US Pharm. 2017;42(6):HS-21-HS-28.
ABSTRACT: Chronic obstructive pulmonary disease (COPD) is a preventable disease in which persistent respiratory symptoms and airflow limitations worsen with time. COPD is the fourth leading cause of death in the world. The treatment of COPD includes various inhaled therapies as well as preventive measures, such as smoking cessation and immunizations. Patient education is crucial to the management of COPD. Medication therapy management (MTM) can play a role in helping patients learn how to manage COPD and decrease complications. Successful MTM interventions can improve patient care and provide a positive impact on the star measures of both pharmacies and healthcare plans.
Medication therapy management (MTM) services have demonstrated impact in a number of areas, including cardiovascular diseases, mental health, and transitions of care.1 MTM services are defined as a service or group of services that optimize therapeutic outcomes for individual patient and include activities such as immunizations, disease state coaching, and medication therapy reviews.2 The purpose of this article is to provide the pharmacist with a brief overview of chronic obstructive pulmonary disease (COPD) and identify areas within an MTM encounter the community pharmacist can capitalize on to improve management of this disease state.
COPD is currently the fourth leading cause of death in the world, with more than 3 million deaths in 2012, and is projected to become the third leading cause by 2020.3,4 It is a preventable disease characterized by persistent respiratory symptoms and airflow limitations, which worsen progressively. The primary risk factor for developing COPD is tobacco smoke, but additional risk factors include exposure to biomass fuel and air pollution.3 Abnormal lung development and accelerated aging may also contribute to development of COPD. The Centers for Medicare and Medicaid Services is evaluating requirements for healthcare facilities, including pharmacies, in order to adequately manage this disease to prevent or reduce the severity of exacerbations.5 A tremendous social and economic burden is associated with the management of COPD, and a direct relationship exists between the disease severity and cost of care.6-8 In the United States, direct and indirect costs of COPD exceed $50 billion. Exacerbations account for the greatest portion of this cost.9 Patient education is crucial to the management of COPD, and MTM can play a role in helping patients learn how to manage COPD and decrease exacerbations and severity.3 These services provide opportunities for counseling, including emphasizing awareness of preventive therapies such as smoking cessation and the different aids available for cessation; assessing inhaler technique to ensure optimal drug delivery; determining adherence to the treatment regimen; educating on the signs and symptoms of infection, such as early signs of pneumonia; and determining when a visit to the physician is warranted. MTM has the potential to play a significant role in managing this costly disease.3
Highlighting COPD During an MTM Encounter
A diagnosis of COPD should be considered in patients presenting with dyspnea, chronic cough or sputum production, and/or a history of exposure to risk factors.10 Cough, either productive or nonproductive, is often the first noticeable symptom, yet it is frequently discounted.11 Dyspnea causes disability in most patients.12,13 Although symptoms may indicate COPD, spirometry is required to confirm the diagnosis.10 Community pharmacists have been shown to be capable of screening and identifying patients that have not yet been diagnosed with COPD. Screening consisted of assessing smoking and signs and symptoms consistent with COPD. Patients found to be at high risk were offered spirometry testing in the pharmacy and referred to a primary care physician for confirmation of diagnosis.14 Use of a quick screening tool to identify patients at risk for COPD but not yet diagnosed can easily be incorporated into an MTM encounter. The pharmacist could then reasonably offer spirometry testing if available in addition to disease-state education and the need for further evaluation.
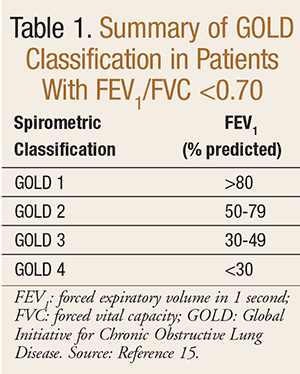
Effectiveness and appropriateness of therapy should also be evaluated during the encounter. Determining which therapies may be appropriate for a patient depends on the classification of the patient’s disease. Disease classification takes into account spirometry, patient symptoms, and exacerbation history.3 Classification based upon spirometry results is outlined in TABLE 1. Two questionnaires are currently recommended to assess symptom prevalence: the Modified British Medical Research Council (mMRC) test or the COPD assessment test (CAT).15 The mMRC determines breathlessness and adequately assesses symptoms and predicts future mortality risk.16 The second questionnaire, CAT, determines health status impairment in COPD and assesses more than breathlessness.17 The mMRC along with the CAT score is used to stage the severity of a patient’s COPD (FIGURE 1). (For more information on each of these questionnaires, the reader is referred to References 16 and 17.)

