US Pharm. 2018;43(6)(Generic Drugs suppl):10-16.
ABSTRACT: The availability of generic drugs provides patients with safe and cost-effective alternatives to many of the brand-name drugs on the market. In the United States, generic medications account for an estimated 89% to 90% of the prescriptions dispensed annually. The FDA Office of Generic Drugs ensures that patients have access to safe, effective, and affordable generic medications by following a rigorous review and approval process. Through this process, the FDA requires a generic-drug company to produce enough data to demonstrate that it can manufacture a medication that can be appropriately substituted for its brand-name counterpart. It is imperative that pharmacists be familiar with the generic-drug approval process in order to answer patient questions or concerns about generic medications.
The accessibility, convenience, and use of generic drugs, which are alternatives to brand-name medications, have had a significant impact on cost savings for healthcare consumers and the healthcare industry. According to the FDA, a generic drug is a medication manufactured to be the equivalent of an already marketed brand-name medication in dosage form, safety, strength, route of administration, quality, performance characteristics, and therapeutic indication.1,2 Moreover, the FDA also mandates that generic drugs must have the same high quality as their brand-name counterparts.
In order to obtain approval from the FDA, generic medications undergo a thorough review process to ensure that they are as safe and effective as their corresponding brand-name products. All approved products—both brand and generic—are listed in the FDA’s publication Approved Drug Products with Therapeutic Equivalence Evaluations, commonly known as the Orange Book.3
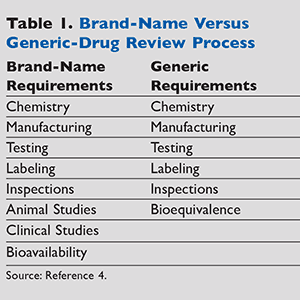
The Drug Price Competition and Patent Term Restoration Act of 1984, also known as the Hatch-Waxman Act, established bioequivalence as the basis for approving generic copies of drug products.3 These amendments permit the FDA to approve applications to market generic versions of brand-name drugs without repeating costly and duplicative clinical trials to establish safety and efficacy.3 Hatch-Waxman established the Abbreviated New Drug Application (ANDA) for generic medications.3 Brand-name drugs must prove their safety and effectiveness through expensive and time-consuming research and development programs, including clinical studies.1,2 In contrast, generic-drug developers can use data from their brand-name counterparts, resulting in much less expensive development programs and in affordable access to treatments for many patients.2
Generic-drug applications are termed abbreviated because they are generally not required to include preclinical (animal) and clinical (human) data to establish safety and effectiveness. Instead, generic applicants must scientifically demonstrate that their drug performs in the same manner as the brand-name drug.1,2 In addition, multiple applications for generic drugs are often approved to market a single product; this creates competition in the marketplace, typically resulting in lower prices.1 When there is more than one manufacturer marketing a generic drug, the resulting competition typically produces drug prices that are about 85% less than those of their brand-name counterparts.1,2
The Bioequivelance Benchmark
In the United States, generic medications must be proven to be bioequivalent to gain FDA approval. The FDA also ensures that generics are as safe and effective as their brand-name counterparts.2,5,6 Generic medications account for an estimated 89% to 90% of the prescriptions dispensed in the U.S.5,7,8 Over the last decade, FDA-approved generic drugs have saved consumers more than $1.67 trillion.2,7 The 2017 Generic Drug Access and Savings in the U.S. report shows that generic pharmaceuticals saved the U.S. healthcare system $253 billion in 2016.7 In addition to healthcare system savings, according to the Kaiser Family Foundation, the availability of generics is a critical value to U.S. patients, 21% of whom skipped filling a prescription for themselves or a family member in the last year owing to cost. Additionally, research from the Association for Affordable Medicines determined that the abandonment rate for branded drugs is 266% higher than it is for generics.7,9 An estimated 90% of generics have a copay of less than $20, while only 39% of branded drugs have copays less than $20.7,9
The FDA requires a generic-drug company to produce enough data to demonstrate that it can make a drug that can be appropriately substituted for its brand-name counterpart. The challenge to the generic-drug maker is to demonstrate to the FDA that the generic version is compatible with a brand-name drug that has been shown to be safe and effective.5,6 The FDA also provides the scientific and regulatory advice needed to bring safe, effective, high-quality generic alternatives to market, which in turn creates more affordable treatment options for patients.8
There are several types of data that generic companies must provide to the FDA for review and evaluation. It is critical that the data demonstrate the manufacturing process and enable the FDA to ascertain if the manufacturer can consistently make a high-quality product.8 More information about the FDA’s generic approval process can be found in TABLE 1 and TABLE 2.

