US Pharm. 2012;37(6):HS11-HS13.
Systemic lupus erythematosus (SLE) is an autoimmune disease that may affect nearly any organ system in the human body. The autoimmunity present in patients with SLE causes local inflammation and tissue damage, and it may present with a variety of symptoms depending on the organ system involved. When this disease affects the kidney, it is termed lupus nephritis. The condition is often associated with poorer outcomes and is of particular interest for this reason. Even with the many recent advances in treatments for lupus nephritis, 10% to 15% of treated patients will still progress to end-stage renal disease (ESRD) and require dialysis or transplant.1
The incidence of SLE is strongly related to race, gender, and socioeconomic status. The prevalence of SLE is much higher in women than in men (up to 12:1 in adults), and the African-American population is up to 10 times more likely to develop the disease as compared to its white counterparts.1 Approximately 35% of adults diagnosed with SLE also present with clinical evidence of nephritis at the time of diagnosis, with 50% to 60% of patients developing nephritis during the first 10 years of disease.2 If renal involvement is present, African Americans have been shown to have poorer outcomes as compared to white patients or those of Hispanic descent. Independent of race, low income has also been shown to be a risk factor for the development and progression of lupus nephritis.3
Declining renal function, as measured by increasing serum creatinine and blood urea nitrogen with concurrent decrease in measured or calculated creatinine clearance, is one of the first signs of lupus nephritis in SLE patients. The American College of Rheumatology (ACR) defines lupus nephritis as persistent proteinuria >0.5 g/day or >3+ by dipstick, and/or cellular casts including red blood cells, hemoglobin, granular, tubular, or mixed.2 In order to verify suspicions of lupus nephritis, a renal biopsy must be performed.2
Lupus Nephritis Classification
The World Health Organization (WHO) originally classified lupus nephritis based upon histological characteristics of a renal biopsy. The WHO classification was revised by the International Society of Nephrology and the Renal Pathology Society (ISN/RPS) into six classes (TABLE 1). Patients often progress from one classification to another, and treatments will differ according to the severity and extent of the disease.4

Treatment
The goal of treatment is to both improve kidney function and delay the onset of kidney failure. Patients who present with ISN/RPS class I or class II mesangial disease generally have good long-term outcomes, and treatment with either an angiotensin-converting enzyme (ACE) inhibitor or angiotensin receptor blocker (ARB) is recommended for any patient with proteinuria ≥0.5 g/day, with the primary goal being the prevention of the progression of nephritis.2
Focal or diffuse lupus nephritis (ISN/RPS classes III-IV) confers a much greater risk of progression, potentially to ESRD, and requires more aggressive treatment with immunosuppressive medications.4 Treatment regimens generally include an aggressive induction phase in which the goal is to induce complete or partial disease remission over the course of a few months. This is followed by a maintenance phase in which lower doses of immunosuppressive agents are used to maintain remission and prevent flare-ups.
