US Pharm. 2011;36(3):33-42.
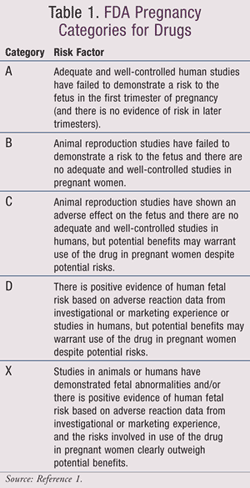
Pharmacists working with pregnant women must weigh the benefits of administering certain drugs and constantly keep in mind that medical compounds may transfer through the placenta and affect the fetus directly. The extent of the drugs’ transfer through the placenta varies with lipid solubility, molecular weight, acid dissociation constant ( pKa), and protein binding of the prescribed drugs. There are different pregnancy categories assigned to drugs based on their risks to the fetus, with Category A carrying the smallest risk and Category X the greatest risk (TABLE 1).1
This article will focus on the area of fetal medication management, whereby physicians prescribe drugs to pregnant women with the intention of addressing and controlling medical conditions associated with their unborn fetuses.
Birth defects of any type affect the lives of children, their mothers, and relatives, and may lead to significant physical disabilities later on in life. Presently, physicians are working diligently to identify problems before a child’s birth—in its fetal stage. Once the problem is uncovered, prevention and/or fetal treatment improve outcomes at the time of birth. Some fetal medical conditions can be corrected via surgical interventions, others with the help of drugs. Fetal medication management involves administration of drugs to pregnant patients with the goal of preventing or correcting certain abnormalities in their fetuses. This process relies upon a multidisciplinary approach—a team of obstetricians, maternal-fetal medicine specialists, geneticists, neonatologists, pediatric surgeons, and other pediatric subspecialists.2 The pediatric pharmacist should play a proactive role as well.
Physicians diagnose fetal patients by using different prenatal screening tests. These tests are done in every trimester as necessary and also during labor. Fetuses can develop different medical conditions; however, this article will focus only on the conditions that can be controlled or eliminated by administering medications to the mother carrying an ill fetus.
Neural Tube Defects
Approximately 1 out of 1,000 pregnancies will develop neural tube defects (NTDs).3,4 NTDs are prebirth defects that occur at a very early phase of the first trimester in pregnancy. The neural tube forms the baby’s spinal cord and brain and the bone surrounding them. NTDs occur primarily when the neural tube forms improperly and fails to close. If present, NTDs can cause miscarriages. If these babies are carried to term, they tend to have a very short life or they develop paralysis of the legs, dysfunction in the bowel and bladder, and hydrocephalus, in addition to learning disabilities.
Because of the devastating outcomes, including fatal complication with NTDs, much research has been conducted in this area. Published articles showed results that lead to a theory that occurrence of NTDs can be partially related to low levels of folic acid in the mother carrying the affected fetus. The CDC and the U.S. Public Health Service have developed the following recommendations to prevent the development of NTDs5:
1) All potential mothers should take folic acid supplements at a dose of 0.4 mg orally on a daily basis, starting with 1 month before the expected conception, and then continue taking it until the end of the first trimester.
2) If previous pregnancies were complicated by fetal NTDs, potential mothers should increase their intake to 4 mg of folic acid orally per day, for 1 month previous to the expected conception, until the end of the first trimester.
Pharmacist’s Role: Pharmacists should pay special attention to women who are taking drugs that can potentially lower levels of folic acid in their bodies, putting their fetus at a higher risk for development of NTDs in case of pregnancy.6 Antihistamines, sulfonamides, carbamazepine, phenytoin, primidone, phenobarbital, valproic acid, triamterene, trimethroprim, and pyrimethamine are considered to lower the levels and effects of folic acid in the human body. Studies show that out of the drugs mentioned above, carbamazepine and valproic acid are the most problematic, whereas phenytoin and phenobarbital are the least problematic in fetal development of NTDs.6-8 Presently, there is conflicting information on the effectiveness of folic acid at different doses for women taking drugs that lower levels of folic acid; however, prenatal diagnostic ultrasound studies of their fetuses are strongly recommended.9-11
Congenital Adrenal Hyperplasia
Another fetal disorder is congenital adrenal hyperplasia (CAH), which is a condition that involves a group of heritable disorders leading to the failure of cortisol synthesis in the adrenal cortex.12,13 The most frequent type (more than 90%) of CAH is the one associated with 21-hydroxy-lase (21-OH) enzyme deficiency. This type of CAH affects humans around the world, ranging from 1 in 12,000 to 1 in 15,000 live births.12,14 Inactivity of the 21-OH enzyme impairs the production of aldosterone and cortisol and causes accumulation of cortisol precursors and adrenal androgen. Insufficient levels of cortisol and aldosterone will promote a “salt wasting” disorder and an increase in the production of adrenal androgens such as testosterone and dihydrotestosterone.15-17 The 21-OH deficiency type of CAH affects females to a greater degree than males, owing to the fact that the major source of male androgens is the testes, whereas females normally synthesize adrenal androgens from progesterone.
