US Pharm. 2013;38(12):30-34.
ABSTRACT: According to the U.S. Preventive Services Task Force, endoscopic screening for colorectal cancer has the potential to save thousands of lives each year. To maximize the diagnostic accuracy of a colonoscopy, it is crucial that the patient be prepared for the screening procedure. Two factors limiting the success of colonoscopy preparations are patient compliance and tolerability. Bowel preparations currently available for colonoscopy include compounds such as polyethylene glycol and sodium phosphate. Despite the emergence of newer alternatives, there is still no ideal preparation for colonoscopy. Community pharmacists can educate patients, address their concerns, and offer them individualized recommendations in order to improve the rates of successful bowel cleansing for colonoscopy.
Colorectal cancer (CRC) is the second leading cause of cancer-related deaths in the United States and the third most common cancer in both men and women.1 The 5-year survival rate for patients whose CRC is diagnosed early (stage I) is greater than 90%, supporting the importance of early detection in reducing mortality.2
The U.S. Preventive Services Task Force has stated that endoscopic screening for CRC can potentially save thousands of lives annually.3 It is essential, therefore, that patients be prepared for their screening procedure in order to maximize its diagnostic accuracy. According to the American Society for Gastrointestinal Endoscopy, colonoscopy is considered the gold standard for CRC screening.4 Inadequate bowel cleansing is responsible for nearly one-third of incomplete procedures, resulting in a reduced rate of polyp and adenoma detection.5 The accuracy of colonoscopy results relies heavily upon the selection of an appropriate bowel-preparation regimen, along with strict adherence to administration. This review will address the various bowel preparations used for colonoscopy and identify strategies for improving patient compliance and tolerability, two factors that limit the success of bowel preparations.
Traditional Bowel Preparations
Bowel preparations used for colonoscopy generally contain a hyperosmotic, isosmotic, or stimulant, or a combination of these agents. Factors that should be considered when a regimen is being selected include coexisting conditions, concomitant medications, age, and patient preference. The most commonly dispensed bowel preparations are polyethylene glycol (PEG) and sodium phosphate (NaP). Poor tolerability of these traditional agents has triggered the development of newer agents, such as SUPREP, Suclear, and Prepopik.
PEG-Based Preparations: PEG-based preparations are isosmotic, nonabsorbable electrolyte solutions that cleanse the bowel through cathartic effects resulting from ingestion of a large fluid volume. Several PEG-based preparations are available, each differing in aspects such as dose volume, electrolyte content, and flavor (e.g., GoLYTELY, Colyte, PEG-3350, NuLYTELY, Trilyte). Decreased tolerance of, and compliance with, older PEG-based solutions is commonly attributed to unpleasant taste. Newer sulfate-free, PEG-based products (e.g., NuLYTELY, Trilyte) have been developed to improve palatability.6 In addition, standard PEG-based preparations are now available with flavor enhancers to improve tolerability.7
Tolerability and compliance are also influenced by the large volume of fluid required for ingestion. For conventional dosing, traditional PEG-based preparations should be reconstituted with water to 4 L and consumed as 8 oz every 10 minutes until finished or rectal fluids are clear.8 As an alternative, split dosing has successfully addressed volume-related challenges while improving efficacy (TABLE 1).9-13 Split-dose regimens generally recommend the ingestion of 2 L the evening before colonoscopy, followed by 2 L the next morning. The second dose, taken approximately 5 hours before the procedure, minimizes the buildup of thick intestinal secretions in the colon, resulting in improved polyp detection.6,12,14
