ABSTRACT: As patents of popular brand-name ophthalmic medications expire and generic formulations of these medications are approved, there is an ongoing debate among healthcare providers regarding the safety and efficacy of generic versus brand-name ophthalmic medications. Clinical studies have demonstrated advantages and disadvantages of using generic and brand-name ophthalmic medications; more research is needed. There are concerns that packaging factors such as bottle material, shape, and size may impact the efficacy of generic ophthalmic medications. Overall, generic ophthalmic medications are safe and effective, but it is imperative that healthcare professionals routinely monitor their patients to ensure safety and to screen for any changes in efficacy or potential adverse effects of these formulations.
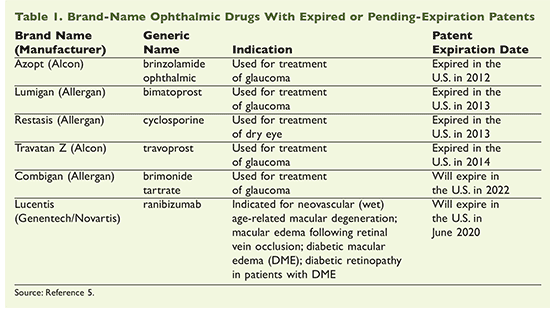
For decades there has been debate among healthcare providers about the efficacy and safety of generic drugs versus brand-name medications, but in March 2011, the patent expiration of the most widely used glaucoma drug in the United States, Pfizer’s Xalatan (latanoprost ophthalmic solution) sparked particular interest regarding generic medications that are used to treat ophthalmic conditions.1 Xalatan is the first of the prostaglandin analogues to become available as a generic formulation. Xalatan is reported to have the largest sales figures of any drug in the ophthalmic industry, and the recent approval of a generic formulation is resulting in significant increases in sales for the generics industry.2 In general, a patient could expect to save approximately $1,300 annually by switching from the brand-name version of this product to the generic formulation.3,4 Interest in the topic of generic ophthalmic drugs continues to escalate as more brand-name ophthalmic medication patents expire (Table 1).5 On February 23, 2015, Lupin Pharmaceuticals received FDA approval to market bimatoprost ophthalmic solution 0.03%, a generic version of Lumigan (Allergan; bimatoprost ophthalmic solution 0.03%).6
In 2012, 84% of prescriptions dispensed were in generic form and more than 75% of drugs listed in the FDA’s Orange Book also had generic counterparts.7-9 A recent study reported that overall, generic-drug use saved the healthcare industry and consumers over $192 billion in 2011 and more than $1 trillion over the past decade.10 In addition, a 2014 market report states that the global generic ophthalmic drug market was valued at $16 billion in 2012 and is expected to escalate to a value of $21.6 billion in 2018.11 These figures clearly illustrate the profound impact that the use of generic drugs has on the healthcare system. Some market research experts believe that the expanding ophthalmic-drug market is primarily the result of the increasing prevalence of ophthalmic disorders, particularly among the growing older adult population.11
In general, generic drugs cost 80% to 85% less than brand-name drugs, and the lower cost is very appealing to many patients.12,13 Generic ophthalmic formulations are available for numerous therapeutic categories and are prescribed daily by many healthcare providers. The availability of generic formulations of ophthalmic drugs enables many patients to afford medical treatment for their ophthalmic conditions. Overall, patients do well with these medications, but some physicians report having patients who do not tolerate generic formulations, possibly owing to differences in packaging, including bottle material and configuration of the dropper, which may affect the amount of drug dispensed or cause difficulties in administering drops for some patients.2,9,14
Assessing Bioequivalence of Generic Ophthalmic Drugs
