US Pharm. 2015;40(5)(Specialty&Oncology suppl):8-11.
ABSTRACT: Bone is a common site for distant metastases of advanced cancers. Metastatic bone disease occurs in most advanced cancers, but it is especially common in cancers of the breast, prostate, and lung. Bone metastases increase the risk of skeletal-related events (SREs) and debilitating pain. Pharmacologic approaches to treat bone pain include the use of analgesics and anti-inflammatory drugs along with bone-modifying agents. