U.S. Pharm. 2012;37(6)(Generic suppl):12-20.
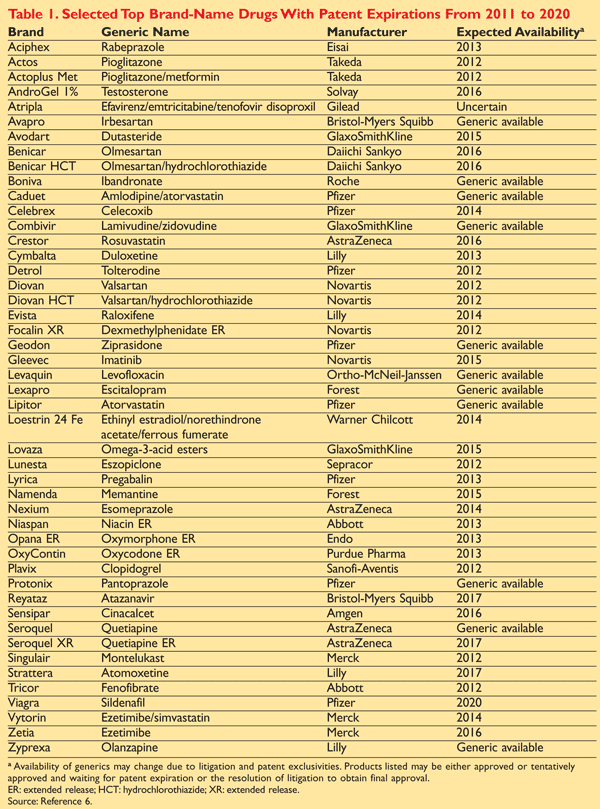
Beginning in 2010, the pharmaceutical industry faced one of the biggest waves of drug patent expirations in history, a phenomenon referred to as the “patent cliff.” A significant number of top-selling drugs in the history of the pharmaceutical industry will experience patent expirations over the next 5 years, paving the way for lower-priced generics. In November 2011 alone, four major drugs—Lipitor (atorvastatin), Caduet (amlodipine/atorvastatin), Combivir (lami vudine/zidovudine), and Solodyn (minocycline extended release tablet)—lost patent protection. Combined, these four drugs accounted for more than $7 billion in sales.1-5 In total, patents on drugs worth $12 billion expired by the end of 2011, and in 2012 that figure is expected to increase to more than $30 billion in annual sales. Furthermore, it is estimated that generic competition will have eroded $67 billion from top drug companies’ annual sales in the United States between 2007 and 2012 alone, as more than three dozen drugs lose patent protection during this time.6 That figure represents approximately 50% of the companies’ combined U.S. sales in 2007. With patents on many so-called blockbuster drugs about to expire, an estimated $250 billion in sales is at risk between now and 2015.1-5 Selected drugs whose patents have expired or will expire over the next several years are shown in TABLE 1 .6
Once drugs lose patent protection, lower-price generics quickly siphon off as much as 90% of their sales.2,3 For consumers, the savings from generics can be substantial. Drugs are granted 20 years of patent protection, although companies often do not get a product to market before as much as half of that period has already elapsed. Once a drug enters the market, however, patent protection can result in high profits, with gross profit margins exceeding 90%.2,3 When patents expire, generic makers offer the products at prices reported to average about 30% of the price of the brand-name originals.2,3 Although the generic industry will benefit in the short term, it will also see a slowdown in revenue growth after 2015 because fewer branded blockbusters will be coming off patent after this time.
