US Pharm.
2008;33(8):54-57.
The
American Diabetes Association (ADA) defines diabetes mellitus as a group of
metabolic diseases characterized by hyperglycemia resulting from defects in
insulin secretion, insulin action, or both. The chronic hyperglycemia of
diabetes is associated with long-term damage, dysfunction, and failure of
various organs, especially the eyes, kidneys, nerves, heart, and blood vessels.1
The ADA estimates that 20.8 million children and adults are diagnosed with
diabetes mellitus, 176,500 of whom are younger than age 20.2
Previously known as juvenile diabetes and insulin-dependent diabetes,
type 1 diabetes mellitus (T1DM) is the most common chronic disease of
childhood and is often diagnosed in children presenting with symptomatic
hyperglycemia.3 Although most attention is focused on type 2
diabetes mellitus (T2DM), the incidence of T1DM is rising worldwide.4
Type 2 diabetes mellitus results from insulin resistance combined with
relative insulin deficiency, while T1DM is primarily the result of insulin
deficiency. Therefore, the focus of pharmacologic treatment in T1DM is
exogenous insulin replacement.
Pathophysiology and
Clinical Manifestations
Pancreatic beta
cells are responsible for the production of insulin, which regulates the
amount of glucose in the body. Insulin regulates glucose metabolism by
peripherally stimulating its uptake and inhibiting its production in the
liver. During the fed state, a bolus of insulin is secreted from the pancreas,
preventing hyperglycemia. Unrelated to food intake, a continuous basal
secretion of insulin is also constantly released. Type 1 diabetes mellitus is
a complex and multifactorial disease characterized by autoimmune-mediated
destruction of pancreatic beta cells. Although genetic defects play a role in
insulin production, autoantibodies against islet cells, insulin, glutamic acid
decarboxylase, and tyrosine phosphatase 1A-2 and 1A-2 beta can be found in
most cases of T1DM. Severe insulin deficiency results in requiring
administration of exogenous insulin drug therapy. Unlike T2DM, several options
are available to cure T1DM, such as islet transplantation along with
immunosuppressive therapy or insulin gene therapy.
The clinical presentation of T1DM can
manifest as classic new onset diabetes, silent diabetes, and diabetic
ketoacidosis (DKA). Classic new onset diabetes is usually diagnosed in the
outpatient setting and presents with symptoms of marked hyperglycemia, such as
polydipsia, polyuria, polyphagia, weight loss, and lethargy. Silent diabetes
is often diagnosed in children undergoing testing for T1DM due to a family
history of the disease. Approximately 30% of children with T1DM present to the
hospital with DKA.5 In addition to the signs and symptoms of
classic new onset diabetes, patients with DKA can also present with
dehydration, vomiting, mental status changes, and reduced arterial pH (i.e.,
acidosis) and serum bicarbonate.
Goals of Therapy
The goals of
therapy in the management of T1DM include achieving near normal blood
glucose and glycated hemoglobin (A1C); preventing DKA and hypoglycemia;
maintaining normal quality of life; understanding carbohydrate insulin dosing;
and preventing acute and chronic diabetes complications.
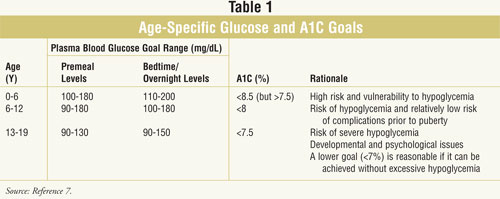
In 2005, the first guidelines
for the management of T1DM in children and adolescents were established. The
ADA recommendations for glycemic control in T1DM are less stringent in
pediatrics than in adults (TABLE 1). Children younger than 6 years are
more likely to develop hypoglycemia; therefore, the thresholds for premeal and
overnight blood glucose levels are higher in children.6 Although
the ideal AlC target is less than 7%, most children and adolescents do not
achieve this goal. As a result, the ADA recommends an A1C test result of less
than 7.5% to prevent the risk of hypoglycemia and the feeling of failure in
patients and families.
When administering drug therapy in
children and adolescents with T1DM, a clear understanding of the physiologic
and pharmacotherapeutic differences between children and adults is necessary
to provide optimal therapy. For example, children require dosage adjustments
as they gain weight, may require family members or legal guardians to
administer drug therapy, may have periods of unpredictable food intake or
varying levels of physical activity, and may not have the cognitive capacity
to recognize hypoglycemic episodes.
