US Pharm. 2013;38(1):35-39.
ABSTRACT: Epilepsy is a disorder comprising a collection of seizures that differ in cause, symptoms, severity, and treatment. Seizures are assigned to one of two major categories: partial or generalized. Epilepsy cases can involve more than one type of seizure. Epilepsy may be classified into etiologic types and then further defined by presenting features. Drug therapy is based on type of seizure, age, gender, comorbidities, adverse-effect potential, drug interactions, and cost. Once treatment is initiated, about 60% of patients achieve adequate seizure control with monotherapy. Monitoring of serum concentrations is beneficial when suspecting toxicity, assessing medication adherence, or making dose adjustments. Pharmacists can improve patient outcomes through dose recommendations, drug interaction surveillance, adherence counseling, and adverse-effect monitoring.
Commonly referred to as seizure disorder, epilepsy is a disorder comprising a collection of seizures that differ in regard to cause, symptoms, severity, and treatment.1 Approximately 2 million people in the United States have epilepsy, and it is estimated that about 140,000 people develop the disorder every year.2 Despite advances in technology and treatment, epilepsy still remains a huge economic burden, with an estimated health care cost of $2.7 billion per year.3
Pathophysiology
Seizures occur when there is a disturbance in the balance of excitation and inhibition in the central nervous system (CNS).4 Many different mechanisms can cause neuronal hyperexcitability and lead to the development of seizures. These factors include changes in ion channel conduction, membrane receptor response, messenger systems, and gene transcription. Imbalances between excitatory (glutamate) and inhibitory (gamma-aminobutyric-acid, or GABA) neurotransmitters, as well as excessive acetylcholine, norepinephrine, and serotonin levels, may also precipitate seizures.1
Excitatory and inhibitory currents are primarily mediated by different channels including voltage- and ligand-gated channels. Voltage-gated channels include sodium and calcium channels. Ligand-gated channels include GABA and glutamate channels. Most of the pharmacologic agents used in treating epilepsy target these different channels.1
Classification
Seizures represent the clinical manifestations or symptoms of disturbed electrical activity, while epilepsy is a disorder of recurrent seizures. A patient is considered to have epilepsy when he or she has had two or more unprovoked seizures.1 Patients with an isolated seizure or seizures secondary to a reversible cause (e.g., hypoglycemia, alcohol withdrawal, high fever) are not considered to have epilepsy.
Seizures are classified according to the classification scheme proposed by the International League Against Epilepsy (ILAE).5 It is recommended that seizures be assigned to one of two major categories: partial seizures or generalized seizures. Partial seizures are further classified into simple partial seizures (patient has no loss of consciousness) or complex partial seizures (patient has loss of consciousness).5
The classification of epilepsies is more complex. Since epilepsy cases can involve more than one seizure type, specific classification of epilepsies is less meaningful. Rather, the newly recommended approach is to refer to epilepsies in regard to etiology—genetic, structural/metabolic, or unknown. The epilepsy can then be further characterized based on different presenting features (e.g., age of onset, EEG findings, neurologic examination).6
Symptoms
Seizures involve abnormal electrical activity that can produce changes in consciousness, motor and sensory activity, and behavior. The symptoms that occur can vary significantly from patient to patient and generally depend on the type of seizure or epilepsy. Generally, partial seizures originate in one hemisphere and can involve disturbances in motor function, sensory perception, autonomic function, and behavior. Sensory symptoms include paresthesias, abnormal tastes or smells, flashing lights, and hearing changes. Patients can report different autonomic symptoms, like sweating, epigastric sensation, or piloerection. Some patients can experience psychic symptoms including dysphasia, fear, and hallucinations. Automatisms, or repetitive movements, can be described in some patients and often present as chewing, swallowing, or sucking. A number of patients can experience an aura or warning symptoms minutes to hours before a seizure. Aura symptoms can differ significantly; examples include irritability, nausea, headache, and fear. Loss of consciousness and memory loss may also occur with partial seizures. Partial seizures may not terminate and can evolve into secondary generalized seizures.5,7
Generalized seizures involve both hemispheres and typically include bilateral motor symptoms and loss of consciousness.5 The many different types of generalized seizures (absence, myoclonic, tonic, clonic, tonic-clonic, atonic) will vary in motor symptom characteristics.
