US Pharm. 2012;37(6):68-70.
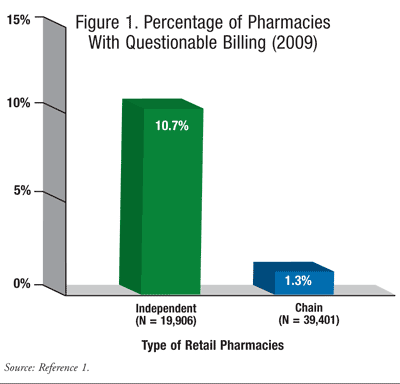
Government investigations and prosecutions, as well as penalties assessed against pharmacies, the pharmacy industry, and other health care providers, are on the rise. On May 10, 2012, the Department of Health and Human Services’ Office of Inspector General (HHS-OIG) released a report analyzing community pharmacy claims for Medicare services from 2009 to 2011.1 The report estimated that approximately 2,600 retail drugstores, or 4% of all U.S. pharmacies participating in Medicare, submitted suspicious or excessive billings to the federal government. The report also stated that 2,637 pharmacies submitted suspect billings for about $5.6 billion in 2009. In the context of all pharmacies operating in the country, chain stores were involved in 1% of the alleged fraudulent practices, and 11% of all independents submitted false or inflated claims for payment (FIGURE 1). The report did not identify any pharmacies by name.1,2
Allegations of Fraud
Some pharmacies dispensed unusually high percentages of painkillers and other controlled substances or expensive brand-name drugs in place of less expensive generics that were available and should have been dispensed according to Medicare plan guidelines.3 Other stores billed Medicare a per patient rate of more than 2.5 times the national average. In total, Medicare paid about $57 billion in 2009 for prescriptions dispensed by about 59,000 drugstores.
Acknowledging that some of the claims might be legitimate, investigators stated that “all pharmacies that bill for such extremely high amounts warrant further scrutiny.”3 Inspector General Daniel R. Levinson recommended that Medicare seek a legislative change mandating that insurers administering Medicare plans be required to report potential fraud to the government. That reporting duty does not exist at the present time, and it is only when the administrators voluntarily report suspicious billing claims that Medicare becomes involved.
Kevin Schweers of the National Community Pharmacists Association (NCPA), the trade-group representing independent pharmacies, noted that some of the “excessive” billings originating from independent pharmacies could occur because they service a higher percentage of aging, nursing home, hospice, and other severely ill patients on Medicare who tend to use larger quantities of more expensive drugs than do the chain pharmacies.3
Federal Investigations
One of the key factors in identifying and targeting suspicious billing activities has resulted from an increased use of multiple cooperating federal agencies. Ongoing interagency collaboration between the Medicare Fraud Strike Force and Health Care Fraud Prevention and Enforcement Action Team (HEAT) has been effective. Ties between the Department of Justice (DOJ), the Federal Bureau of Investigation (FBI), the HHS-OIG, and local U.S. Attorneys’ Offices (USAOs), as well as Medicaid Fraud Control Units (MFCUs), also assist in health care fraud investigations.4 Shared intelligence makes it easier for these agencies to detect, investigate, and prosecute large-scale fraud schemes. Advanced data-mining techniques will strengthen the links between investigatory and enforcement agencies, making large-scale investigations and prosecutions even easier for the government to pursue in the coming years.
The Health Care Fraud and Abuse Control Program (HCFAC) report, released in February 2012, states that “convictions increased by over 27% (from 583 to 743) between 2009 and 2011, and the number of defendants facing criminal charges filed by federal prosecutors in 2011 increased by 74% (from 821 to 1,430) compared with 2008.”5 During 2011, the DOJ opened 1,110 criminal health care fraud investigations involving 2,561 potential defendants. Federal prosecutors investigated 1,873 pending health care fraud criminal cases involving 3,118 potential defendants and filed 489 criminal cases against 1,430 defendants while obtaining 743 convictions against defendants charged with health care fraud-related crimes. In addition, HHS-OIG excluded 2,662 individuals and health care entities from participating in federal reimbursement programs for Medicare, Medicaid, and related health care services.
