US Pharm. 2012;37(3):61-63.
For those who have lost the attachment to a loved one’s mind suffering from Alzheimer’s disease, there is hardly anything more devastating. This spouse, mother, father, sister, brother, or friend that you have known and loved slowly but distinctly loses touch with reality, and the day of reckoning comes when you see this person and they do not know who you are anymore. And the “good days” when the illness retreats and lets memories rise, if only for a while, never seem to make up for the “bad days” when you are just another figure in this person’s life.
The hardship of this demoralizing malady is something that almost everyone with this experience wants to rile against it with all the fervor and frustration of Dylan Thomas’s famous poem written while his own father was dying: “Do not go gentle into that good night…Rage, rage against the dying of the light.”1
The statistics are not very encouraging. Today about 4.5 million Americans have some form of advanced neurodegeneration, and there are estimates that by 2050 the disease state will affect as many as 14 million people. Worse, Alzheimer’s disease is said to affect 1 out of 4 people over age 75 years, 1 out of 3 people over age 80, and nearly half of those over age 85. The Alzheimer’s Drug Discovery Foundation estimates that this disorder costs the United States over $183 million annually, just slightly behind the most expensive diseases—cancer and cardiac complications.2
FDA-Approved Treatments
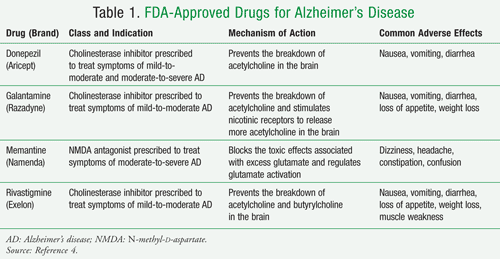
Drugs that have been approved by the FDA have been divided into categories that refer to the degree of Alzheimer’s disease: mild-to-moderate or moderate-to-severe. For mild-to-moderate Alzheimer’s disease, cholinesterase inhibitors such as donepezil (Aricept; also approved for moderate-to-severe disease), galantamine (Razadyne), and rivastigmine (Exelon) are prescribed. According to the National Institute on Aging, “these drugs may help delay or prevent symptoms from becoming worse for a limited time and may help control some behavioral symptoms.”3The N-methyl- D-aspartate (NMDA) antagonist memantine (Namenda) is used to treat moderate-to-severe Alzheimer’s disease. Its main effect is to delay progression of symptoms, which allows patients to maintain certain daily functions (e.g., ability to use the bathroom independently) for a little longer than they would without the drug. Memantine regulates glutamate, an important brain chemical that in excessive amounts may lead to brain cell death. NMDA antagonists and cholinesterase inhibitors can be used in combination because they have very different mechanisms of action (TABLE 1).4
The Search for New Treatments
It should come as no surprise to anyone that drug companies and researchers are urgently seeking compounds to fight this awful illness. It seems that grant money to study drugs that will slow the onset of the disease and alleviate some of its worst symptoms abound.5 Take note that the majority of the ongoing and developing research is not targeting a cure. At this point in time, curing the disease does not seem feasible.6
This little detail does not seem to deter Americans clamoring for hope and relief.7 The journal Science recently published research data showing that a drug used to treat a particular form of skin cancer (bexarotene [Targretin] for cutaneous T-cell lymphoma), improved brain function in mice bred with a form of Alzheimer’s disease.8-10 Improvements in social skills and the sense of smell and a decrease in brain amyloid-beta protein fragments were notable.
The drug has not been tested in humans at this time. Nonetheless, patients, relatives, and friends of patients have deluged physicians across the country begging that bexarotene be prescribed for Alzheimer’s patients. It appears that in just 1 or 2 days following publication of these results, in a scientific journal not necessarily identified as widely read by the general public, physicians and advocacy groups started getting a flurry of calls from patients seeking to use the drug off-label. “The Alzheimer’s community is very desperate for anything that shows any sign of hope or promise,” said Eric Hall, chief executive of the Alzheimer’s Foundation of America, a New York-based advocacy organization that started to field calls from consumers as soon as the paper was published.11
Sam Gandy, MD, PhD, a neurology professor at the Mount Sinai School of Medicine and director of its Alzheimer’s Disease Research Center, was not surprised by the flood of inquiries for the use of bexarotene in his Alzheimer’s patients. He stated, “they also often ask for off-label use of another treatment on the market, intravenous immunoglobulin, which costs about $50,000 a year and is given in time-consuming infusions. To patients and families who are this motivated, the idea of an off-label pill is not a major leap. The cost would be $1,200 or more a month and unlikely to be covered by insurance.”12
Commentary
Depending on one’s perspective, the lack of any human research unfortunately means most physicians will not prescribe the drug for their suffering patients. While there may be some room for moral arguments favoring administration of the drug to individuals inflicted with Alzheimer’s disease, from a legal standpoint, there is no justification for doing so right now.
This situation needs to serve as a reminder to pharmacists that off-label prescribing, while usually legal, can be fraught with many uncertainties including legal liabilities. The old saw, “Prescribers may prescribe and pharmacists may dispense an FDA-approved drug for an indication not approved by the FDA so long as there is a body of evidence, usually in the form of peer-reviewed literature that it is an acceptable practice within the bounds of reasonable standards of health care to do so as long as the unapproved use does not constitute experimentation” holds true.13,14 While pharmacists may not necessarily know exactly what a patient is using any particular drug for, there is always the “reasonable man” test for negligent behavior.15 In this context, the question would be whether a reasonable pharmacist in the same or similar circumstances knows or should know that the drug is being used off-label or experimentally.