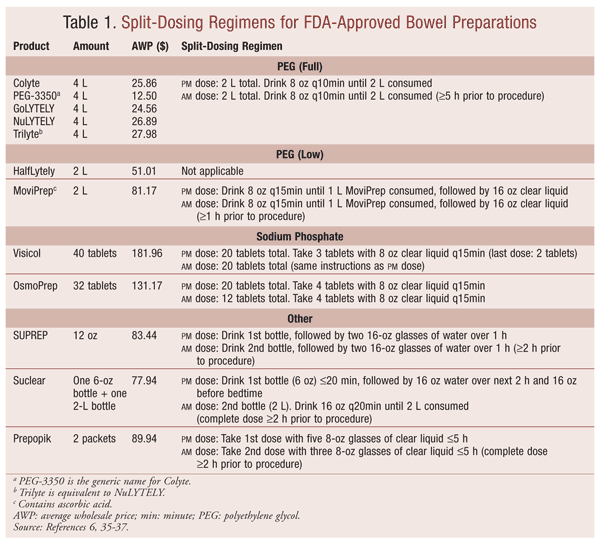
Low-volume 2-L PEG preparations such as HalfLytely (which includes bisacodyl) and MoviPrep (which contains ascorbic acid) are also alternatives to full-volume regimens. The additional ingredients act synergistically, with PEG causing a reduction in volume without compromising safety and efficacy.10 With HalfLytely, after the bisacodyl tablets are taken, the patient should wait for a bowel movement to occur, or at least 6 hours, before consuming the PEG solution.15 MoviPrep may be given as either a one-dose regimen the evening before colonoscopy or a split-dose regimen of 1 L followed by 0.5 L of clear liquid the evening before, then repeated the following morning.16
Most PEG-based solutions contain electrolytes, thereby preventing shifts in fluid and electrolytes. Some providers recommend MiraLAX—PEG-3350 containing no electrolytes—as a more tolerable alternative to the full-volume regimens. Although widely used in practice, MiraLAX as a bowel preparation is an off-label use. As a bowel preparation, MiraLAX should be mixed with 2 L of clear liquid or a sports drink and consumed as 8 oz every 10 minutes the evening before the procedure.17 In theory, the lack of electrolyte supplementation increases the risk of electrolyte imbalances. Nevertheless, MiraLAX has been administered as one-dose and split-dose regimens. The split-dose regimen was found to be less efficacious for bowel cleansing compared with split-dose PEG.18,19
Overall, the efficacy and tolerability of PEG-based preparations are generally considered inferior to those of NaP preparations. Conversely, compared with NaP, PEG-based products are thought to be a safer alternative for patients at risk for developing complications secondary to fluid and electrolyte imbalances (e.g., patients with renal failure, liver failure, or congestive heart failure [CHF]). In addition, PEG-based solutions do not appear to alter the cells of the colonic mucosa, making it an effective option for patients with suspected inflammatory bowel disease.20
NaP Preparation: NaP was originally developed to avoid the large-volume ingestion associated with PEG preparations.17 NaP works as a hyperosmotic laxative, drawing water into the lumen of the colon to stimulate peristalsis and catharsis. Until 2008, when the FDA issued a black box warning, Fleet Phospho-soda was a commonly used OTC NaP preparation for bowel cleansing. Because of the increased risk of acute phosphate nephropathy (APN), which may result in permanent renal impairment, this solution is no longer available on the U.S. market, according to the manufacturer (oral communication, August 2013).8,21 Currently, only tablet formulations of NaP (Visicol, OsmoPrep) are available in the U.S. The main difference between Visicol and OsmoPrep is the presence or absence of microcrystalline cellulose (MCC).8,22,23 This insoluble, inactive compound is thought to produce a residue that theoretically may reduce visualization of the colonic mucosa during colonoscopy.24 Visicol contains MCC and requires the ingestion of 40 tablets to complete the regimen; OsmoPrep, which is MCC-free, requires 32 tablets (TABLE 1).
A common misconception about NaP products is that hydration is not necessary. As with the solution, the tablets induce cathartic effects and must be taken with 4 L of clear liquid to maintain adequate hydration and prevent serious adverse effects (AEs), such as fluid and electrolyte imbalances.22,23,25 Additionally, these tablet formulations carry the risk of APN and should be monitored accordingly.