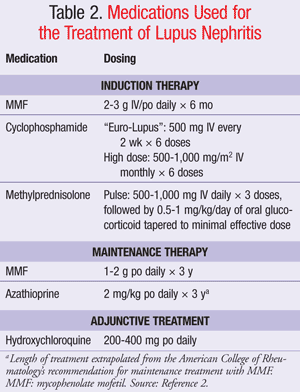
There have been some small observational studies suggesting that antimalarials may reduce the risk of renal flares, improve maintenance of remission, and reduce the risk of progression to ESRD in addition to reducing the risk of thrombosis. The current ACR guidelines recommend the addition of hydroxychloroquine to the regimens of all SLE patients with nephritis (TABLE 2).2,5-9
Cyclophosphamide Induction Therapy
Traditionally, cyclophosphamide combined with corticosteroids has been the standard for induction therapy. Cyclophosphamide, a synthetic antineoplastic drug chemically related to the nitrogen mustards, is often used in chemotherapy regimens targeted against a variety of malignancies. It exerts its antineoplastic and immunosuppressant effects by cross-linking DNA preferentially in quickly dividing cells, such as cancerous cells and leukocytes.10
The combination of cyclophosphamide and corticosteroids as induction therapy has been proven effective in randomized controlled trials and was shown to be more effective than corticosteroids alone. Patients treated with methylprednisolone plus monthly “pulse” dosing of IV cyclophosphamide, followed by quarterly injections, were more likely to achieve and maintain remission as compared to those treated with cyclophosphamide or methylprednisolone alone.11,12 Daily oral cyclophosphamide may also be used for induction therapy; however, IV pulse therapy is preferred due to the decreased cumulative exposure to cyclophosphamide and a lower incidence of cytopenia.13
While this combination is effective, patients treated with cyclophosphamide and methylprednisolone for induction therapy in lupus nephritis are at an increased risk of adverse drug events as compared to those treated with methylprednisolone monotherapy. Adverse events include amenorrhea (43% vs. 7.4%), cervical dysplasia (7.1% vs. 0%), herpes zoster (21% vs. 3.7%), and infection (32% vs. 7.4%).11
In an attempt to avoid these complications of therapy, trials of lower-dose cyclophosphamide have been studied for induction therapy. The Euro-Lupus Nephritis Trial compared traditional high-dose therapy, dosed monthly for 6 months followed by quarterly dosing (0.5 g/m2 initially, then adjusted based on white blood cell count nadir), with low-dose therapy (500 mg every 2 weeks × 6 doses followed by azathioprine maintenance therapy of 2 mg/kg/day).14 After a 41-month follow-up, there were no significant differences in rate of treatment failure or incidence of renal flares between the two groups. The ACR guidelines include recommendations for both induction regimens, with the lower Euro-Lupus dosing being preferred in white patients with Western or Southern European ethnic background.2
Induction Therapy With MMF
Mycophenolate mofetil (MMF) is metabolized to mycophenolic acid, which inhibits inosine monophosphate dehydrogenase and in turn inhibits the de novo pathway for guanine nucleotide synthesis. As the proliferation of B and T cells is highly dependent on this pathway, MMF has a potent cytostatic effect on lymphocytes.15
Within the last decade, MMF has been shown in studies to be effective for induction therapy for lupus nephritis. In a study conducted in Hong Kong, 42 patients with diffuse proliferative lupus nephritis were randomized to either oral MMF (2 g daily for 6 months followed by 1 g daily for 6 months) or oral cyclophosphamide (2.5 mg/kg/day for 6 months) followed by oral azathioprine (1.5 mg/kg/day for 6 months). Both arms of the study also received oral prednisolone daily. After 12 months of follow-up, there were no significant differences between the groups in terms of complete or partial remission rates. There were also no significant differences in relapse rate between the two arms; however, infections were significantly less common in the MMF induction group (13% vs. 40%).16
A larger randomized, controlled trial, the Aspreva Lupus Management Study (ALMS), was conducted to verify the results of some of the smaller studies.17 Three hundred and seventy patients with WHO class III-V lupus nephritis were randomized to 24 weeks of treatment with either MMF (3 g daily) or IV cyclophosphamide (0.5-1 g/m2). Both arms were also treated with prednisolone daily. After the 6-month study period, no significant differences in remission rates or mortality were seen between the groups. Diarrhea occurred more often in the MMF group (28% vs. 12%), while nausea/vomiting (37% vs. 45%) and hair loss (10% vs. 35%) were more common with cyclophosphamide therapy. This study helped confirm that MMF is a viable alternative to cyclophosphamide induction therapy, and may confer less risk of toxicity.17
The ACR recommends 2 to 3 g/day of MMF as part of an induction regimen with pulsed corticosteroids. The 3 g/day dosage is favored in patients with proteinuria and significant rises in serum creatinine.2