Generally speaking, if left uncontrolled, CAH is expressed differently depending on the form of deficiency, varying from conditions in which females do not develop breasts or do not menstruate, to more complicated disorders such as virilization of female fetuses; however, all clinical presentations complicate patients’ normal social and sexual interactions.15,16
The 21-OH deficiency is diagnosed by measuring 17-hydroxyprogesterone and other steroid levels in amniotic fluid, or by molecular genetic methods.12 CAH was found to be associated with elevated release of corticotropin (ACTH) from the pituitary gland in response to low blood levels of cortisol. Furthermore, analysis of fetal development indicated that by the fourth week of gestation, the adrenal cortex is formed, which by the sixth or seventh week of gestation starts to secrete steroids. Therefore, the treatment goals would be administration of medication at the right time, aiming to decrease ACTH release in the prenatal body.16 Dexamethasone is known for lowering ACTH levels; furthermore, physicians choose dexamethasone to control CAH because of its ability to avoid the inactivation reaction by the 11-beta-hydroxy-steroid dehydrogenase placental enzyme. Medication is given to the mother as early as the seventh week of gestation. Treatment may be continued to term unless fetal karyotype is determined to be a male or a female whose DNA is not affected by CAH.12
Pharmacist’s Role: Pharmacists should be well aware of CAH occurrence and assist physicians in the medication management of such a complicated prenatal disorder. Furthermore, pharmacists should advise physicians of drugs that provoke ACTH release, such as levodopa, metoclopramide, and amphetamines. These agents should be used cautiously in pregnant patients, assessing the potential risks to the fetus against the benefits to the mother.18,19
Fetal Hyperthyroidism
Thyroid function in pregnant mothers influences the thyroid function of the fetus. About 1% to 5% of pregnant women diagnosed with Grave’s disease will have a fetus or neonate who develops hyperthyroidism due to the passive transplacental transport of maternal thyroid-stimulating hormone (TSH) receptor antibodies, if it occurred at approximately 20 weeks gestation or later.20,21 Furthermore, high maternal thyroid levels may lead to the development of fetal thyrotoxicosis.22 Fetal hyperthyroidism is characterized by premature labor, low birth weight, advanced bone maturation, and increased heart rate. If left untreated, a fatal cardiac failure can occur.
If hyperthyroidism is suspected, the fetus undergoes prenatal sonography, where its heart rate and intrauterine growth are analyzed. Furthermore, prenatal ultrasonography of the fetal thyroid gland can be used.23 If these studies prove to be inconclusive, fetal blood sampling can be performed as the last resort. Fetal blood sampling is taken percutaneously from the umbilical vein, which is considered to be risky due to its correlation with miscarriages. If fetal hyperthyroidism is confirmed, physicians usually initiate medical treatment. Use of drugs like propylthiouracil (PTU) and methimazole should be considered based on the trimester, at the smallest effective doses, to prevent fetal hypothyroidism.24-26
Pharmacist’s Role: Pharmacists should be aware of the appropriate pharmacologic treatment of fetal hyperthyroidism in pregnant mothers. Every physician should be warned against using radioiodine (I131) in pregnant patients. However, agents like PTU and methimazole would be a good choice, as both drugs cross the placenta and should be used accordingly based on the trimester and potential adverse effects to the fetus. Pharmacists should monitor patients’ response to therapy by checking TSH, 3,5,3'–triiodothyronine (T3), and thyroxine (T4) serum levels. In addition, patients’ medications and laboratory results should be studied to alert physicians about bone marrow suppression, liver status, and any types of bleeding and skin reactions possibly associated with the use of the drugs mentioned above.20,24,25 Pharmacists should also pay attention to other factors causing hyperthyroidism, including excess iodine in foods and exposure to drugs such as amiodarone and iodinated-contrast agents, and advise limiting or even avoiding their use.27
Fetal Hypothyroidism
Another fetal disorder associated with the function of the thyroid gland is fetal hypothyroidism. Fetal hypothyroidism occurs when the TSH level increases and the level of free T4 decreases in the fetal serum. Two nongenetic causes of fetal hypothyroidism are types that can be provoked either by the pregnant mother undergoing medical treatment for hyperthyroidism or iodine deficiency observed in the carrying mother. Fetal hypothyroidism is diagnosed using ultrasonography, and if that gives inconclusive results, the diagnosis can be further confirmed by analyzing fetal blood samples for TSH and T4 concentrations. If fetal hypothyroidism is left untreated, the fetus can suffer brain damage, and later on in life the child may experience social problems related to the neuropsychological harm and below average intellectual capabilities.28,29