Not all of these prosecutions involved pharmacies. Many involved durable medical equipment providers as well as Medicare insurance administrators and long-term-care nursing facilities. Still, a substantial number of pharmacists, pharmacies, and programs operating for the benefit of pharmacy providers were identified in these cases.
Additional Cases
In an older case from 2008, CVS Caremark agreed to pay $38.5 million to settle charges that its Caremark division was illegally switching prescribed drugs to more expensive medications, thereby increasing Caremark’s profit margins.6 More recently, on October 14, 2010, CVS was ordered to pay $77.6 million in fines and returned profits stemming from a lawsuit alleging improper control in the sale of pseudoephedrine, used to make methamphetamine.7 CVS had to forfeit $2.6 million in profits from the illegal sale and pay a fine of $75 million, which at the time was the largest civil penalty ever paid under the Controlled Substances Act (CSA).
While not yet adjudicated, in 2011 the DOJ charged that CVS pharmacies in Sanford, Florida, ordered enough oxycodone to supply a population eight times its size.8 According to the Drug Enforcement Administration (DEA), in 2010 a single CVS pharmacy in Sanford ordered 1.8 million oxycodone pills, an average of 137,994 pills a month. Other pharmacies in Florida averaged 5,364 oxycodone pills a month. DEA investigators serving a warrant to the Sanford pharmacy on October 18, 2011, noted that “approximately every third car that came through the drive-thru lane had prescriptions for oxycodone or hydrocodone.” A pharmacist at that location stated to investigators that “customers often requested certain brands of oxycodone using street slang,” an indicator that the drugs were being diverted and not used for legitimate pain management.9
On April 29, 2011, CVS agreed to pay $17.5 million to settle Medicaid fraud allegations in a case initiated by a whistle-blower pharmacist in Minnesota based on a design flaw inserted into the company’s software billing system that automatically increased claims made on Medicare prescriptions.10 Take note that approximately $2.6 million was awarded to the pharmacist informant.
Also of interest, in 2008 the Walgreen Co. was sued for using a system that fraudulently inflated Medicaid billings in Minnesota.11 Walgreens settled the case for $9.9 million. The two pharmacist whistle--blowers in this case shared a $1.44 million reward for bringing the illegal billing scheme to the attention of government investigators. Perhaps not learning its lesson in that case, Walgreens settled another lawsuit alleging false claims for $7.9 million on April 20, 2012.11
One of the odder cases involved alleged Canadian pharmacies doing business with patients residing in the United States. On August 24, 2011, Google (the Internet search engine company) agreed to pay the federal government $500 million to settle charges that it knowingly accepted illegal advertisements from fraudulent Canadian pharmacies.12 The investigation found that Google was aware that some Canadian pharmacies that advertised on its site did not require a prescription for medications such as Ritalin and OxyContin. Google continued to accept their money and even assisted the pharmacies in placing ads and improving their Web sites. Under federal law, Web site operators are liable for advertising that violates U.S. laws.
In a more typical case, on February 7, 2012, Lam’s Pharmacy in Las Vegas, Nevada, agreed to pay $1 million to settle civil charges that it violated the federal CSA in exchange for dismissal of criminal charges it could have faced for drug trafficking.13 This is the largest civil fine ever levied against an independent pharmacy under the CSA. That, however, is not the end of this story. Lam’s Pharmacy will also surrender its DEA registration, meaning it no longer will be able to order, dispense, or even handle any controlled substances, and ownership of its assets will be sold. The business will be allowed to operate in Las Vegas under new ownership and a different name.
