US Pharm. 2013;38(11):HS15-HS20.
ABSTRACT: Most antiepileptic drugs (AEDs) cause some degree of adverse drug reactions. Behavioral side effects (BSEs) associated with AEDs are often overlooked, but are a significant consideration. Agitation, aggression, psychosis, behavioral disorders, hyperactivity, and restlessness are some AED-related BSEs. Contributing causes may include pharmacologic activity, forced normalization, patient characteristics, individual susceptibility, and medication parameters such as dosage and drug interactions. The pharmacist must educate the patient and caregivers about possible BSEs in order to minimize the impact of behavioral changes and improve quality of life.
The goal of epilepsy treatment is to achieve a seizure-free state with minimal medication side effects. Information about expected or alarming side effects should always be communicated to patients. Although behavioral side effects (BSEs) are fairly common with antiepileptic drugs (AEDs), information in the pharmacy literature is sparse. BSEs associated with AEDs frequently are overlooked, but are an important consideration.
The first AED in clinical use—potassium bromide—was associated with psychiatric toxicities. Bromism, described as somnolence, psychosis, and delirium, has been extensively documented.1 Nearly all anticonvulsants cause some degree of cognitive, behavioral, or psychiatric adverse reactions. One of the newest agents, perampanel, has a black box warning regarding BSEs, including a 0.07% incidence of homicidal ideation.2 A literature review, however, reveals that this BSE may not be unique to perampanel. The purpose of this article is to review the literature on available AEDs and categorize their BSEs so that this information can be shared more effectively with patients and caregivers. Specifically, this article will focus on AED-related agitation, aggression, psychosis, behavioral disorders, hyperactivity, and restlessness.
Pharmacology of AEDs
The AEDs have various major mechanisms of action, including blockade of voltage-gated sodium ion (Na+) and calcium ion (Ca2+) channels; enhancement of gamma-aminobutyric acid (GABA)–ergic neurotransmission; inhibition of glutamatergic neurotransmission; and other mechanisms, such as modulation of synaptic vesicle proteins (levetiracetam), potassium ion (K+) channels (ezogabine), or carbonic anhydrase (topiramate and zonisamide).3 Most AEDs have multiple pharmacologic targets; thus, a number of activities contribute to their efficacy, as well as to their adverse effects.
Many AEDs block voltage-gated Na+ channels. Most AEDs delay the recovery of these channels from their fast-inactivated state, limiting the burst firing of neurons underlying epileptic seizures. One exception is lacosamide, which increases the number of Na+ channels in their slow-inactivated state, decreasing their availability for propagation of action potential.3 Drugs that act as Na+ channel blockers include carbamazepine, ethotoin, phenytoin, primidone, lacosamide, lamotrigine, oxcarbazepine, rufinamide, topiramate, zonisamide, valproic acid, and felbamate.3 Since a number of these drugs are associated with BSEs, it is likely that Na+ transport and homeostasis influence mood and behavior, and some evidence for this exists. Studies indicate that patients with affective disorders exhibit high plasma Na+ levels and that low Na+ diets have positive effects on mood.4,5 Furthermore, many other mood-stabilizing and antidepressant agents share the mechanism of Na+ channel blockade.6