Pharmacist’s Role: To improve the condition of the fetus diagnosed with hypothyroidism, the antithyroid medication of pregnant patients should be adjusted to the smallest and most effective dose based on the maternal TSH, T3, and T4 levels. Iodine supplements should be recommended only during the first or second trimester. If those regimens fail, additional weekly or every-other-week intra-amniotic injections of levothyroxine should be considered. The pharmacist’s key role is to identify and inform physicians about other agents that may produce drug-induced hypothyroidism such as hydrocortisone, amiodarone, lithium, propranolol, and sulfonylureas. These drugs should be adjusted to the smallest effective dose or the patient should be switched to another medication.30,31
Methylmalonic Acidemia
Methylmalonic acidemia is a metabolic defect state that is associated with a vitamin B12 deficiency. This condition is described as being provoked by the high levels of methylmalonic acid as a result of genetically produced malfunction of methylmalonyl-CoA mutase or failure in the formation of its coenzyme adenosylcobalamin via vitamin B12 metabolism. Diagnosis of this condition can be drawn from the presence of methylmalonic acid levels either in amniotic fluid or in the pregnant woman’s urine.2,32,33
Pharmacist’s Role: Pharmacists should be aware of any drugs that can potentially lower the body’s supply of vitamin B12. Medications such as colchicine and metformin, as well as histamine2 blockers and proton pump inhibitors when used for extended periods, have the potential to lower vitamin B12 levels in the blood. Their use should be called to the attention of prescribing physicians when these drugs are ordered for pregnant patients who are carrying a fetus diagnosed with methylmalonic acidemia.34-37
Lung Maturity Induction
In the case of a birth at the gestational age of less than 34 weeks, the neonate will most likely be delivered with immature lungs. Such lungs are not capable of performing gas exchange due to the lack of surfactants in the alveolar spaces. This condition is dangerous to a newborn and, if not detected and treated prenatally, may cause a respiratory distress syndrome, leading to a life-threatening situation. Physicians usually diagnose this condition by measuring the concentration of surfactant in the amniotic fluid.38,39
Pharmacist’s Role: When the diagnosis of fetal premature lungs is confirmed, corticosteroid therapy is given to the pregnant woman to stimulate the synthesis and release of surfactants in the alveolar spaces, to raise beta-receptor density in the fetal lungs, and to promote structural maturation of the fetal lungs. Out of the therapeutically available corticosteroid group, based on clinical efficiency and safety profile, the National Institute of Health Consensus Conference recommends using betamethasone at a prescribed schedule of 12 mg intramuscularly (IM) every 24 hours for 2 doses.2,39 Pharmacists should follow up and confirm the number of prescribed and scheduled doses of betamethasone. Clinical literature shows that multiple courses of antenatal corticosteroidal therapy produce no extra benefits; however, this therapy is associated with decreased fetal growth in utero in addition to long-term adverse events.40,41
Fetal Arrhythmias
Generally speaking, fetal heart conductivity develops around 16 weeks of gestation. Normal fetal heart rate at 30 weeks of gestation is 120 to 160 beats per minute (bpm). By the 40th week, heart beat values drop to 110 to 150 bpm. Any values outside of the normal range are considered to put the affected fetus into a category of potential fetal arrhythmia. In such cases, further investigation and analysis are done to identify and confirm a type of fetal arrhythmia. In the majority of cases, fetal activity does not change and the carrying mother does not show any symptoms associated with arrhythmia. Instruments and techniques like two-dimensional ultrasound, M-mode pulsed Doppler, and tissue Doppler imaging are used to exclude heart rate change associated with possible hypoxia and to confirm and identify a type of fetal heart misconductivity.42-44
Pharmacist’s Role: Medication management of fetal arrhythmias is done by administering drugs to either the carrying mother or the fetus directly. Fetal supraventricular tachycardias (SVTs) are the most frequent type of tachyarrhythmias. Digoxin is primarily the drug of choice for management of fetal SVT. Digoxin should be given first orally or intravenously to the pregnant woman, targeting the serum level between 1 and 2 ng/mL. Literature shows that digoxin can be given directly to the fetus. If SVT is not under control by that time, other drugs can play a role as second-line medications, such as amiodarone, sotalol, and flecainide, and might be necessary as a part of medication management.