As of 2010, patents had expired on a number of widely used drugs, including Advair (fluticasone propionate and salmeterol), Levaquin (levofloxacin), Cozaar (losartan), and Taxotere (docetaxel). In addition to the drugs mentioned above, in 2011 Bristol-Myers/Sanofi-Aventis lost exclusivity for Avapro (irbesartan), an angiotensin II receptor antagonist for treatment of hypertension, and Pfizer lost the glaucoma drug Xalatan (latanoprost ophthalmic solution). Other patents expiring in 2011 included Astelin (azelastine, Wallace) antihistamine nasal spray; Synarel (nafarelin, Roche) nasal spray for precocious puberty and endometriosis; Actos (pioglitazone, Takeda) for type 2 diabetes; the fertility drug Follistim (follitropin beta injection, Organon); and the HIV/AIDs drug Viramune (nevira-pine, Boehringer Ingleheim).1-6
Among drugs scheduled to come off patent in 2012 is AstraZeneca’s cholesterol-lowering drug Crestor (rosuvastatin) and its asthma drug Symbicort (budesonide/formoterol). Merck loses its top-selling asthma drug, Singulair (montelukast), as well as its migraine medication, Maxalt (rizatriptan), in 2012. GlaxoSmithKline’s (GSK) patent on the diabetes drug Avandia (rosiglitazone) and Forest Laboratories’ patent on the antidepressant Lexapro (escitalopram) also expire in 2012. Novartis’s Zometa (zoledronic acid), its bisphosphonate drug, and its Diovan (valsartan), an antihypertensive, both expire in 2012, as does the first patent protection for Pfizer’s Viagra (sildenafil).1-6 Some notable drug patent expirations and the associated financial impact for their manufacturers are presented below.
Lipitor, Protonix, and the Pfizer Product Line
Between 2010 and 2012, drugs that make up 42% of Pfizer’s pharmaceutical revenue, including Lipitor, Protonix (pantoprazole) and Geodon (ziprasidone), will all have lost patent protection. Protonix, a proton pump inhibitor, is used to treat gastroesophageal reflux disease and other gastric acid hypersecretory diseases, with an estimated cost of up to $173 per month. In 2007, Protonix earned Pfizer more than $1.9 billion, but these revenues had fallen to $480 million by 2010. This reduction in profit was, in part, a result of the launch of generic versions of pantoprazole by Teva and Sun Pharmaceuticals in 2007 and 2008. These generic manufacturers claimed that the original Protonix patent was invalid, but a court eventually ruled that the original patent for Protonix was indeed valid, resulting in a halt to generic manufacture and sale by Teva and Sun. Pfizer will continue to manufacture Protonix as well as an authorized generic through Wyeth, a company it acquired in 2009. Generic versions of atorvastatin and ziprasidone are already available in the U.S. 1-5
The anticholesterol drug Lipitor has been one of the best-selling drugs worldwide for a number of years and has been a major source of income for the world’s biggest drug company. Lipitor therapy can cost up to $168 per month. Lipitor was first marketed in 1998, and by 2006 it had reached peak sales of $12.9 billion, accounting for 27% of the company’s revenue. In 2010, with $10.8 billion in sales ($5.3 billion in the U.S.), Lipitor still accounted for nearly 16% of total revenue. In 2008, Pfizer entered into a licensing agreement with Indian generics manufacturer Ranbaxy Laboratories to sell atorvastatin in the U.S. effective in November 2011 and to have exclusivity for 180 days before other drug makers can enter the market. The only differences in the generic version will be in the size, shape, and/or color of the drug product. It is anticipated that Watson Pharmaceuticals and perhaps other manufacturers will also introduce a generic for Lipitor in the future.1-5
Pfizer also faces patent expirations for other big sellers including Viagra, the first of the phosphodiesterase inhibitors to be approved for erectile dysfunction; Celebrex (celecoxib), the first cyclo-oxygenase inhibitor to be marketed for inflammatory disease; and Lyrica (pregabalin), approved for treatment of fibromyalgia and neuropathies. These three drugs represented over $3 billion in sales in the U.S. alone in 2010. 1-5
Seroquel, Nexium, and the AstraZeneca Product Line
Seroquel (quetiapine), an atypical antipsychotic approved for a variety of conditions from depression to bipolar disorder and schizophrenia, was introduced in 1997 by the British pharmaceutical company AstraZeneca. Depending on the dose, the drug costs about $549 per month. Seroquel’s worldwide sales amounted to $5.3 billion in 2010 ($3.7 billion in the U.S), or nearly 16% of Astra’s revenues. Seroquel was originally destined to expire in September 2011 but received a 6-month extension from the FDA through a pediatric exclusivity agreement. A generic version is now available in the U.S. In total, drugs that accounted for approximately 62% of AstraZeneca’s 2009 revenue will likely face competition from lower-priced generics by 2014, including the cholesterol-lowering drug Crestor and the proton pump inhibitor Nexium (esomeprazole). The total U.S. sales for Crestor and Nexium topped $8 billion in 2010, with Nexium accounting for 15% of the company’s total sales.1-5
