US Pharm. 2011;36(6):HS-25-HS-28.
Potassium iodide (KI) is an inorganic compound that is available from three manufacturers under different brand names as an antidote to radiation exposure. From a chemistry point of view, it is made from potassium hydroxide and iodine, and it is the most produced iodide compound in the world. It is preferred over sodium iodide salt because it is less hygroscopic and easier to handle; it is an odorless and stable white crystalline powder. Among its other applications, it is used in the photography industry to form silver iodide and in chemical laboratories as a source of iodide in organic synthesis. KI is also used in biomedical research as a fluorescence quencher through its iodide ion. Upon extended exposure to air, KI becomes yellow as a result of the liberation of iodine, and small quantities of iodate may be formed due to oxidation.1
Although KI has several medical and nutritional applications, its most important application is as an antidote to radiation. Recently, KI has been in high demand in the United States due to the warning about remote radiation exposure from the recent Japanese nuclear plant radioactive releases.2 For medical purposes, a saturated solution of KI is used to treat lung congestion (as an expectorant) and sporotrichosis (a fungal infection) and as an antiseptic in sore throats.
KI's value as a radiation protective or antithyroid agent was demonstrated at the time of the Chernobyl nuclear accident when Russian authorities distributed KI in a 30-km zone around the plant. The purpose was to protect residents from the radioactive iodine, the most abundant and highly carcinogenic material found in nuclear reactors, that had been released by the damaged reactors.2,3
KI is supplied as 130-mg tablets for emergency purposes. It may also be administered as a viscous saturated solution of potassium iodide (SSKI). This formulation provides 250 mg of iodide per each five drops. According to the USP, each two drops of this solution (containing 100 mg of iodide) have the same amount of iodide as a 130-mg tablet of KI (130 mg = 100 mg iodide).2
Thyroid Protection After Nuclear Accidents/Events
KI was approved by the FDA in 1982 to protect the thyroid gland from radioactive iodine released accidentally from nuclear power plants or during a nuclear attack. In these events, many radionuclide products may be discharged to the atmosphere. Of these products, I131 is one of the most common, and it is particularly dangerous to the thyroid gland.1
By saturating the body with a source of stable iodide such as KI prior to exposure, inhaled or ingested I131 will be excreted from the body, and as a result, radioiodine uptake by the thyroid gland will not take place. The protective effect of KI lasts approximately 24 hours. For optimal prophylaxis, KI must be dosed daily until the risk of significant exposure to radioiodine by either inhalation or ingestion no longer exists.
Doses of 130 mg KI provide 100 mg iodide, which is roughly 700 times greater than the normal nutritional need for iodine of 150 mcg (0.15 mg, as iodide) per day for an adult. The KI in iodized salt is insufficient for this use, as 80 tablespoons would be needed to equal one tablet. See TABLE 1 for the recommended FDA dosing regimen for humans.2 KI cannot protect against any other causes of radiation poisoning or against radionuclide other than radioisotopes of iodine.
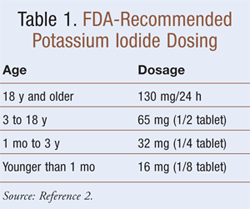
Dosing Equivalency: For protection of the thyroid against I131 contamination, the convenient standard 130-mg KI pill is used, if available. As noted, the equivalent two drops of SSKI may be used for this purpose if the pills are not available.
