US Pharm. 2013;38(10):7.
The FDA, drug manufacturers, and the supply chain work together to achieve availability of drugs when they are needed. According to government reports, however, the number of drug shortages nearly tripled from 61 in 2005 to 178 (including 132 injectables) in 2010; rose to 251 (including 183 injectables) in 2011; and was 237 in August 2013. Such shortages involve a very small proportion (1%) of the drugs on the market, but the 29.5% average annual increase from 2005 to 2010 is a notable trend.
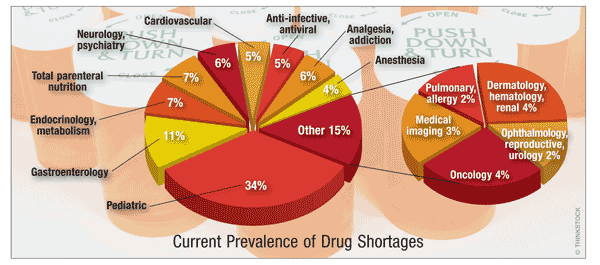
Prevalence: Of the current 237 drug shortages, 6.3%, 36.7%, and 57% were prevalent since June, July, and August 2013, respectively. Shortages include injections (75.1%), tablets/capsules (13.9%), solutions/suspensions/emulsions (5.9%), and gels/ointments (2.1%). Although the use of injectables for cancer treatment has increased an average of 20% per year (2005-2011), it has not been spared shortages (1.8% shortage annually for 2006-2011). Anesthesia, emergency medicine, and electrolytes for IV feeding, among other drugs, also were in short supply during this period.
Contributing Factors: In 2010 and 2011, problems at the manufacturing facility (43%), delays in shipping or manufacturing (15%), and shortages of active pharmaceutical ingredients (10%) were identified as significant contributors to drug shortages. The complexity of the manufacturing process for injectables (e.g., need for a dedicated line) has increased the burden on three generic injectable manufacturers feeding 71% of the market by volume and on one manufacturer that produces 90% of the drug.
According to a study of the Medicare Part B volume of services (VOS) for oncology injectables, drugs that subsequently experienced a shortage were those in which the volume of sales was declining an average of 1.8% annually in the prior period; those that did not experience a shortage had an average 4.3% annual increase in VOS. Oncology injectables with a 5-year average of 5.5% decrease in price were found to be experiencing a shortage, in contrast to an annual increase of 0.7% in price for oncology injectables that were not in short supply.
Corrective Measures: The 2012 Food and Drug Administration Safety and Innovation Act requires manufacturers to help identify shortages and find temporary solutions by alerting the FDA 6 months in advance about anticipated market withdrawal or drug-supply interruptions. The FDA has asked manufacturers to increase production (31%); is considering flexibility in its regulatory discretion regarding quality of products (28%); is expediting review of regulatory submissions (26%); and is exercising discretion regarding controlled importation of similar products approved abroad, but not approved in the United States (5%). FDA actions or interventions have prevented 128 drug shortages between October 2012 and March 2013.
To comment on this article, contact rdavidson@uspharmacist.com.