Zyprexa and the Lilly Product Line
Zyprexa (olanzapine), like Seroquel, is an atypical antipsychotic introduced in 1996 and used to treat the symptoms of schizophrenia and bipolar disorder. Typically, therapy with this drug costs up to $641 per month. Since 2003, Zyprexa consistently earned revenues of over $4 billion, and in 2010 revenues were over $2.5 billion in the U.S. alone, or nearly 22% of Lilly’s annual sales.1-5 But Zyprexa has generated controversy as well as revenues. In 2009, Lilly agreed to pay $1.415 billion to settle criminal and civil allegations for the off-label promotion of Zyprexa to treat dementia. Also in 2005 and 2007, Lilly settled lawsuits for over $500 million with patients over claims that Lilly withheld information about the drug’s link to hyperglycemia. A generic version of Zyprexa was produced in the U.S. in 2011. Other Lilly products facing patent expiration over the next several years include Cymbalta (duloxetine), Evista (raloxifene), and Strattera (atomoxetine), drugs totaling over $3.5 billion in total sales in 2010. 1-5
Plavix and the Bristol-Myers Squibb
and Sanofi-Aventis Product Lines
Plavix (clopidogrel) has been the most prescribed antiplatelet brand-name drug for the prevention of heart attack or stroke, and was the second best-selling drug in the world, with $9.4 billion in global sales in 2010. This drug has been marketed jointly by Sanofi-Aventis and Bristol-Myers Squibb, and the cost of therapy averages about $162 per month. In 2010, Bristol recorded $6.7 billion in Plavix sales, or 34% of its total revenues of $19.5 billion. Sanofi recorded sales of nearly $3 billion, or 7% of its 2010 revenues. In 2006, generic drug-maker Apotex briefly marketed generic Plavix, but was ordered to stop until November 2011. The order was extended by the FDA, and Plavix’s patent expired in May, 2012.
Bristol-Myers Squibb is also facing 2012 patent expiration for its antihypertensive medication Avapro, with global sales in 2010 of $1.2 billion; its deal to market Abilify (aripiprazole), a drug used for schizophrenia and bipolar disorder with sales of $2.6 billion worldwide in 2010, is also due to end in 2012. Their patent on Reyataz (atazanavir), which generated over $400 million in sales in 2010, will also expire in the near future. Sanofi has already lost patent protection in the U.S. on its top seller, Lovenox (enoxaparin), a low-molecular-weight heparin. In 2010, the company said generic competition caused the loss of more than e 2 billion (U.S. $2.6 billion) in sales. Sandoz, the generic pharmaceutical division of Novartis, is already marketing generic enoxaparin, and other generic manufacturers are also looking to launch their own formulations of this drug. 1-5
Levaquin, Concerta, and the Johnson & Johnson
(Janssen Pharmaceuticals) Product Line
Johnson & Johnson had patents expire for the antipsychotic Risperdal (risperidone) in 2008 and the antiepileptic drug Topamax (topiramate) in 2009. Now it will lose patent protection on the widely used antibacterial Levaquin and on Concerta (sustained-release formulation of methylphenidate). Concerta is used to control symptoms of attention-deficit/hyperactivity disorder (ADHD) and narcolepsy and costs up to $236 per month. Levaquin is an antibiotic used to treat pneumonia as well as infections of the sinuses, urinary tract, kidneys and skin. In 2010 Levaquin and Concerta had sales of $1.4 billion and $1.3 billion worldwide, respectively, together comprising 12% of the company's $22.4 billion in pharmaceutical revenues and 4% of its total $61.6 billion in revenues. Hi-Tech Pharmacal Company. Inc. will inherit the exclusive rights to sell a generic version of the Levaquin solution for 6 months after the patent expiration date.1-5
Singulair and the Merck Product Line
