US Pharm. 2013;38(6):68-71.
ABSTRACT: Impetigo is a common skin infection in children between the ages of 2 and 5 years old. It is highly infectious and is caused primarily by gram-positive bacteria, mainly Staphylococcus aureus and group A beta-hemolytic streptococcus. The lesions are characteristic, appearing as bullae that dry to form a honey-colored, thick crust that may cause pruritus. Most impetigo infections resolve without requiring antibiotics; however, to reduce the duration and spread of the disease, topical antibiotics are used. Oral antibiotics are generally reserved for patients with more severe or treatment-refractory infection and include penicillinase-resistant penicillins or first-generation cephalosporins.
Impetigo contagiosa, also referred to as pyoderma, is a highly communicable superficial skin infection that commonly occurs worldwide in children between ages 2 and 5 years.1 About 10% of pediatric patients presenting to U.S. physicians with dermatologic complaints are diagnosed with this infection. During the summer months, the incidence of impetigo increases primarily due to higher humidity and temperature.
The infection is highly contagious, spreading through direct skin contact. It can be further spread via inoculation to oneself or others upon initial infection.2 Although skin-to-skin contact with infected children is the most common mode of transmission of impetigo, fomites also play a role in the spread of the disease.3 The infection consists of purulent blisters that usually occur on the face or limbs. There is no gender or racial predilection related to impetigo.4
Impetigo is caused by gram-positive bacteria, most commonly Staphylococcus aureus and Streptococcus pyogenes (group A beta-hemolytic streptococcus [GABHS]), either alone or in combination.4 The responsible microorganisms have been known to colonize the skin of uninfected individuals, and an infection may develop in areas where the skin is broken.4,5 The infection often spreads in day care centers, in schools, and among siblings.2,6,7 The prognosis is usually good, with healing occurring within 2 weeks if left untreated.2
Clinical Presentation
There are two main types of impetigo—bullous (characterized by bullae) and nonbullous. Bullae are fluid-filled lesions >0.5 cm.8 Impetigo may also be classified as a primary or secondary infection. The former refers to a direct invasion of bacteria to intact skin, and the latter refers to an underlying condition that disrupts the skin.2
Bullous impetigo almost exclusively results from an S aureus dermatologic infection that manifests when the stratum corneum layer of the skin is raised.1,2,9 This form presents as an erythroderma that is toxin mediated, resulting in the sloughing of the epidermal layer of the skin, ultimately leading to large areas of skin loss. Vesicles turn into bullae and fill with fluid, which later rupture to form a light-brown crust.1 The bullous form is the least common type of impetigo.
Nonbullous impetigo is the most common form, occurring in about 70% of cases.2,9 It indicates a host response to the infection. Methicillin-resistant S aureus (MRSA) impetigo is most commonly associated with the nonbullous form.2 Although the lesions are not considered to be bullae, the fluid-filled vesicles may appear transiently and are <0.5 cm in diameter. The infection initially appears as red maculae/papulae on the surface of the skin. Impetigo typically presents in the face, but may be transmitted wherever skin is exposed. After 4 to 6 days, the maculae turn into painful pustules or blisters that rupture and dry into a thick, golden-yellow crust that is often pruritic.1 The impetigo affects areas of the skin (usually on the face and extremities) that have been associated with trauma such as mosquito bites and scratches.