In general, NaP products are considered more effective and tolerable than full-volume PEG products, but they may be less safe, particularly in patients with kidney disease, CHF, advanced liver disease, or a sodium-restricted diet.20
Magnesium Citrate: Although not FDA-approved as a bowel preparation, magnesium citrate is a hyperosmotic laxative that has long been a component of bowel-cleansing regimens. In addition to drawing fluid into the colon and increasing motility, magnesium citrate also stimulates the release of cholecystokinin, leading to intestinal accumulation of fluid and electrolytes and eventual evacuation of the bowels.20,24,26
Magnesium citrate typically is not used as monotherapy for bowel cleansing. It is frequently administered with bisacodyl and has been used as an adjunct to low-volume PEG for colonoscopy preparation. Patients with abdominal pain or hemorrhage or renal dysfunction should avoid its use. Although they are considered less tolerable than NaP products, regimens containing magnesium citrate are generally well tolerated. For this reason, along with its relative affordability, magnesium citrate may be considered an alternative for appropriate patient populations.26
Newer Bowel Preparations
Over the last several years, three new products have been FDA-approved for use in colonoscopy preparation. These agents were developed to better address the challenge of improving tolerability while maintaining efficacy. Caution should be exercised in patients with a history of arrhythmias, seizures, renal impairment, significant gastrointestinal (GI) disease, impaired gag reflex, regurgitation or aspiration, and fluid or electrolyte imbalances.27-29
Oral Sulfate Solution (SUPREP): SUPREP is a low-volume osmotic laxative containing a combination of poorly absorbed sulfate salts (sodium sulfate, potassium sulfate, magnesium sulfate). These salts draw water into the lumen of the GI tract to promote cathartic effects.27
To decrease GI AEs, including vomiting, the manufacturer recommends administering SUPREP as a split-dose regimen. On the evening before the procedure, the first 16-oz bottle should be consumed, followed by 32 oz of water, over 1 hour. On the morning of the procedure, at least 2 hours prior to the procedure, the regimen is repeated with the second 16-oz bottle.11,27
Clinical trials evaluating both one-dose and split-dose regimens have demonstrated similar efficacy and safety compared with PEG preparations. SUPREP, therefore, may be an alternative, particularly in patients unable to tolerate PEG.30
Sodium Picosulfate Preparation (Prepopik): Prepopik (sodium picosulfate, magnesium hydroxide, anhydrous citric acid) is a 10-oz preparation that combines a stimulant laxative with an osmotic laxative, making it the lowest-volume preparation currently available in the U.S. Sodium picosulfate is a prodrug that is hydrolyzed and activated by colonic bacteria to produce peristalsis. Magnesium hydroxide and anhydrous citric acid react with water to form magnesium citrate, further promoting a laxative effect.8,28
Prepopik is contraindicated in patients with severely reduced renal function. Prior or current use of antibiotics may decrease the preparation’s efficacy because of the conversion of sodium picosulfate to its active metabolite by colonic bacteria, so evaluation is necessary in this case.28
When one-dose regimens of low-volume PEG-3350 plus bisacodyl tablets were compared with Prepopik, both preparations were equally effective for bowel cleansing. In a comparison of split-dose regimens, Prepopik had slightly superior efficacy. Tolerability was also improved with Prepopik. Bowel preparations containing this combination have been studied and used extensively outside the U.S. and have achieved similar results, further supporting this preparation’s role as an alternative.24,28,30,31
Combination Osmotic Laxative (Suclear): Suclear (sodium sulfate, potassium sulfate, and magnesium sulfate; PEG-3350, sodium chloride, sodium bicarbonate, potassium chloride), the latest bowel preparation approved by the FDA (January 2013), combines the osmotic effects of oral sulfates and PEG to induce cathartic effects in the GI tract.29
Split-dose administration of Suclear is preferred. On the evening before the procedure, the 6-oz bottle of oral sulfate solution should be diluted and consumed, followed by 32 oz of water, over 2 hours. On the morning of the procedure, the 2-L jug of PEG should be reconstituted and consumed at a rate of 16 oz every 20 minutes, to be completed 2 hours before procedure.29
In two different studies comparing one-dose regimens and split-dose regimens of low-volume PEG-3350, with or without bisacodyl tablets, the proportion of successful colon cleansing was similar between groups. Suclear may be considered an alternative bowel preparation.29
Adjunctive Agents
Before the widespread use of PEG, the enema was an important component of bowel preparation. The increased discomfort and lack of improvement in the quality of bowel cleansing have minimized the popularity of this agent. The enema appears to maintain its role as an adjunct in patients who present for colonoscopy with inadequate bowel preparation.