Merck has already had to absorb lost revenue from several major products due to patent expiration or product withdrawal, including Vioxx (rofecoxib, withdrawn in 2004), Proscar (finasteride, 2005), Fosamax (alendronate, 2008), and, in the past two years, Cozaar and Zocor (simvastatin).1-5 Following the loss last year of patent protection for Merck’s cholesterol-lowering drug Zocor, sales this year are expected to fall 82% from $4.38 billion in 2005. Now the patents on two more of its top sellers—Fosamax and Singulair—will expire in the near future. Combined, Cozaar, Fosamax, and Singulair represented 44% of the company’s annual revenue.1-5 Singulair, a leukotriene blocker used as an oral asthma and allergy treatment, was first approved by the FDA in 1998. Its growth has been consistent despite the FDA adding warnings to its label about side effects including depression and increased suicidal thoughts. In 2010, worldwide sales for Singulair were $5 billion ($3.3 billion in the U.S.), a 7% increase, and nearly 11% of Merck’s total revenue. Now its patent is due to expire in 2012 and a generic version could be available as early as August of this year. Merck is also facing patent expirations in the next several years for its cholesterol-lowering drugs Vytorin (ezetimibe/simvastatin) and Zetia (ezetimibe), drugs with total sales exceeding $2 billion in 2010.1-5
Lexapro and the Forest Laboratories Product Line
Forest Laboratories’ biggest product, the antidepressant Lexapro, currently accounts for approximately 59% of company sales. In 2010 the U.S. sales for this drug approached $2.5 billion, but sales have already been declining as antidepressants from other companies have come off patent and generics have taken their place. Forest’s patent on Lexapro expired in early 2012 and generic products are already available. In the near future Forest’s patent on the Alzheimer’s drug Namenda (memantine) will also expire, a drug that earned over $700 million in 2010.1-5
Actos and the Takeda Product Line
The patent on the type 2 diabetes medication from Japan’s Takeda, which was launched in 1999, expired in January of this year. In 2010 Actos recorded sales of $3.58 billion (nearly $3 billion in the U.S.), or 27% of the company's total revenue. The branded drug costs patients up to $241 per month. But Takeda entered into an agreement with generic manufacturers Ranbaxy, Watson, and Mylan under which they won’t start marketing Actos until August 2012. Sandoz, Teva Pharmaceutical Industries, and several others will enter the market 180 days later. Takeda’s drug sales already were affected by a sharp decrease in Prevacid (lansoprazole) sales due to the loss of exclusivity in the U.S.1- 5
The GlaxoSmithKline Product Line
In the next several years GSK will lose patent protection on its antitriglyceride product Lovaza (omega-3-acid esters), the benign prostatic hyperplasia drug Avodart (dutasteride), and the HIV/AIDs product Combivir. These drugs combined for over $1 billion in earnings in the U.S. alone in 2010, with Lovaza accounting for the majority of the revenue. A generic form of Combivir is already available in the U.S. 1-5
Enbrel (Amgen) and the Problem of
“Biological” and “Biosimilar” Products
Enbrel (etanercept), a large biological
molecule, is used to treat the autoimmune diseases rheumatoid arthritis
and psoriasis. Therapy with this drug, which belongs to a class of
medications called tumor necrosis factor (TNF)-alpha inhibitors, costs
up to $2,784 per month. In 2010, Enbrel earned Amgen more than $3.3
billion in U.S. sales, or over 23% of the company's total revenue.1-5 Also Pfizer, which markets Enbrel outside the U.S., in Canada and Japan, earned $3.2 billion from drug sales. The patent on
Enbrel
expires in August 2012; however, since it is a biological agent it is
subject to different laws with respect to generic manufacture. Under
current law, most biologics are licensed for marketing by the FDA under
the Public Health Service Act. By contrast, so-called small-molecule
drugs—the primary focus of this article—are approved for marketing under
the Federal Food, Drug, and Cosmetic Act. The 1984 landmark
Hatch-Waxman Act created an abbreviated pathway for approval, which
allowed generic versions of brand small-molecule drugs to be approved
without clinical studies. All a generic manufacturer had to demonstrate
to gain FDA approval was that its product was bioequivalent to the
approved brand drug already deemed to be safe and effective. Currently
there is no such pathway available under the Public Health Service Act
for approval of generic biological drugs or so-called biosimilar
products (also known as follow-on biologics or FOBs). Several
proposals to create such a process, however, are under consideration by
Congress as part of patent reform legislation. 1-5
Conclusion
