US Pharm. 2011;36(6):HS-3-HS-7.
In 2010, the urinary bladder was the fourth most common site of new cancer cases in the United States, with an estimated 70,530 new cases and 14,680 deaths.1 Three percent of all cancer-related deaths are due to bladder cancer (BC).2 BC is more common in men, with a male:female ratio of 4:1.3 Approximately 50% to 70% of BC cases will recur, most within 5 years.4 Because of its high recurrence rate and its propensity to progress and metastasize, thereby requiring long-term follow-up and treatment, BC is considered a chronic disease and the most expensive of all cancers.5
Urothelial, or transitional, carcinoma accounts for 90% of all BC. Five percent of cases are squamous cell carcinoma, and 1% are adenocarcinoma.6 BC is classified as carcinoma in situ (CIS), non–muscle-invasive disease, or muscle-invasive disease (discussed below). This article will provide a review of BC and its treatment.
Risk Factors
Cigarette smoking is the largest risk factor for BC. It is thought that the aromatic amines found in cigarette smoke are the carcinogens responsible for the increased risk of BC in smokers.4 Smoking duration and the number of cigarettes smoked per day directly correlate with the incidence of BC. The risk is higher in individuals who started smoking at a young age and in people who were exposed to secondhand smoke as children.7 BC that results from smoking is associated with a higher mortality rate.8 The relative risk of dying from BC caused by smoking is 3.3 for current smokers and 2.1 for former smokers.4,8 BC risk decreases with smoking cessation; after 4 years of cessation the risk decreases by up to 40%, and after 25 years the risk decreases by 60%.4,7
Certain occupations are associated with an increased risk of developing BC. Occupational exposure accounts for 20% to 25% of all BC cases.7 BC usually occurs 30 to 50 years after occupational exposure. High-risk occupations include the painting and leather industries, the automotive industry, truck driving, metalworking, paper and rubber manufacturing, foundry work, dry cleaning, dental technology, and hairdressing.4
Other factors that increase BC risk are radiation exposure, such as that used to treat prostate cancer in men and gynecologic cancers in women; chronic urinary tract infections (UTIs); ongoing irritation from catheters and bladder stones; and nonfunctioning bladder.4,7 Age appears to increase the risk; the median age upon presentation with BC is 72 years for men and 74 years for women.4 BC development usually occurs 6 to 13 years after exposure to cyclophosphamide.4,7 Heredity does not appear to increase the risk, but some genetic markers have been associated with BC.4
Signs and Symptoms
Hematuria is the hallmark sign of BC, occurring in 85% of patients at presentation; however, blood in the urine is also associated with other conditions, such as UTIs, stones, and benign prostatic hyperplasia.4,9 Increased urinary frequency and dysuria may also be presenting symptoms.4,7 Since these are all present in UTIs, which are more common in women, women may be misdiagnosed, which results in delayed treatment and more advanced disease.4 More advanced disease may present with pelvic pain and all the symptoms of urinary obstruction.7
Staging and Diagnosis
The American Joint Committee on Cancer TNM (primary tumor, regional lymph nodes, and distant metastasis) Staging System is used for staging BC (TABLE 1). Correct staging can estimate the prognosis and risk of recurrence and is used to determine treatment strategies.4 Understaging, a common problem, may result in incorrect treatment decisions.10 There is a 10% chance that a high-grade Ta or T1 lesion is really muscle-invasive disease.8

The gold standard for diagnosis is cystoscopy. Intraurethral lidocaine is used to perform this procedure, in which abnormal tissue is resected.4,8 This resection is called transurethral resection of bladder tumor (TURBT). Since cystoscopy may miss CIS, fluorescent agents (most commonly, 5-aminolevulinic acid or hexaminolevulinate) may be used to highlight tissues with an elevated metabolism.4 False-positives with the use of fluorescent agents may be caused by recent TURBT or inflammation.3,8 The tissue retrieved via TURBT should be biopsied.8,11
Urinary cytology is an important adjunct to cystoscopy and is helpful for identifying high-grade tumors, such as CIS. The presence of exfoliated cancerous cells can indicate cancer anywhere along the urinary tract, and the absence of cancer cells does not rule out the presence of a low-grade lesion. A false-positive may result from UTIs, stones, or intravesical instillation.3,8