The FDA defines a generic as a drug product that is comparable to a brand/reference-listed drug product in dosage form, strength, route of administration, quality, performance characteristics, and indications.15 According to the FDA standards, if a formulation is a true generic, there should be no concerns regarding the safety and efficacy of the generic formulation.15,16 Some ophthalmology experts and healthcare providers have raised questions regarding the safety and efficacy of the growing number of generic ophthalmic products. Many health experts believe that a lack of strict requirements for determining bioequivalence and a shortage of therapeutic-equivalence studies has added to these concerns.17 While some ophthalmology professionals believe that generics are preferable because of their lower cost compared to that of brand-name medications, others disagree, owing to a belief that generic formulations are not always the same as their brand-name counterparts. A survey of physicians in the U.S. reported that willingness to prescribe a generic drug over a brand name is reliant upon the physician’s beliefs about and experiences with generic drugs as well as the physician’s knowledge of the FDA standard for bioequivalence.10,18
Although generic drugs work well for the majority of patients, results from various clinical studies have revealed differences, such as the way the drugs are absorbed, when compared to their brand-name counterparts. While generic-drug manufacturers are typically required by the FDA to demonstrate equivalence in order to obtain FDA approval, most ophthalmic solutions are not mandated to prove therapeutic equivalence.7,17 According to the policies of the FDA, ophthalmic generic products introduced prior to 1992 did not have to match the inactive ingredients listed by the brand-name manufacturer.17 The FDA drug-approval process was updated in 1992 after the introduction of the Generic Drug Enforcement Act, which required generic substitutes to include the same active and inactive ingredients as the brand-name drug counterpart.17
A critical concern for generic ophthalmic agents when comparing them to systemic agents is that the FDA does not require pharmacokinetic assays and strict demonstration of human bioequivalence, because those would require invasive ocular procedures.10,17 Bioavailability cannot be directly measured in the human eye; thus, testing may be performed in animals and could result in inaccurate or misleading results, given that drug absorption rates may vary between animal and human eyes.10,19,20
Moreover, the safety of packaging and bottle manufacturing for eyedrop medications is strictly regulated in brand-name drugs; however, generic medications may have differences in packaging and the size of the eyedrop.21,22 Eyedrop size has an impact on the number of doses in a container; the larger the drop size, the fewer doses will be in a container of a given size, the sooner a patient will need to refill, and the more expensive a month’s supply will be.21 More important, researchers have found that when a patient is switched from a brand-name to a generic formulation or from one generic to another, drop size may vary between formulations from an estimated 25 mcL to 70 mcL. If a patient receives a different amount from the one prescribed, adverse effects may occur and efficacy may be affected.10,23,24
Clinical Studies
There is clearly a need for more clinical studies comparing brand-name drugs with generic ophthalmic medications. Some clinical studies have demonstrated variations between brand-name drugs and generic formulations, and others have demonstrated equivalence. Some of these studies are discussed below.
Research published in a 2015 issue of Ophthalmology reports that glaucoma patients are more likely to adhere to medication regimens if they are prescribed generic drugs.25,26 The study was conducted at the University of Michigan Kellogg Eye Center and College of Pharmacy. Researchers studied medication adherence rates 18 months before and after generic latanoprost became available in March 2011.25,26 The study results demonstrated that medication adherence improved on average among all patients who were switched from brand-name to generic latanoprost.25,26 Adherence rates were best among patients who had been on brand-name prostaglandin analogues (PGAs) other than branded latanoprost. In addition, among patients who had poor adherence when only brand-name versions were available, researchers found a correlation between lower medication cost and improved medication adherence.4,25,26