Protecting Effect: When KI is administered simultaneously with radiation exposure, the protective effect is approximately 97%. KI given 12 and 24 hours before exposure yields a 90% and 70% protective effect, respectively. However, KI administered 1 and 3 hours after exposure results in an 85% and 50% protective effect, respectively. KI administered more than 6 hours after exposure is thought to have a negligible protective effect.4
It was reported on March 16, 2011, that KI tablets were given prophylactically to U.S. Naval air crewmembers flying within 70 miles of the Fukushima Daiichi nuclear plant damaged in the massive Japanese earthquake (8.9/9.0 magnitude) and ensuing tsunami on March 11, 2011. The measures were seen as precautions, and the Pentagon said no U.S. forces have shown signs of radiation poisoning. By March 20, the U.S. Navy had also instructed personnel coming within 100 miles of the reactor to take the pills as a precautionary measure.5,6
Radionuclide Iodine in Nuclear Medicine
As opposed to the harmful release of I131 in nuclear disasters, an iodide radionuclide in different radiopharmaceutical forms (and not necessarily as KI) is used in nuclear medicine for diagnostic and therapeutic purposes.7 The thyroid gland absorbs nearly all of the iodine in the blood. When enough of the medical form of I131 is taken into the thyroid, it can destroy the thyroid gland and any other thyroid cells (including cancer cells) that take up iodine, with little effect on the rest of the body. The radioactive iodine is usually given as a capsule or liquid. This treatment can be used to destroy any thyroid tissue not removed by surgery or to treat thyroid cancer that has spread to lymph nodes and other parts of the body.
Radioactive iodine therapy has been shown to improve the survival rate of patients with papillary or follicular thyroid cancer (differentiated thyroid cancer) that has spread to the neck or other body parts, and this treatment is now the standard practice in such cases. Radioactive iodine therapy cannot be used to treat undifferentiated and medullary thyroid carcinomas because these types of cancer do not take up iodine.
Although this intentional hypothyroidism is temporary, it often causes symptoms such as tiredness, depression, weight gain, sleepiness, constipation, muscle aches, and reduced concentration. An injectable form of thyrotropin is now available that can increase a patient's thyroid-stimulating hormone level to overcome some of these symptoms.
Many medical experts recommend that women avoid becoming pregnant for 6 months to a year after treatment. No ill effects have been noted in children whose parents received radioactive iodine in the past.
Other Medical Applications
There are a number of labeled and unlabeled medical applications for KI.
Preoperative Thyroidectomy: KI in doses of 50 to 250 mg (1-5 drops SSKI) three times/day is administered for 10 days before thyroid surgery.8
Thyrotoxic Crisis: SSKI can be an emergency treatment for hyperthyroidism or thyroid storm in doses of 250 mg (5 drops SSKI) every 6 hours. High amounts of iodide temporarily suppress secretion of thyroxine from the thyroid gland.8
Expectorant: KI is used for the symptomatic treatment of chronic pulmonary diseases complicated by mucus. Iodide solutions made from a few drops of SSKI added to drinks have been used as expectorant to increase the water content of respiratory secretions and encourage effective coughing.
Sporotrichosis (Cutaneous, Lymphocutaneous): Oral: Initially, five drops (SSKI) three times/day; increase to 40 to 50 drops (SSKI) three times/day as tolerated until 2 to 4 weeks after lesions have resolved (usual duration 3-6 months). KI has been used in symptomatic treatment of erythema nodosum for persistent lesions whose cause remains unknown. It has also been used in cases of erythema nodosum associated with Crohn's disease.9
Nutritional Applications
As a nutritional supplement, KI is the most common additive to table salt to prevent iodine deficiency in populations that consume little seafood. Because KI is slowly oxidized from air exposure, it is replaced in certain products by potassium iodate, which is not lost by oxidation.10
None of the available pharmaceutical formulations (SSKI or KI tablets) are intended for use as nutritional supplements, due to the very high concentration of iodine. The nutritional requirement for iodine in humans is only 150 mcg (0.15 mg) of iodide per day.
There are natural sources of iodide for nutritional purposes. Kelp is a natural KI source. One-quarter cup of kelp supplies only 9 calories and 276.7% of daily iodine needs. The iodide content is about 89 mcg/g in Asian varieties. Another natural source of iodide is seaweed. Dried, unrinsed seaweed (3-5 g) will provide the recommended daily allowance of 100 to 150 mcg of iodide for nutritional purposes. Other natural sources are seafood, asparagus, turnip greens, spinach, and Swiss chard.10
Adverse Reactions
There have been some reports of KI treatment causing swelling of the parotid gland (one of the three glands that secrete saliva), because of its stimulatory effects on saliva production. Other reported adverse reactions reported include irregular heartbeat, skin rash, diarrhea, metallic taste, stomach upset, joint pain, numbness, and tingling.