The need to ingest a large volume of fluid has been one of the greatest challenges in successful administration of bowel preparations. Laxatives such as senna, bisacodyl, and magnesium citrate have been used for their additive cathartic effects, reducing the volume of PEG required for cleansing.
GI symptoms such as nausea, bloating, and gas also limit the tolerability of bowel preparations. These symptoms can be alleviated with use of adjunct agents. Metoclopramide has been shown to reduce the occurrence of nausea and bloating. Simethicone can reduce bloating and gas, and it also may be used to eliminate foam formation and improve visualization for colonoscopy screenings.14,24,26
Education and Counseling
Patient education regarding preparation, administration, and AE management reduces the rates of missed polyps (up to 48%), procedure complications, and repeat colonoscopies.10,32,33 Community pharmacists can play a key role in ensuring that patients are prepared for colonoscopy. Information on topics such as timing, concomitant medications, diet, improving tolerability, and hydration is helpful for successful bowel preparation.
For optimal colonic cleansing, at least a portion of the bowel preparation must be ingested within 6 to 8 hours of the procedure. To ensure optimal visualization of the colon, patients must precisely follow the instructions provided by their healthcare professional (HCP) or the manufacturer.34
Patients should review their current medication list with their HCP. For patients taking antithrombotic agents, the risks and benefits of interrupting therapy should be weighed. OTC medications with bleeding risk (e.g., aspirin, other nonsteroidal anti-inflammatory drugs) are generally safe to continue, but should be reviewed with the HCP. Patients taking diabetic medications, particularly insulin, may need to adjust the dose when completing a bowel-preparation regimen. All other prescription medications generally can be continued, unless otherwise advised. Patients taking diuretics, ACE inhibitors, or angiotensin receptor blockers should use NaP-containing regimens with caution. OTC bulk-forming laxatives (e.g., Metamucil, Citrucel), as well as iron and iron-containing products (e.g., multivitamins), should be avoided 5 days before the procedure.26
In general, for 2 to 3 days prior to a colonoscopy (range 1-5 days, depending upon HCP instructions), patients are advised to adhere to a low-residue diet, limiting intake of fiber and other indigestible foods. Patients should also maintain a clear liquid diet starting 24 hours before the procedure. Red or purple liquids, alcohol, and milk or milk-based fluids should be avoided. In general, nothing should be ingested 2 hours prior to the procedure.26,34,35
Suggestions addressing taste and volume have been developed to improve the tolerability of bowel preparations. Pharmacists can work with HCPs to suggest split-dose regimens, lower-volume preparations, and the use of adjunctive agents to address concerns regarding fluid volume. To improve taste, patients can chill the solutions, use a straw to bypass the taste buds, or add flavor (e.g., the included flavor packets, clear sugar-free powder mixes, or lemon juice). Sucking on fresh lemon or lime slices or rubbing them on the tongue may also improve palatability, as citrus appears to best mask the saltiness of the preparations.35
Because of the significant fluid and electrolyte imbalance that can result from bowel-cleansing regimens, adequate hydration throughout the preparation period is essential. Patients should be advised to consume at least 64 oz (approximately 2 L) of clear liquid on the day before the colonoscopy. Clear fluids may be consumed for up to 2 hours prior to the procedure. A minimum of 32 oz of fluid should be consumed within 8 hours after the colonoscopy to prevent any complications.25
Patients also should be counseled about common AEs (e.g., diarrhea, nausea, bloating) and the need to stay at home or near a restroom because of the frequent bowel movements triggered by the preparation. After ingestion, the expected onset of action is approximately 1 hour for PEG-based bowel preparations and 3 to 6 hours for NaP preparations.8 Patients may experience anal irritation, which can be alleviated by using wet wipes or petrolatum.26
Conclusion
Over the past three decades, the number of adults screened for CRC in the U.S. has increased significantly.2 The diagnostic accuracy of colonoscopy is limited by inadequate bowel cleansing, often because of poor patient compliance and tolerability. Despite the development of newer bowel preparations, there continues to be no ideal regimen. Adjunctive strategies may be used to improve screening visualization, palatability, and GI AEs. Community pharmacists can be a valuable resource in ensuring a successful colonoscopy.