Voltage-gated Ca2+ channels are another target for AEDs. Ethosuximide, valproic acid, lamotrigine, and zonisamide inhibit low voltage–activated T-type Ca2+ channels, which are implicated in absence seizures.3,7 Additionally, carbamazepine, phenobarbital, felbamate, gabapentin, lamotrigine, levetiracetam, oxcarbazepine, pregabalin, topiramate, zonisamide, and possibly phenytoin block high voltage–activated Ca2+ channels (L-, R-, P/Q-, and N-types), which are involved in neurotransmitter release.3,7 This mechanism may contribute to BSEs, since several studies indicate that Ca2+ homeostasis is important for mood and behavior. Notably, genetic variations in an L-type voltage-gated Ca2+ channel are associated with increased risk of bipolar disorder, depression, and schizophrenia.8 Furthermore, in some experimental models, Ca2+ channel blockers have antidepressant efficacy, and some antidepressants inhibit Ca2+ channel activity.9,10
Several AEDs enhance GABA-ergic neurotransmission by modulating GABAA receptors or by increasing GABA levels in the synaptic cleft. This mechanism is relevant for phenobarbital, clobazam, clonazepam, tiagabine, vigabatrin, primidone (via its metabolite, phenobarbital), valproic acid, felbamate, lamotrigine, topiramate, and zonisamide.3,7 GABA-ergic synapse dysfunction has been shown to contribute to aggressive behavior and schizophrenia.11,12
Inhibition of ionotropic glutamate receptors is another key mechanism for AEDs. Alpha-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid/kainate receptor antagonists include phenobarbital, lamotrigine, perampanel, and topiramate.3,7,13 Additionally, carbamazepine, valproic acid, felbamate, lamotrigine, and oxcarbazepine inhibit N-methyl-D-aspartate receptors.3,7 Recent evidence suggests that aberrant regulation of glutamatergic synapses may contribute to schizophrenia and, possibly, to mood disorders.14 In one study, plasma glutamate levels in patients with major depression and bipolar disorder were elevated compared with control levels.15
While the pharmacologic activities discussed above may cause the BSEs seen with some AEDs, another factor—forced normalization—may also contribute. In this phenomenon described in 1953 by Heinrich Landolt, patients’ electroencephalograms paradoxically normalized and seizure activity was inhibited during psychotic episodes.16 This idea is supported by epidemiologic studies that found lower seizure frequency in epileptic patients with psychosis, and studies citing the relatively few cases involving comorbid schizophrenia and epilepsy.17,18 In other words, AEDs may cause BSEs simply by suppressing seizure activity. The symptom most commonly associated with forced normalization is psychosis, but hypomania/mania, depression, and anxiety also have been described.19
Other Considerations
All AEDs have the potential to affect behavior. While this article focuses on medications as the likely cause of behavioral change, there are other potential factors to consider. The occurrence of BSEs may be related to seizure control (i.e., forced normalization).16 Patient characteristics and individual susceptibility also play a role in BSE incidence. Age, epilepsy type, and presence of a learning disability or other central nervous system or psychiatric disorder may also influence BSEs. Specific medication parameters, including dosage, titration rate, drug interactions, and effectiveness, also may be related to BSEs.20
Role of the Pharmacist
Based on the available data, it can be difficult to determine the true risk of psychiatric side effects with AEDs. The reported incidence of these behavioral reactions varies significantly in the literature. However, in general, AEDs with the highest incidence of aggressive-type behavior are topiramate, tiagabine, clobazam, levetiracetam, vigabatrin, and perampanel (TABLE 1). Psychosis, while reported much less commonly, is most frequently seen with zonisamide and topiramate (TABLE 2). Hyperactivity and restlessness have been reported most often with phenobarbital, clobazam, and vigabatrin (TABLE 3). Finally, the more generalized term “behavioral disorder” has been reported most frequently with topiramate, clobazam, and ethosuximide (TABLE 4).