Managing COPD Treatment Modalities
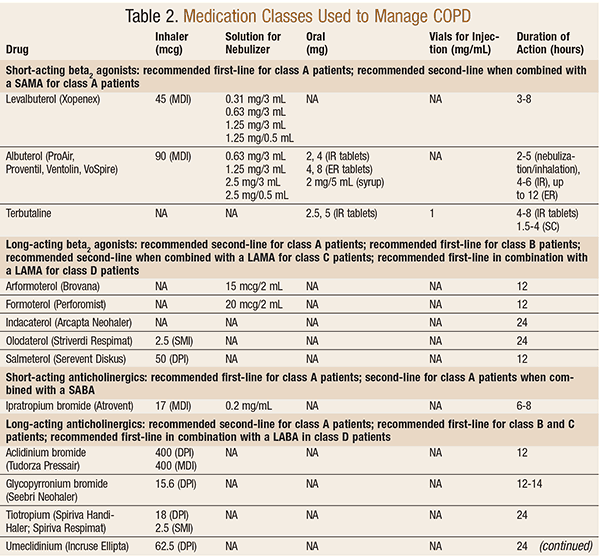
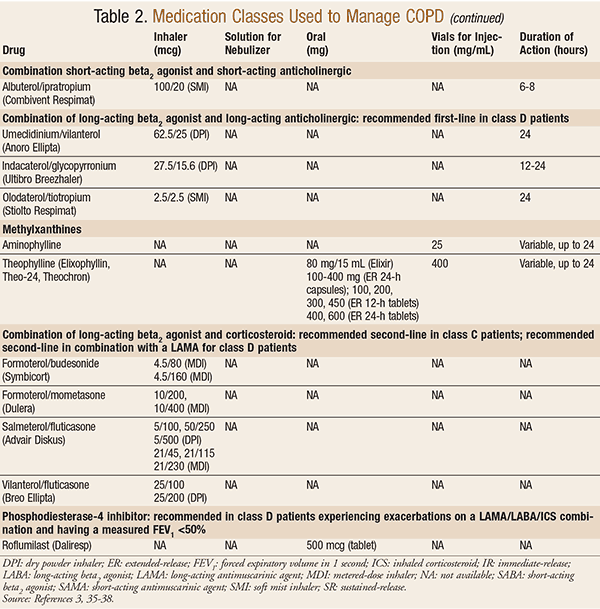
The treatment of COPD includes various inhaled therapies as well as preventive measures, such as smoking cessation and immunizations. The main classes of medications used to treat COPD are short-acting beta agonists (SABA), long-acting beta agonists (LABA), short-acting antimuscarinic agents (SAMA), long-acting antimuscarinic agents (LAMA), inhaled corticosteroids (ICS), phosphodiesterase-4 inhibitors (PD4), and methylxanthines.3 The available dosage forms, duration of action, and classifications these agents are recommended for are outlined in TABLE 2.
Determining a patient’s immunization status is frequently a component of MTM services. The influenza and pneumococcal vaccines are recommended in patients with COPD. The influenza vaccine can reduce serious illness and even death in COPD patients if the influenza virus is contracted in this patient population.18-20 Studies have shown a significant reduction in exacerbations among vaccinated subjects compared with those who received placebo.18 Patients may receive either the inactive or live-attenuated vaccine. The pneumococcal vaccine is also recommended for patients with COPD. In patients age <65 years with an FEV1 less than 40%, the 23-valent pneumococcal polysaccharide vaccine (PPSV23) is recommended, as studies have demonstrated a reduction in the incidence of community-acquired pneumonia in this patient population.21 The 13-valent conjugated pneumococcal vaccine (PCV13) is recommended for patients aged >65 years. In a large randomized, controlled clinical trial, the PCV13 demonstrated significant efficacy for the prevention of community-acquired pneumonia in patients aged >65 years.22
Assessing Inhaler Technique
An MTM encounter is a prime opportunity to evaluate a patient’s inhaler administration technique. Several articles have demonstrated that improper inhaler use is a prevalent issue.23-25 Pharmacists have been able to provide focused education initiatives to improve and sustain a patient’s technique.23,26,27 An additional review of 10 studies evaluating the impact of community pharmacists in the management of COPD confirmed that pharmacist intervention can not only improve inhaler technique but improve medication adherence as well.28 Improving a patient’s inhaler technique has been shown in one trial to not only improve adherence but to also decrease exacerbations.29 One study hypothesizes that inhaler adherence can be improved after a disease state–specific intervention. This intervention included a medication review, education, a COPD action plan, a pulmonary rehabilitation referral, tobacco-cessation counseling, and a referral to a community-based self-management program.30 These interventions are likely to be effective and can easily be incorporated into the medication action plan provided to the patient at the end of an MTM session. Provision of an action plan for the patient with telephone follow-up can also reduce emergency room visits and hospitalizations. This benefit is coupled with an increased utilization of corticosteroids and antibiotics.31
Lastly, inquiring about the patient’s tobacco use is a reasonable query to include in all patients’ MTM encounters, not just in those with COPD. However, for patients with COPD, addressing tobacco use has the greatest potential to impact disease progression.3 A review of 10 studies has shown that pharmacist-delivered interventions can positively influence successful quit attempts.32 Even brief encounters in which the pharmacist uses the ask-advise-refer method have been shown to be effective.33
These interventions, while not in the context of an MTM session, have been shown to improve patients’ health-related quality of life as well.34 The MTM session can be managed in such a way that incorporation of these interventions may be accomplished, resulting in improvement in patient outcomes.