REFERENCES
1. CDC. Colorectal cancer statistics. www.cdc.gov/cancer/colorectal/statistics/index.htm. Accessed July 29, 2013.
2. American Cancer Society. Colorectal Cancer Facts & Figures 2011-2013. Atlanta, GA: American Cancer Society; 2011.
3. U.S. Preventive Services Task Force. Screening for colorectal cancer: recommendation statement.
www.uspreventiveservicestaskforce.org/uspstf08/colocancer/colors.htm. Accessed July 29, 2013.
4. American Society for Gastrointestinal Endoscopy. Colorectal cancer
screening. www.asge.org/press/press.aspx?id=552. Accessed September 4,
2013.
5. Froehlich F, Wietlisbach V, Bonvers JJ, et al. Impact of colonic
cleansing on quality and diagnostic yield of colonoscopy: the European
Panel of Appropriateness of Gastrointestinal Endoscopy European
multicenter study. Gastrointest Endosc. 2005;61:378-384.
6. DiPalma JA, Marshall JB. Comparison of a new sulfate-free
polyethylene glycol electrolyte lavage solution versus a standard
solution for colonoscopy cleansing. Gastrointest Endosc. 1990;36:285-289.
7. Diab FH, Marshall JB. The palatability of five colonic lavage solutions. Aliment Pharmacol Ther. 1996;10:815-819.
8. Lexicomp. www.lexicomp.com. Accessed July 31, 2013.
9. Cohen LB. Split dosing of bowel preparations for colonoscopy: an analysis of its efficacy, safety, and tolerability. Gastrointest Endosc. 2010;72:406-412.
10. Marmo R, Rotondano G, Riccio G, et al. Effective bowel cleansing
before colonoscopy: a randomized study of split-dosage versus non-split
dosage regimens of high-volume versus low-volume polyethylene glycol
solutions. Gastrointest Endosc. 2010;72:313-320.
11. Rex DK, Di Palma JA, Rodriguez R, et al. A randomized clinical
study comparing reduced-volume oral sulfate solution with standard
4-liter sulfate-free electrolyte lavage solution as preparation for
colonoscopy. Gastrointest Endosc. 2010;72:328-336.
12. Belsey J, Crosta C, Epstein O, et al. Meta-analysis: the relative
efficacy of oral bowel preparations for colonoscopy 1985-2010. Aliment Pharmacol Ther. 2012;35:222-237.
13. Kilgore TW, Abdinoor AA, Szary NM, et al. Bowel preparation with
split-dose polyethylene glycol before colonscopy: a meta-analysis of
randomized controlled trials. Gastrointest Endosc. 2011;73:1240-1245.
14. Shawki S, Wexner SD. Oral colorectal cleansing preparations in adults. Drugs. 2008;68:417-437.
15. HalfLytely and Bisacodyl Tablets (PEG-3350, sodium chloride,
sodium bicarbonate and potassium chloride for oral solution and
bisacodyl delayed-release tablet) product information. Braintree, MA:
Braintree Laboratories; July 2010.
16. MoviPrep (PEG-3350, sodium sulfate, sodium chloride, potassium
chloride, sodium ascorbate, and ascorbic acid for oral solution) product
information. Raleigh, NC: Salix Pharmaceuticals; October 2012.
17. Samarasena JB, Muthusamy VR, Jamal MM. Split-dosed
MiraLAX/Gatorade is an effective, safe, and tolerable option for bowel
preparation in low-risk patients: a randomized controlled study. Am J Gastroenterol. 2012;107:1036-1042.
18. Nepal S, Atreja A, Lashner BA. Optimal bowel preparation for colonoscopy. In: Ribeiro da Rocha JJ, ed. Endoscopic Procedures in Colon and Rectum.
InTech; 2011.
www.intechopen.com/books/endoscopic-procedures-in-colon-andrectum/optimal-bowel-preparation-for-colonoscopy.
Accessed July 29, 2013.