With the arrival of the patent cliff, it is estimated that generic competition will erode more than $60 billion from top drug companies’ annual U.S. sales between 2007 and 2012, and potentially hundreds of billions in the near future.1-5 Pharmaceutical companies have explored and continue to investigate a variety of strategies to minimize the impact of this revenue loss and maintain their profitability. Most obviously, there has been continued investment in new drug discovery and development of a “pipeline” of promising agents that address unmet medical needs and are profitable. However, it appears that there will be relatively few drugs with the profit potential of Lipitor, Nexium, and Plavix emerging in the immediate future. In the meantime, companies have raised drug prices (as much as 60% since 2002) and enhanced advertising over the past decade in an attempt to optimize earnings prior to patent expiration. The pharmaceutical industry has also invested hundreds of millions of dollars to lobby Congress on a variety of issues ranging from regulating Medicare drug prices to keeping lower-priced foreign drugs from being imported to protecting lucrative drug patents.1-5
Other approaches aimed at preserving the future profitability of drug companies have involved structural reorganization and downsizing. Overall, it is estimated that as many as 50,000 positions in the pharmaceutical industry will be displaced over the next decade. A number of drug companies have suggested that they will outsource at least some of their manufacturing to places such as Eastern Europe, India, and China, where drug products can be made at significantly less cost. And in the future, outsourcing is expected to extend to drug research and development efforts as well.1-5
There have been and likely will continue to be mergers and acquisitions that will change the face and possibly the direction of the pharmaceutical industry. Some pharmaceutical companies already are entering into agreements with generic manufacturers, licensing them the right to sell “authorized generics” identical to branded drugs that have gone or will go off patent. Others have established their own generic manufacturing companies or subsidiaries. And many pharmaceutical companies have begun to diversify through new business development or acquisition. Two major areas in this category to date include biotechnology, or biologics, and diagnostics. As mentioned above, biologics are especially appealing because no regulatory pathway yet exists in the U.S. for bringing to market generic “biosimilar” drugs. In the end, the financial future of many pharmaceutical companies will depend on how they manage the transition of many of their brand drugs to the generic market, as well as on their own efforts to develop novel therapeutics and diagnostics with significant profit potential, and to adapt to the changing global business climate.1-5
Generics are already available for a number of drugs, listed in TABLE 1, including Boniva (ibandronate), Caduet, Combivir, Geodon, Lexapro, Lipitor, Seroquel, and Zyprexa.6 When generics will emerge for other brands will depend on a host of factors including the ability of generic manufacturers to gain approval and successfully market the products; cooperative agreements that may develop between brand and generic manufacturers; and litigation to stop or delay generic development by the current patent holders. However, while the generic drug industry will certainly benefit in the short term from the patent cliff, it too will see a slowdown in growth after 2015 as fewer high-revenue brand drugs will experience patent expiration.
REFERENCES
1. Method K. Going, going, gone: patents set to expire soon on many brand-name drugs. Drug Topics Supplements; August 2009. http://drugtopics.modernmedicine.com/drugtopics/Modern+Medicine+Now/Patents-soon-to-expire-for-many-brand-name-drugs/ArticleStandard/Article/detail/617015. Accessed May 17, 2012.
2. Wilson D. Drug firms face billions in losses in ’11 as patents end. New York Times; March 6, 2011:A1. www.nytimes.com/2011/03/07/business/07drug.html?pagewanted=all. Accessed May 17, 2012.
3. Alazraki M. The 10 biggest-selling drugs that are about to lose their patent. DailyFinance; February 27, 2011. www.dailyfinance.com/2011/02/27/top-selling-drugs-are-about-to-lose-patent-protection-ready/. Accessed May 17, 2012.
4. Johnson LA. Prescription-drug prices to plunge as patents expire. Seattle Times; July 24, 2011. http://seattletimes.nwsource.com/html/health/2015720110_generics25.html. Accessed May 17, 2012.
5. Krishnan A. Drug patents expiration in 2011 & 2012—a bumpy ride ahead for big pharma as big drugs lose patent protection. IHS Healthcare Practice; October 28, 2011. http://healthcare.blogs.ihs.com/2011/10/28/drug-patents-expiration-2011-2012-a-bumpy-ride-ahead-for-big-pharma-as-big-drugs-lose-patent-protection. Accessed May 17, 2012.
6. PL Detail-Document. Anticipated availability of first-time generics. Pharmacist’s Letter/Prescriber’s Letter. April 2012.
To comment on this article, contact rdavidson@uspharmacist.com