Treatment
Antiepileptic drug (AED) therapy may or may not be indicated after a patient experiences a first seizure. AED therapy is generally reserved for patients diagnosed with recurrent, unprovoked seizures or epilepsy and is typically not indicated in patients who have experienced an isolated seizure or seizures secondary to a reversible cause.
With the many different types of seizures and epilepsy syndromes, there really is no specific agent that is considered the drug of choice. Rather, specific AEDs are generally more effective and useful for certain types of seizures or epilepsy syndromes compared to others. A number of studies have compared agents for different types of seizures. The VA Cooperative Study compared carbamazepine, phenobarbital, phenytoin, and primidone in the treatment of partial and secondarily generalized tonic-clonic seizures.8 Based on the results, carbamazepine and phenytoin had the greatest treatment success. However, this study was conducted in the early 1980s and does not consider the use of newer AEDs. More recent studies have compared newer AEDs as initial monotherapy and have shown little difference in efficacy, but these studies are limited in duration (<1 year) and do not evaluate long-term efficacy.9-11
One of the latest studies, the Standard and New Antiepileptic Drugs (SANAD) trial, followed patients for 3 years.12 Results demonstrated that for partial seizures, lamotrigine and oxcarbazepine had the longest time to treatment failure in comparison to carbamazepine, topiramate, and gabapentin. For time to 12-month remission, lamotrigine and carbamazepine were significantly better versus oxcarbazepine, topiramate, and gabapentin.12 For generalized seizures, valproate and lamotrigine had the longest time to treatment failure, while valproate and topiramate had the shortest time to 12-month remission.13
The ILAE guidelines summarize evidence for efficacy of initial monotherapy for the treatment of newly diagnosed or untreated epilepsy.14 The American Academy of Neurology has issued guidelines assessing the efficacy, tolerability, and safety of newer AEDs in the treatment of newly diagnosed epilepsies.15 Although these guidelines make general recommendations on which AEDs should be considered for use based on efficacy or safety, they do not make specific recommendations on which AED is the clear drug of choice.14,15 With limited data and lack of specific recommendations from evidence-based guidelines, drug therapy is individualized, and selection of the most appropriate AED is based on a number of important considerations including seizure or epilepsy type, age, gender, comorbidities, potential adverse effects, drug interactions, and cost.14 Once treatment is initiated, approximately 60% of patients achieve adequate seizure control with monotherapy.16
Treatment Considerations
Seizure Type: Each AED is generally effective for certain types of seizures and can be classified as either broad or narrow spectrum. Broad-spectrum agents are effective for many different types of seizures. Narrow-spectrum agents are effective for only specific types of seizures. Determining seizure and epilepsy type is important in selecting the most appropriate agent (TABLE 1 lists specific agents and dosing).17
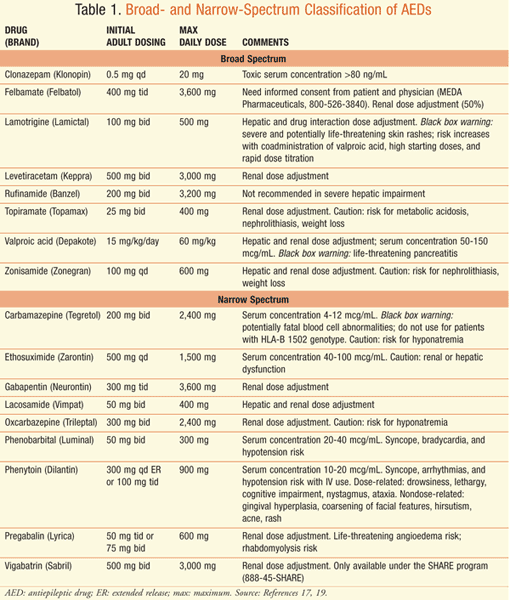
Adverse Effects: Since treatment of epilepsy is long-term and medication adherence is crucial for treatment success, drug tolerability is a major issue. AEDs can cause various adverse effects, and this is an important factor to consider when selecting therapy. Although most AEDs can cause common CNS side effects (dizziness and drowsiness), some agents are more tolerable compared to others. Impaired cognition is one major concern for many patients and family members. In general, newer agents like gabapentin and levetiracetam appear to have the least effects on cognition and may be better options.18