In 2012, Kahook et al compared the concentration of active ingredients and preservatives in commonly used brand-name and generic glaucoma drugs.27 The study concluded that brand-name formulations contained active ingredients and benzalkonium chloride (BAK) in concentrations that were generally in agreement with their package inserts at baseline.27 The two generic formulations of latanoprost contained baseline levels of active ingredients that were 10% greater than their labeled value.27 Generic latanoprost formulations had significant loss of active ingredient concentration after exposure to temperatures at 25°C and 50°C for 30 days.27 Bottles of both generic medications had higher levels of particulate matter compared with those of the brand-name versions.27
A 2012 study investigated whether brand-name glaucoma drops differed from generic counterparts with regard to bottle design, viscosity, surface tension, and volume.28 In North America, researchers found that American and Canadian Timoptic XE ophthalmic drops differed considerably from the generic formulations with regard to drop volume, viscosity, surface tension, and bottle tip.28 Canadian brand-name Timoptic delivered considerably smaller drop volumes than generic Apo-Timop.28 The researchers concluded that consideration should be given to drop viscosity and bottle design when generic ophthalmic products are evaluated for interchangeability and market approval.28
A recent study that compared generic latanoprost to Xalatan found that both treatments demonstrated equivalent safety and tolerability among patients.29 There is some debate about this study, since 99.6% of the study subjects were Caucasian and the results may not be the same for African Americans, Asians, or Hispanics. Further studies are needed.29
Results from a 2001 study showed that the brand-name formulation of timolol gel-forming solution based in gellan gum lowered intraocular pressure (IOP) better than its generic counterpart based in xanthan gum.30 As a result, some healthcare professionals believe that particle size and other properties of a suspension should be evaluated for generic equivalence.30
A 2007 study investigated the variations in particle size between Pred Forte, EconoPred Plus, and generic prednisolone acetate 1%.31 The generic form of prednisolone acetate was reported to have a greater propensity for particles to agglomerate, which subsequently led to inconsistent dosage concentration.31 Moreover, the researchers concluded that the larger particle sizes may possibly clog the tip of the medication bottle during instillation of drops and further change dosage consistency and concentration. In contrast, the particle sizes in Pred Forte were smaller and more uniform, which allowed them to stay in suspension longer and give a more accurate dosage of the drop.31
Results from a 2005 study reported that 20% of generic ciprofloxacin eyedrops purchased in India were underpotent and that some preparations of the antibiotic content were low enough to negatively affect treatment outcomes.32
Factors to Consider With Generic Ophthalmic Medications
Given that studies have demonstrated both strengths and limitations associated with generic and brand-name ophthalmic medications, healthcare professionals are encouraged to closely monitor patients, and pharmacists should thoroughly counsel them on the proper use of their prescribed medications. Areas of concern regarding the use of generic drugs include:
Excipients: While generic drugs may have the same active ingredients as brand name drugs, generic ophthalmic drugs are not required to have the same excipients—preservatives, pH adjusters, antioxidants, thickening agents, tonicity adjusters, and buffers—as brand-name drugs, and this can interfere with the pharmacokinetic and pharmacodynamic properties of active agents.2,10 For example, some clinical studies have shown that the pH of ophthalmic solutions can influence drug ionization, efficacy, and pharmacokinetics.10,33 Some experts believe that the effectiveness and tolerability of ophthalmic medications may be affected or altered by inactive ingredients.