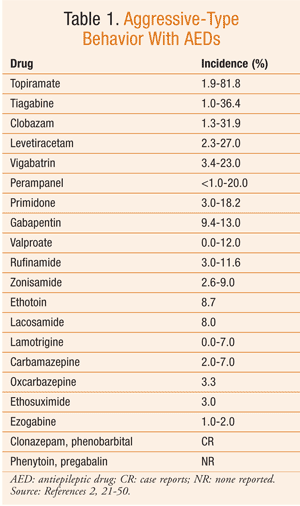
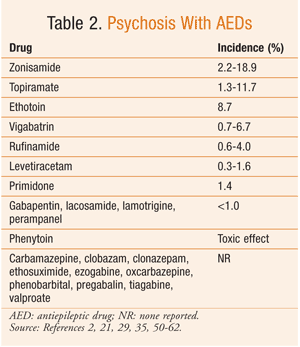
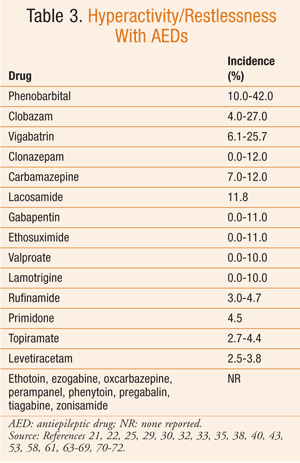
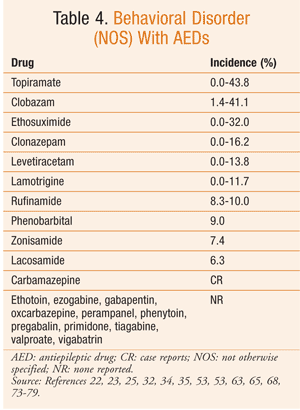
Therefore, especially with these medications, the pharmacist must effectively communicate possible BSEs to the patient and family or caregivers. This dialogue is crucial, since behavioral changes can result in a stressful environment, family instability, and other more serious consequences. Knowledge of these behaviors can minimize the impact of behavioral changes, lead to more rapid therapeutic adjustment, and improve patients’ and caregivers’ quality of life.
REFERENCES
1. Pearce JM. Bromide, the first effective antiepileptic agent. J Neurol Neurosurg Psychiatry. 2002;72:412.
2. Fycompa (perampanel) product information. Woodcliff Lake, NJ: Eisai Inc; October 2012.
3. Porter RJ, Dhir A, Madcdonald RL, Rogawski MA. Mechanisms of action of antiseizure drugs. Handb Clin Neurol. 2012;108:663-681.
4. Ramsey TA, Frazer A, Mendels J. Plasma and erythrocyte cations in affective illness. Neuropsychobiology. 1979;5:1-10.
5. Torres SJ, Nowson CA, Worsley A. Dietary electrolytes are related to mood. Br J Nutr. 2008;100:1038-1045.
6. El-Mallakh RS, Huff MO. Mood stabilizers and ion regulation. Harv Rev Psychiatry. 2001;9:23-32.
7. Perucca P, Mula M. Antiepileptic drug effects on mood and behavior: molecular targets. Epilepsy Behav. 2013;26:440-449.
8. Bhat S, Dao DT, Terrillion CE, et al. CACNA1C (Cav1.2) in the pathophysiology of psychiatric disease. Prog Neurobiol. 2012;99:1-14.
9. Galeotti N, Bartolini A, Ghelardini C. Blockade of intracellular
calcium release induces an antidepressant-like effect in the mouse
forced swimming test. Neuropharmacology. 2006;50:309-316.
10. Kim HJ, Kim TH, Choi SJ, et al. Fluoxetine suppresses synaptically induced [Ca2+]i spikes and excitotoxicity in cultured rat hippocampal neurons. Brain Res. 2013;1490:23-34.
11. Comai S, Tau M, Gobbi G. The psychopharmacology of aggressive behavior: a translational approach part 1: neurobiology. J Clin Psychopharmacol. 2012;32:83-94.
12. Stan AD, Lewis DA. Altered cortical GABA neurotransmission in schizophrenia: insights into novel therapeutic strategies. Curr Pharm Biotechnol. 2012;13:1557-1562.
13. Hanada T, Hashizume Y, Tokuhara N, et al. Perampanel: a novel,
orally active, noncompetitive AMPA-receptor antagonist that reduces
seizure activity in rodent models of epilepsy. Epilepsia. 2011;52:1331-1340.
14. Coyle JT, Basu A, Benneyworth M, et al. Glutamatergic synaptic dysregulation in schizophrenia: therapeutic implications. Handb Exp Pharmacol. 2012;213:267-295.