Patients with renal dysfunction should have dosage adjustment performed for topiramate, oxcarbazepine, levetiracetam, pregabalin, and lacosamide to avoid accumulation of the drug or its metabolites.19
A number of agents can affect the liver. Carbamazepine, phenytoin, and valproic acid can cause elevations in liver enzymes and thus should be avoided in patients with liver disease. Although these agents have also been known to cause hepatotoxicity, more severe liver problems are rare.19
AEDs may also cause different dermatologic reactions. Phenytoin can cause skin rash. Rare, fatal skin reactions like Stevens-Johnson syndrome (SJS) and toxic epidermal necrolysis (TEN) have been reported with most agents, but appear to be more common with carbamazepine, valproic acid, phenytoin, phenobarbital, and lamotrigine.20,21
Suicidal behavior or ideation has been reported in numerous studies evaluating the use of AEDs for epilepsy and psychiatric conditions. Due to the potential risk, a black box warning has been added to the labeling of all AEDs, and distribution of medication guides is required. It is generally recommended to monitor patients on AEDs for any new or worsening thoughts of suicidal behavior.22
Childbearing Plans: Because of significant concerns for teratogenicity, AED selection is also affected by a woman’s childbearing plans. Possible birth defects include intrauterine growth retardation, major malformations, and postnatal developmental delay. Since there is no consensus on the safest agent during pregnancy, the most effective agent is generally used at the lowest effective dose. However, because studies do demonstrate a greater risk for birth defects with valproic acid, it is commonly avoided.23 Women on oral contraceptives should be educated about the potential interaction between enzyme-inducing oral contraceptives and AEDs. When necessary, alternative contraception should be recommended.23
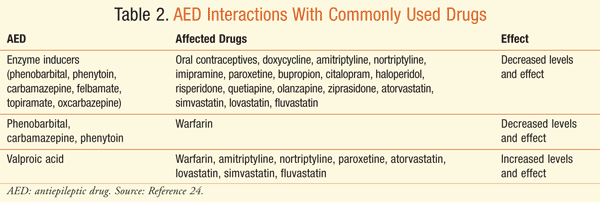
Drug Interactions: Many AEDs are metabolized through the CYP450 system in the liver and can be either enzyme inducers or enzyme inhibitors. Phenytoin, phenobarbital, carbamazepine, and primidone are well-known enzyme inducers, while valproic acid is the only major enzyme inhibitor. Enzyme inducers can enhance the metabolism of warfarin, oral contraceptives, antibiotics, and antidepressants resulting, in decreased serum concentrations and effects. Valproic acid can decrease the metabolism of warfarin, statins, and tricyclic antidepressants, resulting in increased serum concentrations and effects (TABLE 2).24 Carbamazepine is metabolized through CYP3A4 and can interact with CYP3A4 inhibitors, resulting in increased carbamazepine levels (e.g., erythromycin, diltiazem, cimetidine). Combination AED therapy is sometimes necessary, but drug interactions between AEDs are possible too. Since these agents can cause drug interactions with other medications and concomitant AEDs, it is important to screen for potential drug interactions prior to drug selection.24
Therapeutic Drug Monitoring