The skin infection resolves within 2 weeks when left untreated, and often heals without scarring. Diagnosis is usually made through visual observation; however, a culture may be indicated when a patient has not responded to therapy, which may be suggestive of a MRSA infection. Systemic manifestations of impetigo are minimal and include weakness, fever, and diarrhea.1,2
Pathophysiology
Because skin is a host mechanism of defense, unbroken skin is generally unaffected by coagulase-negative staphylococci (CoNS) and GABHS colonization.10 Teichoic acid mediates adhesion in the impetigo-causing microorganisms, but it requires fibronectin in order to colonize the skin. Fibronectin, a cell adhesion molecule that is not available on the surface of intact skin, allows for bacterial cells to attach to collagen and invade disrupted skin surfaces. (TABLE 1 lists various conditions that may predispose children to impetigo). Certain factors modify the skin’s normal flora and enable transient colonization by GABHS and S aureus. These include humidity or high temperature, preexisting skin lesions, recent antibiotic treatment, and young age.10

Colonization with GABHS occurs when infected individuals are in close contact with others. The normal flora of the skin becomes disrupted, allowing for the development of impetigo once the epidermis becomes affected with minor abrasions or insect bites. Often-times, patients who developed impetigo caused by streptococcus may have detectable GABHS in their nasal passages and throat up to 3 weeks after the development of lesions. These patients will not have symptoms of streptococcal pharyngitis since the strain of bacterium that causes impetigo (pattern D) is different from the strains causing pharyngitis (patterns A, B, and C).11
Up to one-third of individuals are colonized with S aureus in the nostrils, and some of these patients experience recurrent impetigo around the lips and nose. Most healthy individuals only transiently host S aureus as part of their normal skin flora. In general, the bacteria are spread via direct skin contact and enter the surface of the skin through a broken site.11
Treatment
Treatment goals for impetigo are three-fold: 1) to relieve discomfort, 2) to improve the appearance of the skin, and 3) to reduce further spread of the infection.2 Currently, there is no accepted standard of therapy, and treatment options include a variety of oral and topical antibiotics as well as topical disinfectants.12 Most impetigo infections resolve without requiring antibiotics; however, to reduce the duration and spread of the disease, topical antibiotics are used. Oral antibiotics are generally reserved for patients with more severe or treatment-refractory infection (TABLES 2 and 3).13,14

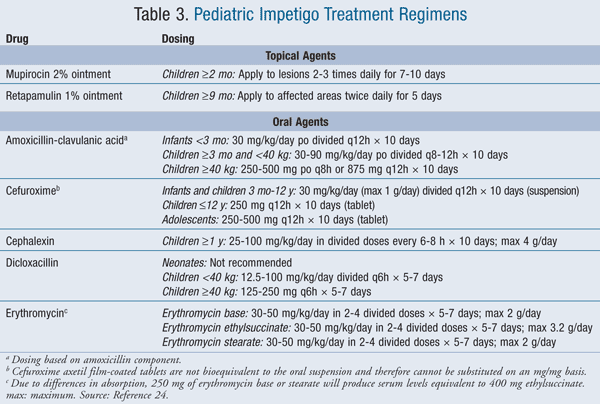
Neonates: Neonates presenting with impetigo should be referred to either an infectious diseases specialist or a pediatric dermatologist. Neonates require oral antibiotics, with erythromycin being the drug of choice for nonbullous impetigo when MRSA is excluded.12 IV vancomycin should be used if MRSA is determined to be the causative agent.12,15 Neonatal patients with the bullous form of impetigo can be treated with nafcillin, oxacillin, or clindamycin. Vancomycin is recommended for MRSA cases.12,15
Infants, Children, and Adolescents: In infants, children, and adolescents with limited infection (i.e., no deep tissue involvement or widespread symptoms), impetigo should be treated with mupirocin or retapamulin topically unless there is resistance.12 Unlike mupirocin, retapamulin should not be used on the nasal mucosa due to the risk of epistaxis.16 For patients with widespread lesions, oral antibiotics such as dicloxacillin and first-generation cephalosporins are the preferred agents. For those with severe immune compromise, parenteral antibiotics should be considered since these patients are at higher risk for developing a complicated course of infection.12,15,17 If MRSA is suspected or confirmed by susceptibility tests, the preferred antibiotic treatment options include clindamycin, trimethoprim-sulfamethoxazole (TMP-SMZ), or doxycycline.18
A Cochrane review of impetigo treatment options found that differences in cure rates were not significant between topical and oral antibiotics.12 In addition, side effects are more common with oral treatments compared to topical antibiotics. However, topical mupirocin was shown to be slightly superior to oral erythromycin. Penicillin was shown to be inferior to both erythromycin and cloxacillin. The study found that there is no evidence supporting the use of disinfectant solutions, as topical antibiotics showed significantly better results. Because of the lack of studies in patients with extensive impetigo, it remains unclear whether oral antibiotics are superior to topical treatments.12
Since S aureus is the most likely cause of bullous pyoderma and a large portion of nonbullous infections, A-1 recommendations from the Infectious Diseases Society of America (IDSA) include penicillinase-resistant penicillins or first-generation cephalosporins.13,19 Oral erythromycin has been a mainstay in the treatment of this infection, but its use may not be warranted in areas with erythromycin-resistant strains of both S aureus and S pyogenes. Topical mupirocin therapy is as effective as systemic antibiotic therapy and may be used when there are a limited number of lesions.4,20,21 Certain strains of staphylococci have developed resistance to mupirocin, and the high cost of the ointment limit its use.4,22
It is important that proper technique be followed when applying topical ointments to the impetigo lesions. The following steps outline the proper administration technique:
1. Caregivers should wash hands thoroughly with warm water and antibacterial soap.
2. Using a clean washcloth soaked in warm soapy water, gently rub the crusty lesions to loosen the debris, thus allowing for adequate antibiotic penetration. In order to prevent further infection, take care to avoid scrubbing the lesions. Some slight bleeding may occur.
3. Apply a thin layer of the prescribed ointment to each sore, including the surrounding area. When finished, wash hands well and, if necessary, apply a piece of gauze to the infected area.
Complications
A rare complication of impetigo is poststreptococcal glomerulonephritis (PSGN). Characterized by kidney inflammation, PSGN develops after streptococcal infection and occurs when antibodies form due to glomerular damage. It results in the appearance of symptoms approximately 2 weeks after infection. It is unclear whether treatment with antibiotics reduces the risk of PSGN. Symptoms include swelling in the face, especially around the eyes, oliguria, hematuria, and increased blood pressure. Most patients are able to recover from this without permanent damage, although PSGN can lead to chronic kidney disease (CKD). CKD is treated with sodium and fluid restriction and diuretics, as the major goal of therapy is to control blood pressure and edema.16,23
Prevention of Impetigo
Proper hygiene, including washing hands vigorously with antibacterial soap and water and taking regular baths, may be helpful in the prevention of impetigo. Patients with impetigo should be instructed to use clean washcloths and towels every time and to avoid sharing any personal care products such as clothing and towels. Children with the infection should not attend school or playgroups until 48 hours after starting treatment or until the sores have blistered and dried up. Children should be urged to wash their hands after touching the skin lesions, and their fingernails should be clipped short and filed to prevent infection through scratching. All minor cuts and scrapes should be cleaned with antibacterial soap and warm water and covered to prevent infection. In addition, toys should be washed or wiped down with a disinfectant wipe to avoid contamination of fomites.
Summary
Impetigo is a common skin infection caused by gram-positive bacteria that most often affects children between the ages of 2 and 5 years. Diagnosis is usually made with visual observation due to the characteristic appearance of the infection. Limited infection is treated with a topical antibiotic cream (mupirocin or retapamulin), and more extensive cases may be treated with oral or IV antibiotics. The vast majority of children with impetigo recover completely with no complications. Patients should be advised to follow-up with their physician if: 1) lesions do not begin to heal following the complete course of antibiotic therapy, 2) lesions continue to spread, or 3) the patient develops systemic symptoms such as fever.
REFERENCES
1. Fish DN. Chapter 42. Skin and soft tissue infections. In: Wofford MR, Posey LM, Linn WD, O’Keefe ME, eds. Pharmacotherapy in Primary Care. New York, NY: McGraw-Hill; 2009.
2. Cole C, Gazewood J. Diagnosis and treatment of impetigo. Am Fam Physician. 2007;75:859-864.
3. Shi D, Higuchi W, Takano T, Saito K, et al. Bullous impetigo in children infected with methicillin-resistant Staphylococcus aureus alone or in combination with methicillin-susceptible S. aureus: analysis of genetic characteristics, including assessment of exfoliative toxin gene carriage. J Clin Microbiol. 2011;49:1972-1974.