Analysis
The cases discussed here are only a small tip of the very large iceberg of fraud, waste, and abuse lawsuits and claims settled and pending over the past few years. One recurring theme seems to keep popping up. The cost of the fines, settlements, attorney fees, and overhead associated with these lawsuits hardly ever seems to stop the very large operators from making the same “mistakes” over and over again. Using the notion that the punishment should fit the crime, it seems at least reasonable that these companies should incur larger, more punitive penalties that would deter such behavior. Otherwise, business as usual will come to mean that the crime really does pay, even if they have to give a little bit back.
REFERENCES
1. Department of Health and Human Services, Office of
Inspector General. Retail pharmacies with questionable Part D billing
(OEI-02-09-00600). May 2012.
http://oig.hhs.gov/oei/reports/oei-02-09-00600.pdf. Accessed May 11,
2012.
2. Medicare overbilling. Detroit Free Press. May 11, 2012. www.freep.com/article/20120511/NEWS07/205110359/4-of-all-drugstores-suspected. Accessed May 11, 2012.
3. Wayne A. Suspicious Medicare bills found at 2,600 U.S. pharmacies. Bloomberg News.
May 11, 2012.
www.bloomberg.com/news/2012-05-10/suspicious-medicare-bills-found-at-2-600-u-s-pharmacies.html.
Accessed May 11, 2012.
4. Miner TA, Dunphy DP, Willis SD, et al. United States:
industry trends in criminal health care fraud enforcement—part III in a
continuing series on health care enforcement. Mintz Levin News. May 4, 2012. www.mondaq.com/unitedstates/x/175740/. Accessed May 11, 2012.
5. New tools to fight fraud, strengthen federal health
programs, and protect taxpayer dollars. Department of Health and Human
Services. February 14, 2012.
www.healthcare.gov/news/factsheets/2012/02/medicare-fraud02142012a.html.
Accessed May 11, 2012.
6. CVS Caremark agrees to pay $38.5M to settle allegations that it did not pass on rebates, discounts to patients, employers. Kaiser Health News. February 15, 2008. http://www.kaiserhealthnews.org/Daily-Reports/2008/February/15/dr00050454.aspx?p=1. Accessed May 11, 2012.
7. Ellis B. CVS to pay $77.6 million in meth case. CNN Money. October 14, 2010. http://money.cnn.com/2010/10/14/news/companies/cvs_meth/index.htm?hpt=T1. Accessed May 11, 2012.
8. Schoenberg T. Cardinal Health blocked from shipping painkiller in Florida. Bloomberg News.
February 29, 2012.
www.bloomberg.com/news/2012-02-29/cardinal-health-blocked-from-shipping-painkiller-in-florida-1-.html.
Accessed May 11, 2012.
9. Leinwand Leger DL. DEA aims big in Cardinal Health painkiller case. USA Today. February 28, 2012. www.usatoday.com/news/nation/story/2012-02-27/painkiller-abuse-DEA/53275844/1. Accessed May 11, 2012.
10. Jones BL. CVS settlement featured in Minnesota Lawyer. Pharmacy Fraud Whistleblowers. April 29, 2011. www.pharmacyfraudwhistleblowers.com. Accessed May 11, 2012.
11. Walgreens pharmacy chain pays $7.9 million to resolve false prescription billing case. Justice News. April 20, 2012. www.justice.gov/opa/pr/2012/April/12-civ-505.html. Accessed May 11, 2012.
12. Miller CC. Google reaches $500 million settlement with government. NY Times.
August 24, 2011.
http://bits.blogs.nytimes.com/2011/08/24/google-reaches-500-million-settlement-with-government/.
Accessed May 11, 2012.
13. Van Wey K. DEA fines Las Vegas pharmacy in violation of federal drug laws. Pill Mill Monitor.
February 8, 2012.
www.pillmillmonitor.com/2012/02/articles/dea-fines-las-vegas-pharmacy-in-violation-of-federal-drug-laws/.
Accessed May 11, 2012.
To comment on this article, contact rdavidson@uspharmacist.com.