Fetal ventricular tachycardias (VTs) can be controlled by agents such as propranolol, procainamide, and lidocaine. Pharmacists should advise physicians to avoid digoxin due to its potential to worsen fetal VT.42-44
Conclusion
Pharmacists play a vital role in fetal medication management. They must verify every drug prescribed to a pregnant woman and work diligently to identify the risks to the mother and her fetus and recognize drug interactions associated with these medications. Micromedex, Lexicomp, and Drugs in Pregnancy and Lactation are excellent resources for additional drug information.
Presently, pharmacists work together with a multi-disciplinary team of prenatal specialists to ensure the safety of both the pregnant woman and the fetus. Pharmacists must acknowledge the reasons for the fetal treatment and monitor the treatment plan accordingly to ensure that all women in their care deliver healthy babies.
REFERENCES
1. FDA pregnancy categories. www.drugs.com/pregnancy-
2. Springer SC. The fetus as a patient, prenatal diagnosis and fetal therapy. eMedicine. http://emedicine.medscape.com/
3. U.S. Preventive Services Task Force. Folic acid for the prevention of neural tube defects: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2009;150:626-631.
4. American Academy of Pediatrics. Committee on Genetics. Folic acid for the prevention of neural tube defects. Pediatrics. 1999;104:325-327.
5. Folic acid. Recommendations. CDC. www.cdc.gov/ncbddd/folicacid/
6. Hernandez-Diaz S, Werler MM, Walker AM, Mitchell AA. Folic acid antagonists during pregnancy and the risk of birth defects. N Engl J Med. 2000;343:1608-1613.
7. Hernandez-Diaz S, Werler MM, Walker AM, Mitchell AA. Neural tube defects in relation to use of folic acid antagonists during pregnancy. Am J Epidemiol. 2001;153:961-968.
8. Wolff T, Takacs Witkop C, Miller T, Syed SB. Folic acid supplementation for the prevention of neural tube defects: an update of the evidence for the U.S. Preventive Services Task Force. Rockville, MD: Agency for Healthcare Research and Quality; May 2009. AHRQ pub no. 09-05132-EF-1.
9. Burry J. Preventing neural tube defects with folic acid in pregnancy. CPJ/RPC. 2008;141:90-94.
10. Yerby MS. Management issues for women with epilepsy: neural tube defects and folic acid supplementation. Neurology. 2003;61(suppl 2):S23-S26.
11. Walling AD. Folic acid for the prevention of neural tube defects. Am Fam Physician. September 15, 1998. www.aafp.org/afp/980915ap/
12. Nimkarn S, New MI. Prenatal diagnosis and treatment of congenital adrenal hyperplasia. Horm Res. 2007;67:53-60.
13. Miller WL. Prenatal treatment of congenital adrenal hyperplasia: a promising experimental therapy of unproven safety. Trends Endocrinol Metab. 1998;9:290-293.
14. American Academy of Pediatrics, Section on Endocrinology and Committee on Genetics. Technical report: congenital adrenal hyperplasia. Pediatrics. 2000;106:1511-1518.
15. Wilson TA. Congenital adrenal hyperplasia. eMedicine. November 18, 2009. www.emedicine.com. Accessed March 14, 2010.
16. Dalls J. Prenatal treatment of congenital adrenal hyperplasia. Newborn screening case management. Department of State Health Services, Texas. Updated 1998.
17. Lajic S, Nordenstrom A, Ritzen EM, Wedell A. Prenatal treatment of congenital adrenal hyperplasia. European J Endocrinol. 2004;151:U63-U69.