4. Liu C, Bayer A, Cosgrove SE, et al. Clinical practice
guidelines by the Infectious Diseases Society of America for the
treatment of methicillin-resistant Staphylococcus aureus infections in adults and children: executive summary. Clin Infect Dis. 2011;52:285-292.
5. Ferrieri P, Dajani AS, Wannamaker LW, Chapman S.
Natural history of impetigo. 1. Site sequence of acquisition and
familial patterns of spread of cutaneous streptococci. J Clin Invest. 1972;51:2851-2862.
6. Brown J, Shriner DL, Schwartz RA, Janniger CK. Impetigo: an update. Int J Dermatol. 2003;42:251-255.
7. Sladden MJ, Johnston GA. Common skin infections in children. BMJ. 2004;329:95-99.
8. Hirschmann JV. Impetigo: etiology and therapy. Curr Clin Top Infect Dis. 2002;22:42-51.
9. George A, Rubin G. A systematic review and
meta-analysis of treatments for impetigo. Br J Gen Pract. 2003;53:480-487.
10. Roth RR, James WD. Microbial ecology of the skin. Annu Rev Microbiol. 1988;42:441-464.
11. Treating impetigo in primary care. Drug Ther Bull. 2007;45:2-4.
12. Koning S, van der Sande R, Verhagen AP, et al. Interventions for impetigo. Cochrane Database Syst Rev. 2012;(1):CD003261.
13. Dagan R, Bar-David Y. Double-blind study comparing
erythromycin and mupirocin for treatment of impetigo in children:
implications of a high prevalence of erythromycin-resistant Staphylococcus aureus strains. Antimicrob Agents Chemother. 1992;36:287-290.
14. Demidovich CW, Wittler RR, Ruff ME, et al. Impetigo:
current etiology and comparison of penicillin, erythromycin, and
cephalexin therapies. Am J Dis Child. 1990;144:1313-1315.
15. Wallin TR, Hern HG, Frazee BW. Community-associated methicillin-resistant Staphylococcus aureus. Emerg Med Clin North Am. 2008;26;431-455.
16. Altabax (retapamulin) ointment. FDA MedWatch.
September 2010.
www.fda.gov/Safety/MedWatch/SafetyInformation/ucm229270.htm. Accessed
April 13, 2013.
17. Moran GJ, Amii RN, Abrahamian FM, Talan DA. Methicillin-resistant Staphylococcus aureus in community-acquired skin infections. Emerg Infect Dis. 2005;11:928-930.
18. Noguchi N, Nishijima S, Kurokawa I, et al.
Antimicrobial agent of susceptibilities and antiseptic resistance gene
distribution among methicillin-resistant Staphylococcus aureus isolates from patients with impetigo and staphylococcal scalded skin syndrome. J Clin Microbiol. 2006;44:2119-2125.
19. Stevens DL, Bisno AL, Chambers HF, et al. Practice
guidelines for the diagnosis and management of skin and soft-tissue
infections. Clin Infect Dis. 2005;41:1373-1406.
20. Britton JW, Fajardo JE, Krafte-Jacobs B. Comparison of mupirocin and erythromycin in the treatment of impetigo. J Pediatr. 1990;117:827-829.
21. Yun HJ, Lee SW, Yoon GM, et al. Prevalence and
mechanisms of low- and high-level mupirocin resistance in staphylococci
isolated from a Korean hospital. J Antimicrob Chemother. 2003;51:619-623.
22. Rodriguez-Iturbe B, Musser JM. The current state of poststreptococcal glomerulonephritis. J Am Soc Nephrol. 2008;19:1855-1864.
23. Ahn SY, Ingulli E. Acute poststreptococcal glomerulonephritis: an update. Curr Opin Pediatr. 2008;20:157-162.
24. Impetigo. Lexi-Drugs Online. Hudson, OH: Lexi-Comp, Inc; 2013. http://online.lexi.com. Accessed April 13, 2013.
To comment on this article, contact rdavidson@uspharmacist.com.