Switching: In 2011, when the patent for Xalatan expired, several generic companies wanted to market their generic formulations. Some ophthalmologists expressed concern about generic formulations because some patients who were using brand-name medications and were switched to generics because of insurance plan requirements experienced increases in IOP by as many as 10 points.2
Studies have shown that while the majority of generic drugs are safe and effective, therapeutic differences have been observed when patients are switched from brand-name to generic products or from one generic formulation to another. In addition, results from various clinical studies suggest that generic formulations do not always provide the same treatment specifications or results as branded medications, thereby contributing to the ongoing debate.34
Packaging: The FDA does not require that generics be packaged the same way as their brand-name counterparts.34 Therefore, some healthcare providers are concerned that while the contents of a generic formulation may be identical to the contents of a brand-name formulation, packaging factors such as bottle material, shape, and size, as well as differences in cap color and drop size, may impact the efficacy of these topical medications.34 Since the generic medications may look different, deliver drops differently owing to the diameter of the bottle opening, and have unfamiliar colors and bottle shapes—for example, Xalatan comes in a small, flat, flexible bottle while some of the generic formulations of latanoprost come in round, more rigid bottles—a patient may be confused by the dissimilarities, thereby increasing the risk of administration and dosing errors and improper use of the medication. A bottle’s shape or size may make it difficult to administer drops, or drops may be wasted because they slip out too fast.7,34 The eyedrop size delivered from the bottle and the bottle fill amount may also vary in generic products, causing patients to run out of the drug prior to its refill date.9 These packaging discrepancies may cause particular challenges for the elderly, as well as those with physical limitations and vision impairments, and affect their compliance with therapy; they may also cause confusion among patients who are switched from brand-name to generic drugs and those who use multiple ophthalmic medications, thereby increasing the potential for drug errors.34
In addition, some generics that are manufactured outside the U.S. may not be equivalent to brand-name counterparts.34
Despite the initial apprehension regarding the use of generic ophthalmic drugs, many healthcare professionals are gaining confidence in the effectiveness and safety of switching patients to newly approved generic formulations, particularly if patients are monitored routinely to ensure that they are responding well to the selected therapy. The majority of healthcare professionals prescribe generics every day and would agree that, overall, generic ophthalmic medications are quite safe and effective, and the lower cost is appealing to patients, especially those on multiple drug regimens. In some cases, patients do not respond well to generic formulations, and in those cases cost savings should not be the most important factor, but rather what is best for the patient and the optimal therapeutic outcome. Healthcare professionals should always monitor and follow-up with patients when switching them from brand-name to generic drugs, to ensure they are achieving the desired therapeutic response. Pharmacists can play an important role in the patient education that is critical to ensuring that patients are using the medications correctly. If patients are on multiple medications, the high cost of those drugs may deter them from adhering to their medication regimens, and generic formulations may be cost-effective options.
As more generics become available, the FDA continues to find ways to improve generic manufacturing. In November 2013, the agency proposed a rule that would enable generic-drug manufacturers to update their packaging and labels if they receive information about potential safety concerns.34 The FDA plans to make a final ruling on this issue in September 2015.35 In addition, in early February 2015 the FDA awarded a grant to a researcher at Texas A&M College of Pharmacy to develop a testing method for determining therapeutic equivalency of topical ophthalmic medications.36 The method will demonstrate whether brand and generic formulations of ophthalmic medications are equally effective and will benefit healthcare professionals by providing more in-depth clinical information.36
The Role of the Pharmacist
As more brand-name ophthalmic medications become available in generic formulations, insurance companies are likely to switch patients from brand-name to generic drugs. As one of the most accessible healthcare professionals, pharmacists are in a pivotal position to provide patients with pertinent information regarding any concerns or questions they may have about generic medications. Some patients may notice differences in how generic ophthalmic drops feel and how well they work. During counseling, patients should be encouraged to discuss any concerns with their primary healthcare provider.
Conclusion
When the patents of commonly used brand-name medications expire and generic formulations become available, it is important that healthcare providers effectively monitor patients and educate them on the proper use of their medications to ensure safety and efficacy. Generic drugs are used every day and are here to stay. Generally, they are safe and effective for the majority of patients and provide a cost-effective means for patients to receive the medications they need in order to maintain their overall health. Until the results of more clinical studies comparing generic drugs to branded drugs are available, healthcare professionals should routinely monitor their patients, especially if they are switched from brand-name to generic drugs, to ensure patient safety and to screen for any changes in drug efficacy or potential adverse effects. Most people do well with generic medications. The approval of generic formulations enables healthcare providers and their patients to have an open dialogue about what is best for each patient and to make informed decisions about that individual patient's health.
REFERENCES
1. Murphy P. Generic latanoprost: one year after approval. Optometric Management. March 1, 2012. www.optometricmanagement.com/articleviewer.aspx?articleID=106839. Accessed May 12, 2015.