Generic Drugs in the News
In September 2017, the FDA announced a new consumer and healthcare-provider education campaign that is intended to raise awareness about the value of generic drugs. The FDA wants consumers to know that generic drugs have the same safety, efficacy, and quality standards as their brand-name counterparts.11
The FDA is also encouraging patients and their prescribers to talk with each other about exploring generic alternatives to brand-name medications. The agency hopes that pharmacists can relay these messages to their patients.11
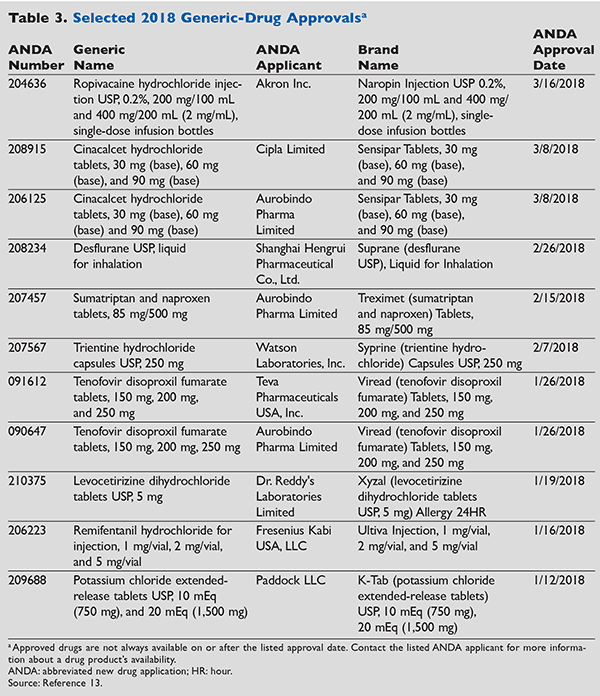
Additionally, in 2017, the FDA’s Office of Generic Drugs had a record-setting year for generic approvals. In 2017, 1,027 new generic drugs were approved, 214 more than 2016 when 813 generic medications were approved.12 Annually, the FDA’s Center for Drug Evaluation and Research approves an extensive range of new drug products.13 (TABLE 3 lists selected new generic-drug approvals, including the corresponding brand names, available as of press time for 2018.)
REFERENCES
1. Generic drugs; questions & answers. FDA website. Updated January 1, 2018. www.fda.gov/Drugs/ResourcesForYou/Consumers/QuestionsAnswers/ucm100100.htm#q1. Accessed March 8, 2018.
2. Generic drug facts. FDA website. Updated October 6, 2017. www.fda.gov/Drugs/ResourcesForYou/Consumers/BuyingUsingMedicineSafely/GenericDrugs/ucm167991.htm. Accessed March 8, 2018.
3. Abbreviated new drug application. FDA website. Updated November 28, 2017. www.fda.gov/Drugs/DevelopmentApprovalProcess/HowDrugsareDevelopedandApproved/ApprovalApplications/AbbreviatedNew
DrugApplicationANDAGenerics/default.htm. Accessed March 8, 2018.4. Generic drugs. FDA website. www.fda.gov/downloads/AboutFDA/Transparency/Basics/UCM226568.pdf. Accessed March 8, 2018.5. Generic drugs. FDA website. Updated March 4, 2018. www.fda.gov/Drugs/ResourcesForYou/Consumers/BuyingUsingMedicineSafely/GenericDrugs/default.htm. Accessed March 8, 2018.6. What is the approval process for generic drugs? FDA website. Updated August 31,2017. www.fda.gov/Drugs/ResourcesForYou/Consumers/BuyingUsingMedicineSafely/GenericDrugs/ucm506040.htm. Accessed March 8, 2018.
7. Frederick J. Savings take center stage for generic makers. Drug Store News. Published online February 4, 2018. www.drugstorenews.com/pharmacy/generic-makers-focus-savings. Accessed March 6, 2018.
8. The generic approval process. FDA website. www.fda.gov/Drugs/NewsEvents/ucm508150.htm. Accessed March 11, 2018.
9. 2017 generic drug access and savings in the U.S. Association for Accessible Medicines website. https://accessiblemeds.org/sites/default/files/2017-07/2017-AAM-Access-Savings-Report-2017-web2.pdf. Accessed March 10, 2018.
10. FDA fact sheet: what’s involved in reviewing and approving generic drug applications. FDA website. www.fda.gov/downloads/Drugs/ResourcesForYou/Consumers/BuyingUsingMedicineSafely/GenericDrugs/UCM510852.pdf. Accessed March 11, 2018.
11. FDA promotes talking about generics in new public education campaign. Pharmacy Today. 2017;23(11):19. www.pharmacytoday.org/article/S1042-0991(17)31675-4/fulltext. Accessed March 11, 2018.
12. Uhl K. 2017 was another record setting year for generic drugs. FDA website. Published February 7, 2018. https://blogs.fda.gov/fdavoice/index.php/2018/02/2017-was-another-record-setting-year-for-generic-drugs. Accessed March 10, 2018.
13. First generic drug approvals. FDA website. www.fda.gov/Drugs/DevelopmentApprovalProcess/HowDrugsareDevelopedandApproved/DrugandBiologicApprovalReports/ANDAGenericDrugApprovals. Accessed March 10, 2018.
To comment on this article, contact rdavidson@uspharmacist.com.