Insulin Agents
Although potential
new drug therapies are in development for the treatment of T1DM, insulin
remains the standard of care. All patients who lack insulin production from
pancreatic beta islet cells will require lifelong exogenous insulin
replacement. Commercial sources of insulin include purified pork pancreas and,
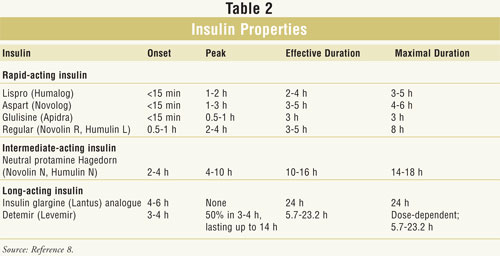
more commonly, human recombinant DNA technology. Insulins are categorized as
rapid-acting (insulin glulisine, lispro, and aspart), short-acting (regular),
intermediate-acting (neutral protamine Hagedorn [NPH]), and long-acting
(glargine and detemir). The currently available types of insulin based
on onset of action, peak, and effective and maximal duration of action are
summarized in TABLE 2. Although premixed combination products are
available to simplify regimens, these agents require children to commit to
fixed dosages, making it problematic in situations of unpredictable
carbohydrate intake, exercise, or changes in weight and blood glucose.
Rapid-acting Insulin Analogue: With
the emergence of rapid-acting insulin analogues, the use of regular insulin in
treatment regimens is becoming less favorable. In comparison to regular
insulin, rapid-acting insulin analogues improve pediatric lifestyles by
offering a faster onset, a peak effect independent of dosage, and the
flexibility to be administered immediately prior to meals or 15 minutes after
meals since most children may not always plan their meals.9,10
Although no significant hypoglycemic differences were noted between
rapid-acting and regular insulin, the Cochrane Metabolic and Endocrine
Disorders Group did report a significant reduction in A1C with rapid-acting
analogues in T1DM.11,12
Long-acting Insulin
Analogues: Because
the administration of intermediate-acting insulin with concomitant rapid or
short-acting insulin achieves goal A1C in only 50% to 70% of T1DM patients,
the use of long-acting insulin analogues is recommended.13
Long-acting insulin imitates the constant, consistent basal release of insulin
from the pancreas. Insulin glargine was the first FDA-approved long-acting
analogue followed by detemir. Insulin glargine is manufactured as an acidic
solution forming microprecipitants after administration, thus producing a
peakless drug effect and allowing once-daily subcutaneous injections. Although
insulin glargine is currently approved for children older than 6 years,
pediatric studies in children as young as 2 years have shown efficacy with
minimal hypoglycemic episodes.14 Unlike insulin glargine, detemir
is approximately 98% reversibly bound to albumin, producing slow absorption
and prolonged action. In comparison to intermediate-acting agents, insulin
detemir has been shown to produce less nighttime and asymptomatic hypoglycemia
in children and adolescents.15,16 Major adverse effects reported
with the use of detemir include serious allergic reactions manifesting as
rash, shortness of breath, tachycardia, and sweating.
Insulin Regimens: Both
regular and rapid-acting insulins are administered preprandially to mimic the
increased production of endogenous insulin associated with food intake, while
the intermediate and long-acting insulins maintain constant basal levels of
insulin. At the initiation of drug therapy, the recommended total daily dose
(TDD) of insulin is 0.5 to 1 units/kg/day, with 50% of the dosage dedicated to
basal insulin and the rest for rapid (prior to or with meals) or short-acting
insulin (30 minutes prior to meals). Due to hormonal changes, pubertal
adolescence usually requires higher doses of 1 to 1.5 units/kg/day. A number
of studies have attempted to prove that regimens with rapid- and long-acting
agents are more effective than regimens with short- and intermediate-acting
agents in reducing A1C and preventing hypoglycemic events in children with
T1DM. However, the results remain inconclusive and the issue continues to be
controversial. Adverse effects to monitor in T1DM patients receiving insulin
include hypoglycemia, lipodystrophy, injection site reactions, muscle
weakness, weight gain, and fatigue.
Some common initial insulin
regimens for T1DM include
1) Intermediate-acting PLUS regular insulin given twice a day
2) Glargine plus rapid-acting insulins given with meals
3) Detemir given once or twice a day
plus rapid-acting insulins given with meals.