CT and MRI may be used to determine the stage of BC; however, they are unable to accurately detect early metastatic disease.4 Ultrasonography is being used more frequently and is advantageous because it does not require contrast agents.8 Another imaging modality is IV urography. If invasive disease is suspected, diagnostic imaging should be done prior to TURBT because inflammation from TURBT can be impossible to distinguish from tumor growth in the perivesical fat.7
Treatment
TURBT performed during cystoscopy is a treatment as well as a diagnostic tool. All visible lesions should be removed, along with muscle tissue, to ensure complete resection and proper staging.3,6,8 Performance of a second TURBT is recommended at 2 to 6 weeks. Residual tumors may be identified and removed with a follow-up TURBT, and the correct disease stage can be confirmed.8,10 Thirty percent of patients undergoing a second TURBT are upstaged to muscle-invasive disease.3
TURBT alone is not recommended for most patients. Forty-five percent of patients receiving TURBT alone have a recurrence within 1 year.6 Only patients with disease limited to the superficial muscle layer and negative follow-up biopsies may be considered as candidates for TURBT alone.7 Treatment used adjunctively with TURBT depends on whether the disease is non–muscle invasive or muscle invasive.
Non–Muscle-Invasive Disease
Approximately 70% to 85% of new cases of BC are non–muscle-invasive.4,5 Non–muscle-invasive disease includes CIS, Ta, and T1 lesions, and 10% to 30% of cases will progress to muscle-invasive disease.4 An estimated 5,000 cases are CIS, a flat, high-grade lesion that is considered a precursor to invasive disease.9,12 CIS is often present with Ta and T1 tumors.8
Treatment decisions are made based on the risk of recurrence and progression to muscle-invasive disease. Factors used to predict these risks include the number of tumors, stage, grade, recurrence at 3 months, presence of CIS, and initial tumor >3 cm.4,8 The European Organisation for Research and Treatment of Cancer has a detailed formula for predicting recurrence and progression based on the number of tumors, tumor size, prior recurrence rate, T category, concomitant CIS, and tumor grade (www.eortc.be/tools/
Intravesical chemotherapy (IC) is the instillation of an agent into the bladder during or immediately (£24 h) after TURBT.4 The American Urological Association (AUA) and the European Society for Medical Oncology (ESMO) recommend an immediate instillation of IC following TURBT for uncomplicated non–muscle-invasive disease.6,11 Because it is confined to the bladder, IC reduces systemic toxicity and destroys circulating and residual tumor cells.8,12 Contraindications include deep resection, bladder perforation, and prior allergy to the agent.10 IC reduces the risk of recurrence (by approximately 500 days), but not the risk of progression.4,8 TABLE 2 lists the chemotherapeutic agents used intravesically. Device-assisted therapy with IC is presently being studied. Electromotive drug administration enhances drug penetration into the urothelium. Preliminary results have shown improved response and overall survival rates; however, more research needs to be done.10
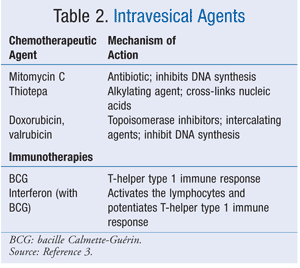
Intravesical bacille Calmette-Guérin (BCG) is sometimes used in addition to TURBT and IC. BCG may reduce the risk of recurrence, but not progression.6,7 There is no evidence that a single instillation of BCG is superior to mitomycin C; however, induction followed by maintenance therapy with BCG may be superior to the equivalent with mitomycin C, especially in high-risk patients.6,10 The ESMO recommends that high-risk patients receive BCG after an initial TURBT.9,11,13 High-risk patients are those with recurrence, large tumors, deeply invasive tumors, poorly differentiated tumors, or CIS.11 BCG is not indicated in patients with a low risk of recurrence or progression.4
In primary CIS, intravesical BCG has demonstrated a complete response rate of 83% to 93%, and the European Association of Urology (EAU) recommends this as first-line treatment in patients with this form of disease. Fifty percent of these patients will experience recurrent disease that is more advanced; therefore, patients with an incomplete response at 9 months, tumor recurrence within the bladder, or extravesical recurrence should undergo radical cystectomy (RC).7 Likewise, patients with high-risk T1 disease are candidates for cystectomy.2