Maintenance Therapy
Once remission has been induced, long-term maintenance therapy should be initiated to reduce the risk of recurrence and long-term complications of the disease. Various therapies have been studied for maintenance therapy, including azathioprine, MMF, and cyclophosphamide, all combined with corticosteroids. One study compared azathioprine (1-3 mg/kg/day), MMF (0.5-3 g/day), and IV cyclophosphamide (0.5-1 g/m2) for the maintenance therapy of proliferative lupus nephritis.18 The study involved 59 patients followed over a period of 2 years. After follow-up, the percentage of patients reaching the primary composite endpoint of death or glomerular filtration rate (GFR) <60mL/min/1.73 m2 were 20%, 25%, and 55% in the MMF, azathioprine, and cyclophosphamide groups, respectively. Adverse effects were also significantly less in the MMF and azathioprine groups as compared to the cyclophosphamide group.18
In order to compare azathioprine to MMF directly, another study was conducted for maintenance therapy for lupus nephritis. The MAINTAIN Nephritis Trial was conducted to compare maintenance of azathioprine 2 mg/kg/day with MMF 2 g/day in 105 patients with proliferative lupus nephritis.19 After a median 53-month follow-up, there were no significant differences between the groups in the rate of relapse or adverse events, aside from cytopenias being more common in the azathioprine group. The results of this trial show that both MMF and azathioprine are effective as maintenance regimens for lupus nephritis.19
The ACR guidelines recommend that patients be placed on maintenance regimens of either azathioprine or MMF. Available studies are not adequate to determine how rapidly azathioprine or MMF may be tapered; however, the guidelines recommend 3 years of MMF maintenance therapy.2 Corticosteroids may also be utilized in maintenance regimens; however, doses should be tapered over time to reduce the risk of adverse reactions.
Membranous Lupus Nephritis Treatment
The treatment of membranous lupus nephritis, ISN/RPS class V, differs from that of proliferative nephritis. Milder disease with stable kidney function, subnephrotic proteinuria, and the absence of proliferative lesions may only require treatment with an ACE inhibitor or an ARB in order to suppress the renin-angiotensin-aldosterone system (RAAS). For disease that is more severe or includes proliferative lesions, studies have demonstrated that treatment with a corticosteroid in combination with either cyclosporine, MMF, or cyclophosphamide is an effective regimen.20,21 The ACR recommends first-line treatment with MMF and prednisone followed by maintenance therapy with MMF or azathioprine.2
Other Therapies
Many other agents are currently undergoing studies for their role in the treatment of lupus nephritis, including rituximab, belimumab, and tacrolimus. Additional data from randomized controlled studies are needed before recommending these agents as first line; however, the ACR recommends treatment with one of these agents if a patient fails both cyclosporine and MMF induction therapy.2
Conclusion
Current therapies are quite effective in the treatment of lupus nephritis (TABLE 3 provides counseling points). Cyclophosphamide in combination with a corticosteroid remains an effective and reliable regimen for the induction phase of therapy for proliferative and membranous lupus nephritis. IV pulse therapy reduces cumulative cyclophosphamide exposure and may confer less risk of toxicity. MMF has also been shown to be an effective agent for the induction in combination with a corticosteroid, and appears to have less risk of serious toxicities as compared to cyclophosphamide regimens. Either medication is recommended by the ACR as first-line induction therapy for lupus nephritis.2 Once remission has been induced, maintenance regimens containing either azathioprine or MMF in combination with a corticosteroid appear to be most effective, while having the lowest risk for adverse events. The treatment of lupus nephritis will continue to evolve and improve as new treatments and data become available.

REFERENCES
1. Cameron JS. Lupus nephritis. J Am Soc Nephrol 1999;10:413-424.
2. Hahn BH, McMahon MA, Wilkinson A, et al. American
College of Rheumatology guidelines for screening, treatment, and
management of lupus nephritis. Arthritis Care Res. 2012;64:797-808.
3. Contreras G, Lenz O, Pardo V, et al. Outcomes in African Americans and Hispanics with lupus nephritis. Kidney Int. 2006;69:1846-1851.
4. Weening JJ, D’Agati VD, Schwartz MM, et al. The
classification of glomerulonephritis in systemic lupus erythematosus
revisited. J Am Soc Nephrol. 2004;15:241-250.
5. Tsakonas E, Joseph L, Esdaile JM, et al. A
long-term study of hydroxychloroquine withdrawal on exacerbations in
systemic lupus erythematosus. The Canadian Hydroxychloroquine Study
Group. Lupus. 1998;7:80-85.