Besides maintaining normal
serum glucose levels, preventing complications from T1DM is another goal of
therapy. As a result, the Diabetes Control and Complication Trial (DCCT)
evaluated the risk of long-term diabetic complications such as retinopathy,
nephropathy, and neuropathy in children with T1DM receiving intensive insulin
regimens.17,18 The trial defined intensive insulin regimen as
targeting premeal blood glucose 70 to 120 mg/mL, postprandial blood glucose
less than 180 mg/mL, and an A1C less than 6.05%. Results from the DCCT
suggested that intensified regimens could reduce the risk of T1DM-associated
complications; however, there was a significantly higher risk for serious
hypoglycemic events. The Epidemiology of Diabetes Interventions and
Complications study, a follow-up to the DCCT, also concluded that intensive
treatment reduced the risk of cardiovascular events.19,20 Although
intensive insulin regimens appear more favorable in reducing long-term
complications, the risk of hypoglycemic events minimizes their utility in
pediatric patients. As a result, the ADA recommends higher thresholds for
targets of blood glucose and A1C (TABLE 1).
Adjunctive Therapy
A number of
patients with T1DM cannot maintain target blood glucose levels with currently
available insulin regimens. As a result, other pharmacologic treatment options
can be considered. In 2005, the FDA approved pramlintide (Symlin) for
adjunctive therapy for type 1 and 2 diabetes patients who failed to achieve
target blood glucose levels with prandial insulin therapy.21 As a
synthetic analogue of human amylin, pramlintide works by slowing carbohydrate
absorption by delaying gastric emptying time, reducing postprandial glucagon
secretion, and decreasing caloric intake via appetite suppression. In adults
with T1DM, pramlintide is dosed at 15 mcg subcutaneously immediately prior to
meals. When no significant nausea is seen for at least three days, the dose
may be increased by 15-mcg increments up to 30 or 60 mcg as tolerated.
Additionally, the patient's concomitant preprandial rapid-acting or regular
insulin dose is reduced by 50%. Although this medication is not
currently approved in pediatrics, Heptulla and colleagues evaluated the
efficacy and safety of pramlintide 30 to 45 mcg plus insulin boluses in
children age 12 to 18 with T1DM.22 Results revealed that
pramlintide significantly suppressed glucagon production, and reduced
postprandial hyperglycemia and A1C by a mean of 0.67%. As a result,
pramlintide appeared to be beneficial for postprandial glycemic control in
adolescents. Like other medications, pramlintide is not without adverse
effects. A major adverse effect associated with pramlintide, stated as a black
box warning, is severe hypoglycemia occurring three hours after injection.
Other side effects include nausea, vomiting, anorexia, and injection site pain.
Insulin Pump Therapy
First identified in
the late 1970s, insulin pump therapy usage has been increasing and is becoming
more common in the pediatric population.23 Insulin pump, also known
as continuous subcutaneous insulin infusion (CSII), is a computerized,
battery-driven portable device that administers preprogrammed basal rates and
bolus doses of insulin. Candidates for insulin pump therapy include patients
who are interested or who have the following24:
• More than the
equivalent of four injections per day
• Greater than four blood glucose tests per day
• Failure to achieve A1C goals
• Recurrent hypoglycemic events
• Changing eating and lifestyle habits
• Recurrent DKA.
Several advantages of CSII in
T1DM include the use of rapid-acting insulin analogues, lifestyle flexibility
due to fewer injections, reduction of hypoglycemic events, and significant
reduction in A1C compared to multiple daily insulin injections.25,26
Disadvantages of CSII include pump failure, device complexity, cost, and user
self-consciousness.
Conclusion
Although T1DM management in
pediatrics can appear overwhelming, pharmacists need to consider
pediatric-specific blood glucose goals, drug selection, and dosing regimens
compared to adults. Insulin therapy remains the standard of care for the
treatment of T1DM. Goals of therapy should include obtaining target blood
glucose and A1C levels based on the ADA recommendations, maintaining normal
quality of life, and preventing acute and chronic complications. Pharmacists
can play a major role in optimizing insulin drug regimens, selecting the most
appropriate insulin, and counseling for adverse effects. Future studies are
necessary to determine whether noninsulin therapies are effective for the
treatment of T1DM. Insulin pump therapy is becoming more favorable.
REFERENCES
1. American Diabetes Association. Diagnosis and classification of diabetes mellitus. Diabetes Care. 2008;31:S55-S60.
2. American Diabetes Association. Total prevalence of diabetes and pre-diabetes. http://diabetes.org/diabetes-statistics/prevalence.jsp. Accessed January 24, 2008.
3. Haller MJ, Atkinson MA, Schatz D. Type 1 diabetes mellitus: etiology, presentation, and management. Pediatr Clin North Am. 2005;52:1553-1578.
4. Center for Disease Control.
Diabetes projects: children and diabetes. www.cdc.gov/
diabetes/projects/diab_children.htm.
Accessed February 1, 2008.