Often, a single BCG treatment is not enough; 20% to 40% of patients have recurrence. A second treatment can improve the recurrence rate.4 The EAU recommends that BCG be given as maintenance therapy; however, the optimum dose, schedule, and BCG strain remain unclear.4,8,9 EAU guidelines recommend 3 weekly instillations of BCG, and patients with a high risk of progression should be given BCG for a year.8 AUA guidelines are a little more stringent, with an initial BCG (2-3 wk post-TURBT) followed by maintenance therapy at weeks 3, 6, 12, 18, 24, 30, and 36 (if tolerated).6 There is no consensus on what constitutes BCG failure; however, if at 3 months there is evidence of persistent disease, the prognosis is poor. BCG is associated with many adverse events (AEs), including sepsis and death; however, fewer than 5% of patients experience serious AEs. Cystitis is the most common AE.4
With BCG failure, treatment options are limited and RC should be considered.2 Determining whether a patient should undergo RC or salvage BCG treatment after BCG failure depends on risk stratification that takes into account the probability of progression and treatment failure.10 With failure occurring after 12 months of initial induction, a lower dose of BCG plus interferon-alpha is an option.13 A third course is never indicated.10
The advantages of early RC with BCG failure are improved chance of a cure and appropriate treatment for understaged carcinoma.10 Delayed RC in high-risk patients may be associated with increased mortality.8 Regardless, substantial morbidity (30%) and mortality (1%-6%) are associated with RC. Changes in sexual, gastrointestinal, and genitourinary function compromise quality of life.10 Some patients are not candidates for RC because they have comorbidities or wish to spare the bladder.13
There has been some success with weekly gemcitabine instillations for BCG failure, but this regimen is not recommended for high-grade lesions.10,13 In patients with CIS that is refractory to intravesical BCG, valrubicin may be used. A 21% complete response rate has been demonstrated with valrubicin; however, at 30 months, only 8% of subjects were disease free, and 56% underwent RC.2,13 Because of the propensity of BC to recur and progress, follow-up is critical to successful treatment.
For follow-up of non–muscle-invasive patients, the EAU recommends 3-month post-TURBT cystoscopy in all patients. Patients with a low risk of recurrence and progression should undergo cystoscopy 1 year after TURBT, and then annually for 5 years. Patients with a high risk of progression should undergo urine cytology and cystoscopy 3 months after TURBT. If negative, cystoscopy and cytology should be performed every 3 months for 2 years, every 4 months in year 3, every 6 months in years 4 and 5, and annually thereafter. For this group, annual upper-UT examination is also recommended. Approximately one-third of patients have an intermediate risk of progression and should be followed up with cystoscopy and cytology, with the intervals determined by physician and patient preference.8
Muscle-Invasive Disease
Approximately one-third of patients diagnosed with muscle-invasive BC have metastatic disease at the time that the first tumor is treated. Although men are more likely to develop BC, women are more likely to be diagnosed with metastatic disease.7 It is important to determine the presence of distant metastasis prior to treatment selection. The most common sites of metastasis are the lungs, bones, and liver.4
For localized muscle-invasive disease, RC is the standard treatment.7,11 In this procedure, the bladder and adjacent organs (prostate and seminal vesicles in men; uterus and adnexa in women) are removed.7 Although it is desirable to preserve the bladder, delaying RC is thought to increase the risk of lymph node metastasis by 26%.7
Neoadjuvant therapy (NT)—administering chemotherapy prior to RC—is a hotly debated subject. NT can help determine the sensitivity of the carcinoma to the selected chemotherapeutic agents. Other advantages include better patient tolerability to chemotherapy prior to RC and delivery of therapy at the earliest time, when occurrence of micrometastatic disease is expected to be low. Among the disadvantages associated with the use of NT are staging errors and overtreatment. Also, the delay in RC may worsen outcomes in patients not sensitive to chemotherapy. AEs may affect the outcome of surgery and the type of urinary diversion.7