2. Karmel M. Generic vs. branded: just how equal is equal? American Academy of Ophthalmology. June, 2011. http://development.aao.org/publications/eyenet/201106/upload/CUcomp-June-2011.pdf. Accessed May 6, 2015.
3. McCarthy C. Generics improve compliance in glaucoma patients. Optometry Times. March 13, 2015. http://optometrytimes.modernmedicine.com/optometrytimes/news/generics-improve-compliance-glaucoma-patients?page=full. Accessed May 12, 2015.
4. Shin H. Generic drugs may help glaucoma patients. American Academy of Ophthalmology, eyeSmart [website]. February 24, 2015. www.geteyesmart.org/eyesmart/eye-health-news/generic-drugs-help-glaucoma-patients.cfm. Accessed May 12, 2015.
5. PRNewswire. Ophthalmic drugs; data, analysis and forecasts to 2024. www.prnewswire.com/news-releases/ophthalmic-drugs-data-analysis-and-forecasts-to-2024-300004949.html. Accessed May 12, 2015.
6. Lupin receives FDA approval for generic Lumigan Ophthalmic Solution 0.03%. Lupin Pharmaceuticals. February 23, 2015. www.lupinworld.com/pdf/2015/Lupin-Receives-FDA-Approval-for-Generic-Lumigan-Ophtahlmic-Solution-0-03.pdf. Accessed May 12, 2015.
7. Pickles V. Branded vs. generic ophthalmic drugs: balancing cost and care. Ophthalmology Management. 2013;17(7):7-14.
8. IMS Institute for Healthcare Informatics. Declining medicine use and costs: for better or worse. May 2013. http://static.correofarmaceutico.com/docs/2013/05/20/usareport.pdf. Accessed May 12, 2015.
9. Cantor L. Generic ophthalmic medications: as good as a Xerox? Medscape Ophthalmology, 2008. www.medscape.org/viewarticle/583866. Accessed May 21, 2015.
10. Zore M, Harris A, Tobe LA, et al. Generic medications in ophthalmology. Br J Ophthalmol. 2013;97(3):253-257.
11. Ophthalmic drug market expected to reach $21.6 billion globally in 2018. Optometry Times. February 4, 2014. http://optometrytimes.modernmedicine.com/optometrytimes/content/tags/eye-and-vision-disorders/ophthalmic-drug-market-expected-reach-216-billi?page=full. Accessed May 12, 2015.
12. Benkoff A. Ophthalmic generic formulations. Optometrist Continuing Education.com. http://optometristcontinuingeducation.com/wp-content/uploads/2012/01/BENKOFF-OPTHALMIC-GENERIC-FORMULATIONS-CORRECTED.pdf. Accessed May 12, 2015.
13. FDA. Facts about generic drugs. September 19, 2012. www.fda.gov/ drugs/resourcesforyou/consumers/buyingusingmedicinesafely/understandinggenericdrugs/ucm167991.htm. Accessed May 12, 2015.
14. Krader C. Branded or generic drugs: what’s in a name? Ophthalmology Times. January 15, 2014. http://ophthalmologytimes.modernmedicine.com/ophthalmologytimes/content/tags/drug-therapy-ophthalmology/branded-or-generic-drugs-whats-name?page=full. Accessed May 12, 2015.
15. FDA. Generic drugs. www.fda.gov/downloads/Drugs/DevelopmentApprovalProcess/SmallBusinessAssistance/ucm127615.pdf. Accessed May 12, 2015.
16. Realini T. Facts and myths about generic formulations of ophthalmic products. ASCRS EyeWorld. March 2014. www.eyeworld.org/article-facts-and-myths-about-generic-formulations-of-ophthalmic-products. Accessed May 12, 2015.
17. Aref AA. Generic drugs for the treatment of ocular conditions: changing the treatment landscape. Expert Rev Clin Pharmacol. 2014;7(5):551-553.
18. Banahan BF, Kolassa EM. A physician survey on generic drugs and substitution of critical dose medications. Arch Intern Med. 1997;157:2080-2088.