5. Scibilia J, Finegold D, Dorman J. Why do children with diabetes die? Acta Endocrinol Suppl. 1986;279:326-333.
6. Mortensen HB, Hougaard P. Comparison of metabolic control in a cross-sectional study of 2,873 children and adolescents with IDDM from 18 countries. Diabetes Care. 1997;20:714-720.
7. Silverstein J, Klinggensmith G, Copeland K, et al. Care of children and adolescents with type 1 diabetes. Diabetes Care.2005;28:186-212.
8. National Diabetes Education Program. Diabetes medications supplement. National Institutes of Health and the Centers for Disease Control and Prevention. Pub. No. NDEP-54-S, March 2007. http://ndep.nih.gov/diabetes/pubs/Drug_tables_supplement.pdf.
9. Owens DR, Zinman B, Bolli GB. Insulins today and beyond. Lancet. 2001;358:729-746.
10. Reynolds NA and Wagstaff AJ. Insulin aspart: a review of its use in the management of type 1 or 2 diabetes mellitus. Drugs. 2004;64:1957-1967.
11. Siebenhofer A, Plank J, Berghold A, et al. Short acting insulin analogues versus regular insulin in patients with diabetes mellitus. Cochrane Database of Systemic Reviews. 2004;3.
12. Siebenhofer A, Plank J, Bergold A, et al. Meta-analysis of short-acting insulin analogues in adult patients with type 1 diabetes: continuous subcutaneous insulin infusion versus injection therapy. Diabetologia. 2004;47:1895-1905.
13. Danne T, Mortensen HB, Hougaard P, et al. Persistent differences among centers over 3 years in glycemic control and hypoglycemia in a study of 3,805 children and adolescents with type 1 diabetes from the Hvidore Study Group. Diabetes Care. 2001;24:1342-1347.
14. Alemzadeh R, Berhe T, Wyatt DT. Flexible insulin therapy with glargine insulin improved glycemic control and reduced severe hypoglycemia among preschool aged children with type 1 diabetes mellitus. Pediatrics. 2005;115:1320-1324.
15. Kordonouri O, Datz N, Hoeffe J, et al. Efficacy and safety of bedtime insulin detemir vs. insulin semilente in children, adolescents, and young adults with type 1 diabetes. A randomized, open-label, cross-over study [abstract]. Pediatr Diabetes. 2006;7(suppl 5):68-69.
16. Deiss D, Kordonouri O, Hartmann R, et al. Treatment with insulin glargine reduces asymptomatic hypoglycemia detected by continuous subcutaneous glucose monitoring in children and adolescents with type 1 diabetes. Pediatr Diabetes. 2007;8:157-162.
17. The DCCT Research Group. The effect of intensive diabetes treatment on the development and progression of long-term complications in adolescents with insulin-dependent diabetes mellitus. N Engl J Med. 1993;329:977-986.
18. The DCCT Research Group. The effect of intensive diabetes treatment on the development and progression of long-term complications in adolescents with insulin-dependent diabetes mellitus: the Diabetes Control and Complication Trial. J Pediatr. 1994;125:177-188.
19. DCCT/EDIC Research Group. Beneficial effects of intensive therapy of diabetes during adolescence: outcomes after the conclusion of the Diabetes Control and Complications Trial. J Pediatr. 2001;139:804-812.
20. The DCCT/EDIC Study Research Group. Intensive diabetes treatment and cardiovascular disease in patients with type 1 diabetes. N Engl J Med. 2005;353:2643-2653.
21. Symlin (pramlintide) package insert. San Diego, CA: Amylin Pharmaceuticals; 2007.
22. Heptulla RA, Rodriguez LM, Bomgaars, et al. The role of amylin and glucagon in the dampening of glycemic excursions in children with type 1 diabetes. Diabetes. 2005;54:1100-1107.
23. Ahern JA. Insulin pump therapy in pediatrics: a therapeutic alternative to safely lower HbA1c levels across all age groups. Pediatr Diabetes. 2002;3:10-15.
24. Owen S. Pediatric pumps: barriers and breakthrough. Diabetes Educ. 2006;32:29S-38S.
25. Ratner RE, Hirsh IB, Neifing JL, et al. Less hypoglycemia with insulin glargine in intensive insulin therapy for type 1 diabetes. U.S. Study Group of Insulin Glargine in Type 1 Diabetes. Diabetes Care.2000;23:639-643.
26. Vague P, Selam JL, Skeie S, et al. Insulin detemir is associated with more predictable glycemic control and reduced risk of hypoglycemia than NPH insulin in patients with type 1 diabetes on a basal-bolus regimen with premeal insulin aspart. Diabetes Care. 2003;26:590-596.
To comment on this article, contact rdavidson@jobson.com.