Conclusion
In summary, COPD is a disease that benefits from various interventions made by pharmacists. Decreasing or preventing COPD exacerbations, in turn, decreases healthcare costs and social burdens patients may face. Pharmacists can perform mMRC and CAT questionnaires and interview patients as well to determine exacerbation history and staging. Once the disease is properly staged, the patient’s current therapeutic regimen can be reviewed for appropriateness and symptom control. Other interventions include informing a physician that a particular inhaler is not providing relief for the patient, ensuring that a patient is up-to-date on vaccinations, and assessing a patient’s willingness to quit smoking. Pharmacists can also provide education on inhaler technique to ensure proper drug delivery as well as education on when a visit to the physician is warranted if a patient continuously experiences exacerbations. Pharmacists can reasonably complete these activities during an MTM encounter and, with successful interventions, improve patient care.
REFERENCES
1. American Pharmacists Association. Pharmacist-delivered patient care services evidence examples. www.pharmacist.com/sites/default/files/EvidenceforPharmacistsServices2000-
2016.pdf. Accessed May 11, 2017.2. American Pharmacists Association. APhA MTM Central. www.pharmacist.com/node/29279?is_sso_called=1. Accessed May 11, 2017.3. Agusti A, Decramer M, Celli B, et al. From the Global Strategy for the Diagnosis, Management and Prevention of COPD, Global Initiative for Chronic Obstructive Lung Disease (GOLD) 2017. http://goldcopd.org.4. Mathers CD, Loncar D. Projections of global mortality and burden of disease from 2002 to 2030. PLoS Med. 2006;3(11):e442.5. CMS: Centers for Medicare & Medicaid Services. Medicare 2017 Part C & D Display Measure Technical Notes. Center for Medicare. Updated December 19, 2016. www.cms.gov/Medicare/Prescription-DrugCoverage/PrescriptionDrugCovGenIn/PerformanceData.html. Accessed May 12, 2017.6. Lozano R, Naghavi M, Foreman K, et al. Global and regional mortality from 235 causes of death for 20 age groups in 1990 and 2010: a systematic analysis for the Global Burden of Disease Study 2010. Lancet. 2012;380(9859):2095-2128.7. Vos T, Flaxman AD, Naghavi M, et al. Years lived with disability (YLDs) for 1160 sequelae of 289 diseases and injuries 1990-2010: a systematic analysis for the Global Burden of Disease Study 2010. Lancet. 2012;380(9859):2163-2196.8. Murray CJ, Lopez AD. Alternative projections of mortality and disability by cause 1990-2020: Global Burden of Disease Study. Lancet. 1997;349(9064):1498-1504.9. Guarascio AJ, Ray SM, Finch CK, Self TH. The clinical and economic burden of chronic obstructive pulmonary disease in the USA. Clinicoecon Outcomes Res. 2013;5:235-245.10. Buist AS, McBurnie MA, Vollmer WM, et al. International variation in the prevalence of COPD (the BOLD Study): a population-based prevalence study. Lancet. 2007;370(9589):741-750.11. Cho SH, Lin HC, Ghoshal AG, et al. Respiratory disease in the Asia-Pacific region: cough as a key symptom. Allergy Asthma Proc. 2016;37(2):131-140.12. Miravitlles M, Worth H, Soler Cataluna JJ, et al. Observational study to characterise 24-hour COPD symptoms and their relationship with patient-reported outcomes: results from the ASSESS study. Respir Res. 2014;15:122.13. Elliott MW, Adams L, Cockcroft A, et al. The language of breathlessness. Use of verbal descriptors by patients with cardiopulmonary disease. Am Rev Respir Dis. 1991;144(4):826-832.14. Castillo D, Burgos F, Guayta R, et al. Airflow obstruction case finding in community-pharmacies: a novel strategy to reduce COPD underdiagnosis. Respir Med. 2015;109:475-482.15. Han MK, Muellerova H, Curran-Everett D, et al. GOLD 2011 disease severity classification in COPDGene: a prospective cohort study. Lancet Respir Med. 2013;1(1):43-50.16. Fletcher CM. Standardised questionnaire on respiratory symptoms: a statement prepared and approved by the MRC Committee on the Aetiology of Chronic Bronchitis (MRC