6. Pons-Estel GJ, Alarcón GS, McGwin G Jr, et al; Lumina
Study Group. Protective effect of hydroxychloroquine on renal damage in
patients with lupus nephritis: LXV, data from a multiethnic US cohort. Arthritis Rheum. 2009;61:830-839.
7. Sisó A, Ramos-Casals M, Bové A, et al. Previous antimalarial therapy in patients diagnosed with lupus nephritis: influence on outcomes and survival. Lupus. 2008;17:281-288.
8. Kasitanon N, Fine DM, Haas M, et al. Hydroxychloroquine
use predicts complete renal remission within 12 months among patients
treated with mycophenolate mofetil therapy for membranous lupus
nephritis. Lupus. 2006;15:366-370.
9. Barber CE, Geldenhuys L, Hanly JG. Sustained remission of lupus nephritis. Lupus. 2006;15:94-101.
10. Cytoxan (cyclophosphamide) package insert. Deerfield, IL: Baxter Healthcare Corporation; 2009.
11. Gourley MF, Austin HA 3rd, Scott D, et al.
Methylprednisolone and cyclophosphamide, alone or in combination, in
patients with lupus nephritis. A randomized, controlled trial. Ann Intern Med. 1996;125:549-557.
12. Illei GG, Austin HA, Crane M, et al. Combination
therapy with pulse cyclophosphamide plus pulse methylprednisolone
improves long-term renal outcome without adding toxicity in patients
with lupus nephritis. Ann Intern Med. 2001;135:248-257.
13. Houssiau FA. Cyclophosphamide in lupus nephritis. Lupus. 2005;14:53-58.
14. Houssiau FA, Vasconcelos C, D’Cruz D, et al.
Immunosuppressive therapy in lupus nephritis: the Euro-Lupus Nephritis
Trial, a randomized trial of low-dose versus high-dose intravenous
cyclophosphamide. Arthritis Rheum. 2002;46:2121-2131.
15. CellCept (mycophenolate mofetil) package insert. South San Francisco, CA: Genentech; February 2010.
16. Chan TM, Li FK, Tang CS, et al. Efficacy of mycophenolate mofetil in patients with diffuse proliferative lupus nephritis. N Engl J Med. 2000;343:1156-1162.
17. Appel GB, Contreras G, Dooley A, et al. Mycophenolate
mofetil versus cyclosporine for induction treatment of lupus nephritis. J Am Soc Nephrol. 2009;20:1103-1112.
18. Contreras G, Pardo V, Leclercq B, et al. Sequential therapies for proliferative lupus nephritis. N Engl J Med. 2004;350:971-980.
19. Houssiau FA, D’Cruz D, Sangle S, et al. Azathioprine
versus mycophenolate mofetil for long-term immunosuppression in lupus
nephritis: results from the MAINTAIN Nephritis Trial. Ann Rheum Dis. 2010;69:2083-2089.
20. Radhakrishnan J, Moutzouris DA, Ginzler EM, et al.
Mycophenolate mofetil and intravenous cyclophosphamide are similar as
induction therapy for class V lupus nephritis. Kidney Int. 2010;77:152-160.
21. Austin HA 3rd, Ilei GG, Braun MJ, Ballow JE.
Randomized, controlled trial of prednisone, cyclophosphamide, and
cyclosporine in lupus membranous nephropathy. J Am Soc Nephrol. 2009;20:901-911.
22. Plaquenil (hydroxychloroquine sulfate) package insert. Bridgewater, NJ: Sanofi-Aventis; August 2009.
23. Imuran (azathioprine) package insert. Hunt Valley, MD: Prometheus Laboratories; April 2009.
24. Solu-Medrol (methylprednisolone sodium succinate) package insert. Kalamazoo, MI: Pharmacia and Upjohn; October 2001.
25. Deltasone (prednisone) package insert. New York, NY: Pharmacia and Upjohn; August 2006.
To comment on this article, contact rdavidson@uspharmacist.com.