Chemotherapy should include a cisplatin-containing regimen, either MVAC (methotrexate, vinblastine, doxorubicin, and cisplatin) or GC (gemcitabine and cisplatin). Neither combination has been shown to be superior to the other; however, GC is less toxic.7,11,14
One study compared neoadjuvant therapy (3 cycles of MVAC) plus cystectomy with cystectomy alone in 307 randomly assigned patients with muscle-invasive BC. The median survival of patients receiving cystectomy alone was 46 months versus 77 months for participants receiving combination therapy.15 In general, there is a 5% to 7% overall survival advantage with NT; therefore, it is recommended that cisplatin-based NT be used in patients with muscle-invasive disease.7,11 NT is not recommended in patients with impaired renal function.7
In addition to renal impairment, certain factors—such as advanced age, poor performance status, and comorbidities (neuropathy, congestive heart failure, hearing loss)—may preclude the use of cisplatin. In these patients, carboplatin may be used.2,16 One combination is paclitaxel, gemcitabine, and carboplatin.17
The use of adjuvant chemotherapy postoperatively has not been shown to have a benefit. Additionally, patients often cannot tolerate systemic chemotherapy after RC. The EAU guidelines recommend adjuvant therapy for clinical trials, but not for routine use.7
Antiangiogenic therapy is under investigation as second-line therapy.11 Angiogenesis, the development of blood vessels from existing vasculature, is necessary for tumor growth and metastasis.18 As the rate-limiting step in tumor growth, it is likely involved in tumor invasion.17 Vascular endothelial growth factor (VEGF) is the most important stimulator of angiogenesis, and higher serum levels of VEGF indicate a more advanced cancer stage.17,18 Bevacizumab is a monoclonal antibody that binds to all isoforms of VEGF, thereby blocking its activity. Clinical trials are currently being performed to evaluate the role of bevacizumab (with other agents) in the treatment of BC.18 Sunitinib also inhibits VEGF, as well as other angiogenic factors; several studies are underway to determine its role in BC treatment.18
Epidermal growth factor receptor (EGFR) regulates the activity of VEGF and other factors that stimulate angiogenesis.18 Overexpression of EGFR in bladder cancer (31%-48%) is associated with poorer outcomes.18 Cetuximab has activity against EGFR and has been shown to inhibit BC tumor growth in vitro and in vivo.18 Current studies examining cetuximab (with other agents) have produced favorable results.17,18 Another promising agent affecting the activity of EGFR is gefitinib.17
Systemic chemotherapy is used for metastatic disease. EAU guidelines recommend first-line treatment (for patients suited to cisplatin use) to be GC, MVAC (preferably with granulocyte colony-stimulating factor [G-CSF]), or high-dose MVAC with G-CSF. Patients unsuited to cisplatin use should undergo combination carboplatin therapy or single-agent therapy. Vinflunine should be used as second-line treatment in patients who progress after platinum-based chemotherapy. Patients with metastatic bone disease should receive biphosphonates.7
There are no established guidelines for follow-up. The ESMO suggests that patients treated with bladder-sparing strategies undergo cystoscopy and urinary cytology every 3 months for the first 2 years and every 6 months thereafter, and that clinical control be evaluated in RC patients every 3 months for the first 2 years and every 6 months thereafter.11
Conclusion
BC is associated with frequent recurrences and a propensity to progress. Because of this, it is considered a chronic disease and one of the most expensive cancers to treat. Many variables need to be taken into account in determining treatment and follow-up of BC patients. Many treatment strategies may be used, and patient and physician preferences should be factors when a course of action is being decided on.
RC is often required to achieve a cure, but there are chemotherapeutic regimens that may be used in lieu of, or in addition to, RC. MVAC and GC are of equal efficacy, with GC having a more favorable toxicity profile. Other agents currently being studied are aimed at inhibiting angiogenesis, and preliminary results look promising.