breathlessness score). BMJ. 1960;2:1662.17. Jones PW, Harding G, Berry P, et al. Development and first validation of the COPD Assessment Test. Eur Respir J. 2009;34(3):648-654.18. Poole PJ, Chacko E, Wood-Baker RW, Cates CJ. Influenza vaccine for patients with chronic obstructive pulmonary disease. Cochrane Database Syst Rev. 2006;(1):CD002733.19. Nichol KL, Margolis KL, Wuorenma J, Von Sternberg T. The efficacy and cost effectiveness of vaccination against influenza among elderly persons living in the community. N Engl J Med. 1994;331(12):778-784.20. Fiore AE, Shay DK, Broder K, et al. Prevention and control of seasonal influenza with vaccines: recommendations of the Advisory Committee on Immunization Practices (ACIP), 2009. MMWR Recomm Rep. 2009;58(RR-8):1-52.21. Alfageme I, Vazquez R, Reyes N, et al. Clinical efficacy of anti-pneumococcal vaccination in patients with COPD. Thorax. 2006;61(3):189-195.22. Bonten MJ, Huijts SM, Bolkenbaas M, et al. Polysaccharide conjugate vaccine against pneumococcal pneumonia in adults. N Engl J Med. 2015; 372(12):1114-1125.23. Hämmerlein A, Müller U, Schulz M. Pharmacist-led intervention study to improve inhalation technique in asthma and COPD patients. J Eval Clin Prac. 2011;17:61-70.24. Pothirat C, Chaiwong W, Phetsuk N, et al. Evaluating inhaler use technique in COPD patients. Int J Chron Obstruct Pulmon Dis. 2015;10:1291-1298.25. Melani AS, Bonavia M, Cilenti V, et al. Inhaler mishandling remains common in real life and is associated with reduced disease control. Respir Med. 2011;105(6):930-938.26. Bouwmeester C, Kraft J, Bungay KM. Optimizing inhaler use by pharmacist-provided education to community-dwelling elderly. Respir Med. 2015;109:1363-1368.27. Axtell S, Haines S, Fairclough J. Effectiveness of Various Methods of Teaching Proper Inhaler Technique: the Importance of Pharmacist Counseling. J Pharm Prac. 2017;30(2):195-201.28. Hesso I, Gebara SN, Kayyali R. Impact of community pharmacists in COPD management: Inhalation technique and medication adherence. Respir Med. 2016;118:22-30.29. Takemura M, Mitsui K, Ido M, et al. Effect of a network system for providing proper inhalation technique by community pharmacists on clinical outcomes in COPD patients. Int J Chron Obstruct Pulmon Dis. 2013;8:239-244.30. Davis E, Marra C, Gamble JM, et al. Effectiveness of a pharmacist-driven intervention in COPD (EPIC): study protocol for a randomized controlled trial. Trials. 2016;17:502.31. Howcroft M, Walters EH, Wood-Baker R, Walters JA. Action plans with brief patient education for exacerbations in chronic obstructive pulmonary disease. Cochrane Database Syst Rev. 2016;12:CD005074.32. Mdege ND, Chindove S. Effectiveness of tobacco use cessation interventions delivered by pharmacy personnel: a systematic review. Res Social Adm Pharm. 2014;10(1):21-44.33. Patwardhan PD, Chewning BA. Effectiveness of intervention to implement tobacco cessation counseling in community chain pharmacies. J Am Pharm Assoc. 2003;52(4):507-514.34. Suhaj A, Manu MK, Unnikrishnan MK, et al. Effectiveness of clinical pharmacist intervention on health-related quality of life in chronic obstructive pulmonary disorder patients—a randomized controlled study. J Clin Pharm Ther. 2016;41:78-83.35. Appleton S, Poole P, Smith B, et al. Long-acting beta2-agonists for poorly reversible chronic obstructive pulmonary disease. Cochrane Database Syst Rev. 2006;3(3):CD001104.36. Barr RG, Bourbeau J, Camargo CA, Ram FS. Inhaled tiotropium for stable chronic obstructive pulmonary disease. Cochrane Database Syst Rev. 2005;(2):CD002876.37. Vogelmeier C, Hederer B, Glaab T, et al. Tiotropium versus salmeterol for the prevention of exacerbations of COPD. N Engl J Med. 2011;364(12):1093-1103.38. Decramer ML, Chapman KR, Dahl R, et al. Once-daily indacaterol versus tiotropium for patients with severe chronic obstructive pulmonary disease (INVIGORATE): a randomised, blinded, parallel-group study. Lancet Respir Med. 2013;1(7):524-533.
To comment on this article, contact rdavidson@uspharmacist.com.