While there are, in fact, drugs now being used for off-label purposes to treat Alzheimer’s disease, anyone prescribing bexarotene for an Alzheimer’s patient is taking a mighty big risk. The same would hold true for pharmacists if they know this drug is being prescribed for Alzheimer’s disease and not for the very specific type of skin cancer for which it is approved. As badly as Alzheimer’s advocates, patients, and loved ones are begging for a chance (an “anything with promise is better than nothing” mentality) to try this drug, it does not, at this moment in time, meet the test set forth above.16 There is no body of evidence in any peer-reviewed journals suggesting use of this drug for Alzheimer’s is reasonable with the state of knowledge we have before us today. Such use would no doubt come under the heading of unacceptable human experimentation, opening prescribers and pharmacists to potential criminal and civil charges.
The manufacturer of bexarotene, Eisai Co., based in Japan, is being a bit vague in its public statements about this potential off-label use of its product. Instead of coming right out and saying that the drug should not be used in humans until further research is performed, its spokesperson was quoted as saying, “It is our policy to not comment on off-label uses of our products. Physicians are able to prescribe products as they see medically necessary.” The company also disavowed any knowledge of the research study published in Science.17
That is a great way for a company to have its cake and eat it too. Companies can say that they do not have any control of how their products are used once they leave their factories, but they will be happy to take the profits from off-label uses if such an occurrence happens. While this may well be an accurate point of view at some time down the road when human-use evidence is collected, from an ethical perspective, the company should be saying this drug has been approved in the U.S. for a very narrow and specific use and should not be used for other purposes at this time. Perhaps that time could come in a relatively short period if clinical trials were opened under the auspices of an FDA Investigational New Drug Application (INDA) with the approval of the Institutional Research Board (IRB) where the research might be conducted.
Depending on which data is used or the type of diseases involved, the rate of off-label prescribing of prescription drugs in the U.S. ranges from a conservative 20% to as high as 75%.18,19 In the majority of cases, using drugs for purposes not approved by the FDA will not expose pharmacists to extraordinary risks of legal liability. However, when a report like the one discussed here is released and throngs of patients, caregivers, and even physicians start demanding a prescription drug under these circumstances, pharmacists should be very wary of complying. Perhaps the line between experimentation and acceptable off-label use is gray, but a reasonable pharmacist staying abreast of the current literature should know the difference.
REFERENCES
1. Thomas D. Do not go gentle into that good night. www.poets.org. Accessed February 11, 2012.
2. A word from Dr. Fillit. Alzheimer’s Drug Discovery
Foundation. www.alzdiscovery.org/index.php/alzheimers-disease/dr-fillit.
Accessed February 11, 2012.
3. Alzheimer’s Disease Medications Fact Sheet. National
Institute on Aging.
www.nia.nih.gov/alzheimers/publication/alzheimers-disease-medications-fact-sheet.
Accessed February 11, 2012.
4. See Note 3, supra.
5. Grant opportunities. Alzheimer’s Drug Discovery
Foundation.
www.alzdiscovery.org/index.php/research-programs/grant-opportunities.
Accessed February 11, 2012.
6. Alzheimer’s disease. PubMed Health. www.ncbi.nlm.nih.gov/pubmedhealth/PMH0001767/. Accessed February 11, 2012.
7. Wang SS. Alzheimer’s families clamor for drug. Wall Street Journal. February 11, 2012. http://online.wsj.com/article/SB10001424052970204642604577215382600942356.html. Accessed February 11, 2012.
8. Cramer PE, Cirrito JR, Wesson DW, et al. ApoE-directed
therapeutics rapidly clear beta-amyloid and reverse deficits in AD mouse
models. Science. February 9, 2012. www.sciencemag.org/content/early/2012/02/08/science.1217697. Accessed February 11, 2011.
9. Drug quickly reverses Alzheimer’s symptoms in mice. ScienceDaily. February 8, 2012. www.sciencedaily.com/releases/2012/02/120209144005.htm. Accessed February 12, 2012.
10. Bexarotene. PubMed Health. www.ncbi.nlm.nih.gov/pubmedhealth/PMH0000433/. Accessed February 11, 2012.
11. See Note 7, supra.
12. See Note 7, supra.
13. This is one of those pharmacy jurisprudence statements
often heard in college classrooms that is more cliché than quotable.
Yet, there is a plethora of literature to support it. See, e.g.,
Stafford RS. Regulating off-label drug use—rethinking the role of the
FDA. N Engl J Med. 2008;358:427-429. April 3, 2008. www.nejm.org/doi/full/10.1056/NEJMp0802107. Accessed February 11, 2012.
14. “Off-label” drug use: shoppers guide to prescription drugs–number 6. Consumer Reports.
2007.
www.consumerreports.org/health/resources/pdf/best-buy-drugs/money-saving-guides/english/Off-Label-FINAL.pdf.
Accessed February 12, 2012.
15. Negligence breach of duty—reasonable man test. Law Teacher. www.lawteacher.net/tort-law/lecture-notes/negligence-breach-lecture.php. Accessed February 11, 2012.
16. See Note 7, supra.
17. See Note 7, supra.
18. Rubin R. Study: off-label drugs often prescribed. USA Today. May 8, 2006. www.usatoday.com/news/health/2006-05-08-offlabel-drugs_x.htm. Accessed February 12, 2012.
19. Angarola RT, Joranson DE. Off-label uses of prescription drugs in pain management. APS Bulletin. 1995;5:14-15. www.painpolicy.wisc.edu/publicat/95apsol.htm. Accessed February 12, 2012.
To comment on this article, contact rdavidson@uspharmacist.com.