REFERENCES
1. Jemal A, Siegel R, Xu J, Ward E. Cancer statistics, 2010. CA Cancer J Clin. 2010;60:277-300.
2. Vaishampayan U. Systemic therapy of advanced urothelial cancer. Curr Treat Options Oncol. 2009;10:256-266.
3. Sexton WJ, Wiegand LR, Correa JJ, et al. Bladder cancer: a review of non-muscle invasive disease. Cancer Control. 2010;17:256-268.
4. Jacobs BL, Lee CT, Montie JE. Bladder cancer in 2010: how far have we come? CA Cancer J Clin. 2010;60:244-272.
5. van Rhijn BW, Burger M, Lotan Y, et al. Recurrence and progression of disease in non-muscle-invasive bladder cancer: from epidemiology to treatment strategy. Eur Urol. 2009;56:430-442.
6. Hall MC, Chang SS, Dalbagni G, et al. Guideline for the management of nonmuscle invasive bladder cancer (stages Ta, T1, and Tis): 2007 update. J Urol. 2007;178:2314-2330.
7. Stenzl A, Cowan NC, De Santis M, et al. Guidelines on Bladder Cancer: Muscle-Invasive and Metastatic. Arnhem, Netherlands: European Association of Urology; 2010. www.uroweb.org/gls/pdf/Muscle%
8. Babjuk M, Oosterlinck W, Sylvester R, et al. Guidelines on TaT1 (Non-Muscle Invasive) Bladder Cancer. Arnhem, the Netherlands: European Association of Urology; 2010. www.uroweb.org/gls/pdf/TaT1%
9. Williamson SR, Montironi R, Lopez-Beltran A, et al. Diagnosis, evaluation and treatment of carcinoma in situ of the urinary bladder: the state of the art. Crit Rev Oncol Hematol. 2010;76:112-126.
10. Kulkarni GS, Hakenberg OW, Gschwend JE, et al. An updated clinical analysis of the treatment strategy for newly diagnosed high-grade T1 (previously T1G3) bladder cancer. Eur Urol. 2010;57:60-70.
11. Bellmunt J, Albiol S, Kataja V. Invasive bladder cancer: ESMO clinical recommendations for diagnosis, treatment and follow-up. Ann Oncol. 2010;20:iv79-iv80.
12. Burke J. Virus therapy for bladder cancer. Cytokine Growth Factor Rev. 2010;21:99-102.
13. Yates D, Rouprêt M. Failure of bacille Calmette-Guérin in patients with high risk non-muscle-invasive bladder cancer unsuitable for radical cystectomy: an update of available treatment options. BJU Int. 2010;106:162-167.
14. von der Maase H, Hansen SW, Roberts JT, et al. Gemcitabine and cisplatin versus methotrexate, vinblastine, doxorubicin, and cisplatin in advanced or metastatic bladder cancer: results of a large, randomized, multinational, multicenter, phase III study. J Clin Oncol. 2000;17:3068-3077.
15. Grossman HB, Natale RB, Tangen CM, et al. Neoadjuvant chemotherapy plus cystectomy compared with cystectomy alone for locally advanced bladder cancer. N Engl J Med. 2003;349:859-866.
16. Sonpavde G, Sternberg CN, Rosenberg JE, et al. Second-line systemic therapy and emerging drugs for metastatic transitional-cell carcinoma of the urothelium. Lancet Oncol. 2010;11:861-870.
17. Yang X, Flaig TW. Novel targeted agents for the treatment of bladder cancer: translating laboratory advances into clinical application. Int Braz J Urol. 2010;36:273-282.
18. Pinto A, Redondo A, Zamora P, et al. Angiogenesis as a therapeutic target in urothelial carcinoma. Anticancer Drugs. 2010;21:890-896.
19. Urinary bladder. In: Edge SB, Byrd DR, Compton CC, et al, eds. AJCC Cancer Staging Manual. 7th ed. Chicago, IL: American Joint Committee on Cancer; 2010:497-506.
To comment on this article, contact rdavidson@uspharmacist.com.