15. Altamura CA, Mauri MC, Ferrara A, et al. Plasma and platelet excitatory amino acids in psychiatric disorders. Amer J Psychiatry. 1993;150:1731-1733.
16. Landolt H. Psychic disorders in epilepsy. Clinical and electroencephalographic research. Dtsch Med Wochenschr. 1962;87:446-452.
17. Trimble MR. The Psychoses of Epilepsy. New York, NY: Raven Press; 1991.
18. Schmitz B, Trimble M. Epileptic equivalents in psychiatry: some 19th century views. Acta Neurol Scand Suppl. 1992;140:122-126.
19. Trimble MR, Schmitz B. Forced Normalization and Alternative Psychoses of Epilepsy. Petersfield, England: Wrightson Biomedical Press; 1998.
20. Austin JK, Caplan R. Behavioral and psychiatric comorbidities in pediatric epilepsy: toward an integrative model. Epilepsia. 2007;48:1639-1651.
21. Shehata GA, Bateh Ael-A, Hamed SA, et al. Neuropsychological
effects of antiepileptic drugs (carbamazepine versus valproate) in adult
males with epilepsy. Neuropsychiatr Dis Treat. 2009;5:527-533.
22. Silverstein FS, Parrish MA, Johnston MV. Adverse behavioral reactions in children treated with carbamazepine (Tegretol). J Pediatr. 1982;101:785-787.
23. Clobazam in treatment of refractory epilepsy: the Canadian
experience. A retrospective study. Canadian Clobazam Cooperative Group. Epilepsia. 1991;32:407-416.
24. Clobazam has equivalent efficacy to carbamazepine and phenytoin
as monotherapy for childhood epilepsy. Canadian Study Group for
Childhood Epilepsy. Epilepsia. 1998;39:952-959.
25. Commander M, Green SH, Prendergast M. Behavioural disturbances in children treated with clonazepam. Dev Med Child Neurol. 1991;33:362-363.
26. Chien J. Ethosuximide-induced mania in a 10-year-old boy. Epilepsy Behav. 2011;21:483-485.
27. Biton V, Gates JR, Ritter FJ, Loewenson RB. Adjunctive therapy for intractable epilepsy with ethotoin. Epilepsia. 1990;31:433-437.
28. Potiga (ezogabine) product information. Research Triangle Park, NC: GlaxoSmithKline; March 2012.
29. Khurana DS, Riviello J, Helmers S, et al. Efficacy of gabapentin therapy in children with refractory partial seizures. J Pediatr. 1996;128:829-833.
30. Lee DO, Steingard RJ, Cesena M, et al. Behavioral side effects of gabapentin in children. Epilepsia. 1996;37:87-90.
31. Wehner T, Bauer S, Hamer HM, et al. Six months of postmarketing
experience with adjunctive lacosamide in patients with pharmacoresistant
focal epilepsy at a tertiary epilepsy center in Germany. Epilepsy Behav. 2009;16:423-425.
32. Villari V, Rocca P, Frieri T, Bogetto F. Psychiatric symptoms
related to the use of lamotrigine: a review of the literature. Funct Neurol. 2008;23:133-136.
33. Beran RG, Gibson RJ. Aggressive behaviour in intellectually challenged patients with epilepsy treated with lamotrigine. Epilepsia. 1998;39:280-282.
34. Mula M, Trimble MR, Yuen A, et al. Psychiatric adverse events during levetiracetam therapy. Neurology. 2003;61:704-706.
35. Weintraub D, Buchsbaum R, Resor SR Jr, Hirsch LJ. Psychiatric and
behavioral side effects of the newer antiepileptic drugs in adults with
epilepsy. Epilepsy Behav. 2007;10:105-110.
36. Kothare SV, Khurana DS, Mostofi N, et al. Oxcarbazepine
monotherapy in children and adolescents: a single-center clinical
experience. Pediatr Neurol. 2006;35:235-239.
