 2007;137(suppl):S1-S31.
4. Fokkens W, Lund V, Mullol J; European Position Paper on
Rhinosinusitis and Nasal Polyps Group. EP30S 2007: European position
paper on rhinosinusitis and nasal polyps 2007. A summary for
otorhinolaryngologists. Rhinology. 2007;45:97-101.
5. Hickner JM, Bartlett JG, Besser RE, et al. Principles
of appropriate antibiotic use for acute rhinosinusitis in adults:
background. Ann Intern Med. 2001;134:498-505.
6. Gill JM, Fleischut P, Haas S, et al. Use of antibiotics
for adult upper respiratory infections in outpatient settings: a
national ambulatory network study. Fam Med. 2006;38:349-354.
7. Young J, DeSutter A, Merenstein D, et al. Antibiotics
for adults with clinically diagnosed acute rhinosinusitis: a
meta-analysis of individual patient data. Lancet. 2008;371:908-914.
8. Chow AW, Benninger MS, Brook I, et al. IDSA clinical
practice guidelines for acute bacterial rhinosinusitis in children and
adults. Clin Infect Dis. 2012;54:e1-e45.
9. Gorbach SL, Falagas M, eds. The 5-Minute Infectious Diseases Consult. Boston, MA: Wolters Kluwer; 2001.
10. Scheid DC, Hamm RM. Acute bacterial rhinosinusitis in adults: part I. Evaluation. Am Fam Physician. 2004;70:1685-1692.
11. Ah-See KW, Evans AS. Sinusitis and its management. BMJ. 2007;334:358-361.
12. Gwaltney JM jr. Acute community-acquired sinusitis. Clin Infect Dis. 1996;23:1209-1223.
13. Stringer SP, Mancuso AA, Avino AJ. Effect of a topical vasoconstrictor on computer tomography of paranasal sinus disease. Laryngoscope. 1993;103:6-9.
14. Lanza DC, Kennedy DW. Adult rhinosinusitis defined. Otolaryngol Head Neck Surg. 1997;117(suppl):S1-S7.
15. Cooper DH, Krainik AJ, Lubner SJ, et al, eds. The Washington Manual of Medical Therapeutics. 32nd ed. St. Louis, MO: Wolters Kluwer; 2007.
16. Meltzer EO, Caballero F, Fromer LM, et al. Treatment of congestion in upper respiratory diseases. Int J Gen Med. 2010;3:69-91.
17. Kassel JC, King D, Spurling GK. Saline nasal irrigation for acute upper respiratory tract infections. Cochrane Database Syst Rev. 2010;(3):CD006821.
18. Slapak I, Skoupa J, Strnad P, et al. Efficacy of
isotonic nasal wash (seawater) in the treatment and prevention of
rhinitis in children. Arch Otolaryngol Head Neck Surg. 2008;134:67-74.
19. Zicam cold remedy nasal products (cold remedy nasal
gel, cold remedy nasal swabs, and cold remedy swabs, kids size). FDA.
Safety alerts for human medical products. June 16, 2009.
www.fda.gov/Safety/MedWatch/SafetyInformation/SafetyAlertsforHumanMedicalProducts/ucm166996.htm.
Accessed March 22, 2012.
20. Rabago D, Zgierska A, Mundt M, et al. Efficacy of
daily hypertonic saline nasal irrigation among patients with sinusitis: a
randomized controlled trial. J Fam Pract. 2002;51:1049-1055.
21. Pleis JR, Lucas JW, Ward BW. Summary health statistics for US adults: National Health Interview Survey, 2008. Vital Health Stat 10. 2009;(242):1-157.
To comment on this article, contact rdavidson@uspharmacist.com.