Therapeutic drug monitoring for AEDs is commonly used to help guide and assist clinicians with optimal dosing in patients. Monitoring serum concentrations can allow clinicians to achieve seizure control while minimizing adverse effects. Established drug levels for various AEDs should be primarily viewed as reference ranges and not therapeutic levels (see TABLE 1 for agents with established reference ranges). A serum concentration that falls below the reference range is unlikely to result in a therapeutic response, while a serum concentration that falls above the reference range is likely to result in toxicity. Yet it is important to recognize that because of individual variation and patient response, many patients can still achieve adequate seizure control at concentrations outside established reference ranges or develop toxicity at concentrations that fall within reference ranges. Thus, reference ranges serve merely as a guide, and each patient will have an individualized therapeutic level. Monitoring serum concentrations is most beneficial when suspecting toxicity, assessing medication adherence, or assisting with dosing in patients with altered pharmacokinetics.25
Epilepsy and the Pharmacist’s Role
Epilepsy is a chronic disorder that continues to be a huge economic burden. Although newer AEDs aim to increase treatment success and decrease the risk for adverse effects, there is still a strong need for improving patient outcomes. Pharmacists can play a significant role in optimizing therapy for patients with epilepsy. Patient counseling on the potential adverse effects of AEDs is important. Patients and family members should be educated on expected CNS and cognitive side effects, potential skin reactions, and the risk for suicidal behavior. Patient education should also address the importance of medication adherence. Patient medication profiles should be reviewed for possible drug interactions, and dosage adjustments or alternative agents should be recommended if necessary. Additionally, pharmacists can advise clinicians on appropriate therapeutic drug monitoring.
REFERENCES
1. Rogers SJ, Cavazos JE. Chapter 65. Epilepsy. In: Talbert RL, DiPiro JT, Matzke GR, et al, eds. Pharmacotherapy: A Pathophysiologic Approach.
8th ed. New York, NY: The McGraw Hill Companies; 2011.
www.accesspharmacy.com/content.aspx?aID=7985219. Accessed August 22,
2012.
2. Hirtz D, Thurman DJ, Gwinn-Hardy K, et al. How common are the “common” neurologic disorders? Neurology. 2007;68:326-337.
3. Vivas AC, Baaj AA, Benbadis SR, Vale FL. The health
care burden of patients with epilepsy in the United States: an analysis
of a nationwide database over 15 years. Neurosurg Focus. 2012;32:E1.
4. Lowenstein DH. Chapter 369. Seizures and epilepsy. In: Fauci AS, Kasper DL, Jameson JL, et al, eds. Harrison’s Principles of Internal Medicine.
18th ed. New York, NY: The McGraw Hill Companies; 2012.
www.accesspharmacy.com/content.aspx?aID=9145219. Accessed August 20,
2012.
5. Commission on Classification and Terminology of the
International League Against Epilepsy. Proposal for revised clinical and
electroencephalographic classification of epileptic seizures. Epilepsia. 1981;22:489-501.
6. Berg ET, Berkovic SF, Brodie MJ, et al. Revised
terminology and concepts for organization of seizures and epilepsies:
report of the ILAE Commission on Classification and Terminology,
2005–2009. Epilepsia. 2010;51:676-685.
7. Symptoms of a seizure. About epilepsy. www.epilepsy.com/101/ep101_symptom. Accessed December 5, 2012.
8. Mattson RH, Cramer JA, Collins JF, et al. Comparison of
carbamazepine, phenobarbital, phenytoin, and primidone in partial and
secondarily generalized tonic-clonic seizures. N Engl J Med. 1985;313:145-151.
9. Brodie MJ, Richens A, Yuen AW. Double-blind comparison
of lamotrigine and carbamazepine in newly diagnosed epilepsy. UK
Lamotrigine/Carbamazepine Monotherapy Trial Group. Lancet. 1995;345:476-479.