18. Moore E. Adrenal insufficiency. Acquired and autoimmune adrenal failure. Suite101. May 1, 2006. http://autoimmunedisease.
19. ACTH. American Association for Clinical Chemistry. March 30, 2010. www.labtestsonline.org/
20. Perros P. Thyrotoxicosis and pregnancy. PLoS Med. 2005;2:1248-1252.
21. Lee SP, Webster J, Thomas WEG. Treatment of thyrotoxicosis. Surgery. 2003;12:293-296.
22. Anselmo J, Cao D, Karrison T, et al. Fetal loss associated with excess thyroid hormone exposure. JAMA. 2004;292:691-695.
23. Luton D, Le Gac I, Vuillard E, et al. Management of Graves’ disease during pregnancy: the key role of fetal thyroid gland monitoring. J Clin Endocrinol Metab. 2005;90:6093-6098.
24. Ross RS. Diagnosis and treatment of hyperthyroidism during pregnancy. UpToDate. www.uptodate.com. Accessed April 1, 2010.
25. Patil-Sisodia K, Mestman JH. Graves hyperthyroidism and pregnancy: a clinical update. Medscape. www.medscape.com. Accessed April 1, 2010.
26. Porreco RP, Bloch CA. Fetal blood sampling in the management of intrauterine thyrotoxicosis. Obstet Gynecol. 1990;76:509-512.
27. Reid JR, Wheeler SF. Hyperthyroidism: diagnosis and treatment. Am Fam Physician. 2005;72:623-630.
28. Ghidini A. Fetal blood sampling: indications and invasive fetal therapy. UpToDate. www.uptodate.com. Accessed April 9, 2010.
29. Smallridge RC, Ladenson PW. Hypothyroidism in pregnancy: consequences to neonatal health. J Clin Endocrinol Metabol. 2001;86:2349-2353.
30. Goldberg JF. Management of hypothyroidism in patients on lithium prophylaxis for bipolar disorder. Medscape. www.medscape. Accessed April 11, 2010.
31. Ogunyemi DA. Autoimmune thyroid disease and pregnancy. eMedicine. www.emedicine.com. Accessed April 11, 2010.
32. Bodamer OA. Methylmalonic acidemia. eMedicine. http://emedicine.medscape.com/
33. Shigematsu Y, Hata I, Nakai A, et al. Prenatal diagnosis of organic acidemias based on amnioric fluid levels of acycarnitines. Pediatr Res. 1996;39:680-684.
34. Vitamin B12. Micromedex. www.micromedex.com. Accessed February 7, 2011.
35. Oh R, Brown DL, Vitamin B12 deficiency. Am Fam Physician. 2003;67:979-986.
36. Schrier SL. Etiology and clinical manifestation of vitamin B12 and folic acid deficiency. UpToDate. www.uptodate.com. Accessed February 7, 2011.
37. Webb DI, Chodos RB, Mahar CQ, Faloon WW. Mechanism of vitamin B12 malabsorption in patients receiving colchicines. N Engl J Med. 1968:279:845-850.
38. Briggs GG, Wan SR. Drug therapy during labor and delivery, part 1: fetal lung immaturity. Medscape. www.medscape.com. Accessed May 1, 2010.
39. Lee MJ, Guinn D. Antenatal use of glucocorticoids in women at risk for preterm delivery. UpToDate. www.uptodate.com. Accessed May 1, 2010.
40. Bonanno C, Fuchs K, Wapner RJ. Single versus repeat course of antenatal steroids to improve neonatal outcomes: risks and benefits. Obstet Gynecol Surv. 2007;62:261-271.
41. Murphy KE, Hannah ME, Willan AR, et al. Multiple courses of antenatal steroids for preterm birth (MACS): a randomized controlled trial. Lancet. 2008;372:2143-2151.
42. Krapp M, Kohl T, Simpson JM, et al. Review of diagnosis, treatment, and outcome of fetal atrial flutter compared with supraventricular tachycardia. Heart. 2003;89:913-917.
43. Zaidi AN, Ro PS. Treatment of fetal and neonatal arrhythmias. US Pediatrics. 2008;4:27-29.
44. Levine JC. Diagnosis and treatment of fetal cardiac arrhythmias. UpToDate. www.uptodate.com. Accessed April 28, 2010.
To comment on this article, contact rdavidson@uspharmacist.com.