37. French JA, Krauss GL, Steinhoff BJ, et al. Evaluation of
adjunctive perampanel in patients with refractory partial-onset
seizures: results of randomized global phase III study 305. Epilepsia. 2013;54:117-125.
38. Wolf SM, Forsyth A. Behavior disturbance, phenobarbital, and febrile seizures. Pediatrics. 1978;61:728-731.
39. Vining EP, Mellitis ED, Dorsen MM, et al. Psychologic and
behavioral effects of antiepileptic drugs in children: a double-blind
comparison between phenobarbital and valproic acid. Pediatrics. 1987;80:165-174.
40. Briggs JN, Tucker J. Primidone (Mysoline) in the treatment of clinical petit mal in children. Lancet. 1954;266:19-21.
41. Herranz JL, Armijo JA, Arteaga R. Clinical side effects of
phenobarbital, primidone, phenytoin, carbamazepine, and valproate during
monotherapy in children. Epilepsia. 1988;29:794-804.
42. Coppola G, Grosso S, Franzoni E, et al. Rufinamide in children
and adults with Lennox-Gastaut syndrome: first Italian multicenter
experience. Seizure. 2010;19:587-591.
43. Banzel (rufinamide) product information. Woodcliff Lake, NJ: Eisai Inc; May 2013.
44. Sveinbjornsdottir S, Sander JW, Patsalos PN, et al.
Neuropsychological effects of tiagabine, a potential new antiepileptic
drug. Seizure. 1994;3:29-35.
45. Gabitril (tiagabine) product information. Frazer, PA: Cephalon, Inc; June 2012.
46. Glauser TA, Clark PO, McGee K. Long-term response to topiramate in patients with West syndrome. Epilepsia. 2000; 41(suppl 1): S91-S94.
47. Kelly K, Stephen LJ, Sills GJ, Brodie MJ. Topiramate in patients with learning disability and refractory epilepsy. Epilepsia. 2002;43:399-402.
48. Guberman A. Vigabatrin. Can J Neurol Sci. 1996;23(4 suppl 2):S13-S17.
49. Sabril (vigabatrin) product information. Cincinnati, OH: Patheon; February 2012.
50. Zonegran (zonisamide) product information. Woodcliff Lake, NJ: Elan Pharma International Ltd; January 2012.
51. Halász P, Kälviäinen R, Mazurkiewicz-Beldzinska M, et al.
Adjunctive lacosamide for partial-onset seizures: efficacy and safety
results from a randomized controlled trial. Epilepsia. 2009;50:443-453.
52. Brandt C, Fueratsch N, Boehme V, et al. Development of psychosis
in patients with epilepsy treated with lamotrigine: report of six cases
and review of the literature. Epilepsy Behav. 2007;11:133-139.
53. White JR, Walczak TS, Leppik IE, et al. Discontinuation of
levetiracetam because of behavioral side effects: a case-control study. Neurology. 2003;61:1218-1221.
54. Dodrill CB, Troupin AS. Neuropsychological effects of carbamazepine and phenytoin: a reanalysis. Neurology. 1991:41:141-143.
55. Mattson RH, Cramer JA, Collins JF, et al. Comparison of
carbamazepine, phenobarbital, phenytoin, and primidone in partial and
secondarily generalized tonic-clonic seizures. N Engl J Med. 1985;313:145-151.
56. Pålhagen S, Canger R, Henriksen O, et al. Rufinamide: a
double-blind, placebo-controlled proof of principle trial in patients
with epilepsy. Epilepsy Res. 2001;43:115-124.
57. Brodie MJ, Rosenfeld WE, Vazquez B, et al. Rufinamide for the
adjunctive treatment of partial seizures in adults and adolescents: a
randomized placebo-controlled trial. Epilepsia. 2009;50:1899-1909.
58. Gerber PE, Hamiwka L, Connolly MB, Farrell K. Factors associated
with behavioral and cognitive abnormalities in children receiving
topiramate. Pediatr Neurol. 2000;22:200-203.