19. Ekong A. Generic vs. branded eye drops—just how equal is equal? Illinois Retina Institute. www.illinoisretinainstitute.com/web_documents/generic_v_branded_eye_drops_single_page.pdf. Accessed May 12, 2015.
20. Cantor LB. Ophthalmic generic drug approval process: implications for efficacy and safety. J Glaucoma. 1997;6:344-349.
21. Generic medications: are they the same? Glaucoma Research Foundation. May 1, 2014. www.glaucoma.org/treatment/generic-medications-are-they-the-same.php. Accessed May 12, 2015.
22. Chu E, Hamp A. Generic vs. brand drugs: which is better? Review of Optometry. February 15, 2012. www.reviewofoptometry.com/content/d/therapeutics/c/32673/. Accessed May 12, 2015.
23. Van Santvliet L, Ludwig A. Determinants of eye drop size. Surv Ophthalmol. 2004;49:197-213.
24. German EJ, Hurst MA, Wood D. Reliability of drop size from multi-dose eye drop bottles: is it cause for concern? Eye. 1999;13:93-100.
25. Generic eye drops for glaucoma encourage greater use. U.S. News & World Report Health. February 19, 2015. http://health.usnews.com/health-news/articles/2015/02/19/generic-eye-drops-for-glaucoma-encourage-greater-use. Accessed May 12, 2015.
26. Stein JD, Shekhawat N, Talwar N, Balkrishnan R. Impact of the introduction of generic latanoprost on glaucoma medication adherence. Ophthalmology. 2015;122(4):738-747.
27. Kahook MY, Fechtner RD, Katz LJ, et al. A comparison of active ingredients and preservatives between brand name and generic topical glaucoma medications using liquid chromatography-tandem mass spectrometry. Curr Eye Res. 2012; 37(2):101-108.
28. Mammo ZN, Flanagan JG, James DF, Trope GE. Generic versus brand-name North American topical glaucoma drops. Can J Ophthalmol. 2012;47(1):55-61.
29. Allaire C, Dietrich A, Allmeier H, et al. Latanoprost 0.005% test formulation is as effective as Xalatan in patients with ocular hypertension and primary open-angle glaucoma. Eur J Ophthalmol. 2012;22(1):19-27.
30. Fiscella RG, Gaynes BI, Jensen M. Equivalence of generic and brand-name ophthalmic products. Am J Health Syst Pharm. 2001;58(7):616-617.
31. Roberts CW, Nelson PL. Comparative analysis of prednisolone acetate suspensions. J Ocul Pharmacol Ther. 2007;23(2):182-187.
32. Weir RE, Zaidi FH, Charteris DG, et al. Variability in the content of Indian generic ciprofloxacin eye drops. Br J Ophthalmol. 2005; 89(9):1094-1096.
33. Brochure WF, Maren TH. pH and drug ionization affects ocular pressure lowering of topical carbonic anhydrase inhibitors. Invest Ophthalmol Vis Sci. 1993;34:2581-2587.
34. Kahook M. Branded vs. generic: proceed with caution. Review of Ophthalmology. April 2, 2014. www.reviewofophthalmology.com/content/d/glaucoma/c/47483/. Accessed May 12, 2015.
35. Johnsen M. FDA exploring a change in generic drug product label updates. Drug Store News. February 24, 2015. www.drugstorenews.com/article/fda-exploring-change-generic-drug-product-labels-updates. Accessed May 12, 2015.
36. Shipman C. Texas A&M researcher receives FDA grant to develop test to bring generic eye medications to market. Vital Record. Texas A&M Health Science Center. February 5, 2015. http://news.tamhsc.edu/?post=texas-am-researcher-receives-fda-grant-for-test-to-bring-generic-eye-medications-to-market. Accessed May 6, 2015.
To comment on this article, contact rdavidson@uspharmacist.com.