19. Enestvedt BK, Fennerty MB, Eisen GM. Randomised clinical trial:
MiraLAX vs. Golytely—a controlled study of efficacy and patient
tolerability in bowel preparation for colonoscopy. Aliment Pharmacol Ther. 2011;33:33-40.
20. Barkun A, Chiba N, Enns R, et al. Commonly used preparations for
colonoscopy: efficacy, tolerability and safety—a Canadian Association of
Gastroenterology position paper. Can J Gastroenterol. 2006;20:699-710.
21. Fleet. Phospho-soda—important prescribing and safety information.
www.fleetlabs.com/phospho-soda.php. Accessed July 29, 2013.
22. Visicol (sodium phosphate monobasic monohydrate, USP and sodium
phosphate dibasic anhydrous, USP) product information. Morrisville, NC:
Salix Pharmaceuticals; March 2009.
23. OsmoPrep (sodium phosphate monobasic monohydrate, USP and sodium
phosphate dibasic anhydrous, USP) product information. Morrisville, NC:
Salix Pharmaceuticals; October 2012.
24. Wexner SD, Beck DE, Baron TH, et al. A consensus document on
bowel preparation before colonoscopy: prepared by a task force from the
American Society of Colon and Rectal Surgeons (ASCRS), the American
Society for Gastrointestinal Endoscopy (ASGE), and the Society of
American Gastrointestinal and Endoscopic Surgeons (SAGES). Dis Colon Rectum. 2006;49:792-809.
25. Lichtenstein GR, Cohen LB, Uribarri J. Review article: bowel preparation for colonoscopy—the importance of adequate hydration. Aliment Pharmacol Ther. 2007;26:633-641.
26. Atreja A, Nepal S, Lashner BA. Making the most of currently available bowel preparations for colonoscopy. Cleve Clin J Med. 2010;77:317-326.
27. SUPREP (sodium sulfate, potassium sulfate, and magnesium sulfate)
product information. Braintree, MA: Braintree Laboratories; August
2010.
28. Prepopik (sodium picosulfate, magnesium oxide, and anhydrous
citric acid) product information. Parsippany, NJ: Ferring
Pharmaceuticals; July 2012.
29. Suclear (sodium sulfate, potassium sulfate and magnesium sulfate
oral solution; and PEG-3350, sodium chloride, sodium bicarbonate and
potassium chloride for oral solution) product information. Braintree,
MA: Braintree Laboratories; January 2013.
30. Di Palma J, Rex D. Advances in bowel preparations: new formulation and clinical results. Gastroenterol Nurs. 2011;34(suppl 2):S2-S8.
31. Katz PO, Rex DK, Epstein M, et al. A dual-action, low-volume
bowel cleanser administered the day before colonoscopy: results from the
SEE CLEAR II study. Am J Gastroenterol. 2013;108:401-409.
32. Abuksis G, Mor M, Segal N, et al. A patient education program is
cost-effective for preventing failure of endoscopic procedures in a
gastroenterology department. Am J Gastroenterol. 2001;96:1786-1790.
33. Tae JW, Lee JC, Hong SJ, et al. Impact of patient education with
cartoon visual aids on the quality of bowel preparation for colonoscopy. Gastrointest Endosc. 2012;76:804-811.
34. Cohen LB, Tennyson C. Bowel preparation for colonoscopy: maximizing efficacy, minimizing risk. Gastroenterol Endosc News. March 2011;1-8.
35. A-Rahim YI, Falchuk M. Bowel preparation for colonoscopy and flexible sigmoidoscopy in adults. UpToDate. www.uptodate.com/contents/bowel-preparation-for-colonoscopy-and-flexible-sigmoidoscopy-in-adults. Accessed July 29, 2013.
36. RedBook [online database]. www.redbook.com/redbook/online. Accessed July 31, 2013.
37. Comparison of bowel preparations for colonoscopy (January 2009).
University of Michigan.
hwww.med.umich.edu/1info/fhp/practiceguides/adult.cancer/bowel_prep_comparison.pdf.
Accessed July 30, 2013.
To comment on this article, contact rdavidson@uspharmacist.com.