Chronic iodine poisoning from prolonged treatment or high doses can cause iodism, hypersensitivity reactions that are manifested as angioedema, cutaneous and mucosal hemorrhage, and serum sickness-like symptoms.4
Drug Interactions
Potassium salts may enhance the hyperkalemic effect of angiotensin-converting enzyme inhibitors, angiotensin II receptor blockers, and potassium-sparing diuretics. KI may enhance the hypothyroid effect of lithium. Antithyroid agents may diminish the anticoagulant effect of vitamin K antagonists. In all these cases, therapy should be carefully monitored, and dosing adjustments should be made.4
Pregnancy/Lactation Considerations
Iodide crosses the placenta and, therefore, may cause hypothyroidism and goiter in the fetus or newborn. The use of KI is contraindicated as an expectorant during pregnancy by the American Academy of Pediatrics (AAP). Its use for protection against thyroid cancer secondary to radioactive iodine exposure is considered acceptable based upon risk-to-benefit ratio, dose, and duration. The AAP recommends avoiding breast-feeding following radioactive iodine exposure. It may cause skin rash in a nursing infant.4
Conclusions
KI cannot prevent radioactive iodine from entering the body. KI can protect only the thyroid from radioactive iodine, but not other parts of the body. In addition, KI cannot reverse the health effects caused by radioactive iodine once damage to the thyroid has happened. KI cannot protect the body from radioactive elements other than radioactive iodine; if radioactive iodine is not present, taking KI is not protective.
Three companies (Anbex Inc., Fleming Pharmaceuticals, and Recipharm AB) currently have FDA approval for manufacturing and testing of KI. These companies offer IOSAT (130 mg), Thyroshield (65 mg), and Thyro-Safe (65 mg), respectively.
REFERENCES
1. U.S. Food and Drug Administration. FDA's guidance on protection of children and adults against thyroid cancer in case of nuclear accident. FDA talk paper. www.fda.gov/Drugs/
2. CDC fact sheet: Potassium iodide (KI). October 11, 2006. www.bt.cdc.gov/radiation/pdf/
3. Nauman J, Wolff J. Iodide prophylaxis in Poland after the Chernobyl reactor accident: benefits and risks. Am J Med. 1993;94(5);524-532.
4. Micromedex health series, 2010.
5. Availability of U.S. Food and Drug Administration (FDA)-approved potassium iodide for use in emergencies involving radioactive iodine. Nuclear Regulatory Commission Notice 88-15.
6. U.S. embassy gives potassium iodide to citizens. Wall Street Journal. March 25, 2011. http://blogs.wsj.com/
7. Kowalsky RJ, Falen SW. Radiopharmaceuticals in Nuclear Pharmacy and Nuclear Medicine. 2nd ed. Washington DC: American Pharmacists Association; 2007.
8. Nayak B, Burman K. Thyrotoxicosis and thyroid storm. Endocrinol Metab Clin North Am. 2006;35(4):663-86,vii.
9. Kauffman CA, Bustamante B, Chapman SW, et al. Clinical practice guidelines for the management of sporotrichosis: 2007 update by the Infectious Diseases Society of America. Clin Infect Dis. 2007;45(10):1255-1265.
10. Institute of Medicine. Dietary Reference Intakes for Vitamin A, Vitamin K, Arsenic, Boron, Chromium, Copper, Iodine, Iron, Manganese, Molybdenum, Nickel, Silicon, Vanadium, and Zinc. Washington DC: National Academy Press; 2001.
To comment on this article, contact rdavidson@uspharmacist.com.