59. Crawford P. An audit of topiramate use in a general neurology clinic. Seizure. 1998;7:207-211.
60. Sander JW, Hart YM, Trimble MR, Shorvon SD. Vigabatrin and psychosis. J Neurol Neurosurg Psychiatr. 1991;54:435-439.
61. Ferrie CD, Robinson RO, Panayiotopoulos CP. Psychotic and severe behavioural reactions with vigabatrin: a review. Acta Neurol Scand. 1996;93:1-8.
62. Miyamoto T, Kohsaka M, Koyama T. Psychotic episodes during zonisamide treatment. Seizure. 2000;9:65-70.
63. Sheth RD, Ronen GM, Goulden KJ, et al. Clobazam for intractable pediatric epilepsy. J Child Neurol. 1995;10:205-208.
64. Onfi (clobazam) product information. Deerfield, IL: Lundbeck; May 2013.
65. Kalachnik JE, Hanzel TE, Sevenich R, et al. Benzodiazepine
behavioral side effects: review and implications for individuals with
mental retardation. Am J Ment Retard. 2002;107:376-410.
66. Glauser TA, Cnaan A, Shinnar S, et al. Ethosuximide, valproic acid, and lamotrigine in childhood absence epilepsy. N Engl J Med. 2010;362:790-799.
67. Heyman E, Lahat E, Levin N, et al. Preliminary efficacy and safety of lacosamide in children with refractory epilepsy. Eur J Paediatr Neurol. 2012;16:15-19.
68. Bootsma HP, Ricker L, Hekster YA, et al. The impact of side
effects on long-term retention in three new antiepileptic drugs. Seizure. 2009;18:327-331.
69. Camfield CS, Chaplin S, Doyle AB, et al. Side effects of phenobarbital in toddlers; behavioral and cognitive aspects. J Pediatr. 1979;95:361-365.
70. Kim SH, Eun SH, Kang HC, et al. Rufinamide as an adjuvant treatment in children with Lennox-Gastaut syndrome. Seizure. 2012;21:288-291.
71. Coppola G, Caliendo G, Veggiotti P, et al. Topiramate as add-on
drug in children, adolescents and young adults with Lennox-Gastaut
syndrome: an Italian multicentric study. Epilepsy Res. 2002;51:147-153.
72. Lenz RA, Elterman RD, Robieson WZ, et al. Divalproex sodium in children with partial seizures: 12-month safety study. Pediatr Neurol. 2009;41:101-110.
73. Matthews-Ferrari K, Karroum N. Mania and anticonvulsant therapy. J Am Acad Child Adolesc Psychiatry. 1992;31:1168.
74. Guilhoto LM, Loddenkemper T, Gooty VD, et al. Experience with
lacosamide in a series of children with drug-resistant focal epilepsy. Pediatr Neurol. 2011;44:414-419.
75. Kluger G, Kurlemann G, Haberlandt E, et al. Effectiveness and
tolerability of rufinamide in children and adults with refractory
epilepsy: first European experience. Epilepsy Behav. 2009;14:491-495.
76. Mueller A, Boor R, Coppola G, et al. Low long-term efficacy and
tolerability of add-on rufinamide in patients with Dravet syndrome. Epilepsy Behav. 2011;21:282-284.
77. Topamax (topiramate) product information. Titusville, NJ: Janssen Pharmaceuticals, Inc; October 2012.
78. Sachdeo RC, Glauser TA, Ritter F, et al. A double-blind,
randomized trial of topiramate in Lennox-Gastaut syndrome. Topiramate YL
Study Group. Neurology. 1999;52:1882-1887.
79. Hirai K, Kimiya S, Tabata K, et al. Selective mutism and obsessive compulsive disorders associated with zonisamide. Seizure. 2002;11:468-470.
To comment on this article, contact rdavidson@uspharmacist.com.