10. Chadwick DW, Anhut H, Greiner MJ, et al. A
double-blind trial of gabapentin monotherapy for newly diagnosed partial
seizures. International Gabapentin Monotherapy Study Group 945-77. Neurology. 1998;51:1282-1288.
11. Privitera MD, Brodie MJ, Mattson RH, et al.
Topiramate, carbamazepine and valproate monotherapy: double-blind
comparison in newly diagnosed epilepsy. Acta Neurol Scand. 2003;107:165-175.
12. Marson AG, Al-Kharusi AM, Alwaidh M, et al. The SANAD
study of effectiveness of carbamazepine, gabapentin, lamotrigine,
oxcarbazepine, or topiramate for treatment of partial epilepsy: an
unblinded randomised controlled trial. Lancet. 2007;369:1000-1015.
13. Marson AG, Al-Kharusi AM, Alwaidh M, et al. The SANAD
study of effectiveness of valproate, lamotrigine, or topiramate for
generalised and unclassifiable epilepsy: an unblinded randomised
controlled trial. Lancet. 2007;369:1016-1026.
14. Gluaser T, Ben-Menachem E, Bourgeois B, et al. ILAE
treatment guidelines: evidence-based analysis of antiepileptic drug
efficacy and effectiveness as initial monotherapy for epileptic seizures
and syndromes. Epilepsia. 2006;47:1094-1120.
15. French JA, Kanner AM, Bautista MD, et al. Efficacy and
tolerability of the new antiepileptic drugs. I: Treatment of new onset
epilepsy. Report of the Therapeutics and Technology Assessment
Subcommittee and Quality Standards Subcommittee of the American Academy
of Neurology and the American Epilepsy Society. Neurology. 2004;62;1252-1260.
16. Brodie MJ, Kwan P. Staged approach to epilepsy management. Neurology. 2002;58(suppl 5):S2-S8.
17. Fisher RS. Summary of antiepileptic drugs. Epilepsy.com. September 2009. www.epilepsy.com/epilepsy/newsletter/sept09/aeds. Accessed September 6, 2012.
18. Meador KJ. Cognitive and memory effects of new antiepileptic drugs. Epilepsy Res. 2006;68:63-67.
19. Lacy CF, Armstrong LL, Goldman MP, Lance LL. Drug Information Handbook 2011-2012. 20th ed. Hudson, OH: Lexi-Comp; 2011.
20. Rzany B, Correia O, Kelly JP, et al. Risk of
Stevens-Johnson syndrome and toxic epidermal necrolysis during first
weeks of antiepileptic therapy: a case-control study. Study Group of the
International Case Control Study on Severe Cutaneous Adverse Reactions.
Lancet. 1999;353:2190-2194.
21. Roujeau JC, Kelly JP, Naldi L, et al. Medication use and the risk of Stevens-Johnson or toxic epidermal necrolysis. N Engl J Med. 1995;333:1600-1607.
22. Suicidal behavior and ideation and antiepileptic
drugs. FDA. May 5, 2009.
www.fda.gov/Drugs/DrugSafety/PostmarketDrugSafetyInformationforPatientsandProviders/ucm100190.htm.
Accessed December 5, 2012.
23. Tomson T, Perucca E, Battino D. Navigating toward
fetal and maternal health: the challenge of treating epilepsy in
pregnancy. Epilepsia. 2004;45:1171-1175.
24. Diaz RA, Sancho J, Serratosa J. Antiepileptic drug interactions. Neurologist. 2008;14(suppl 1):S55-S65.
25. Patsalos PN, Berry DJ, Bourgeois BF, et al.
Antiepileptic drugs—best practice guideline for therapeutic drug
monitoring: a position paper by the subcommission on therapeutic drug
monitoring, ILAE Commission on Therapeutic Strategies. Epilepsia. 2008;49:1239-1276.
To comment on this article, contact rdavidson@uspharmacist.com.





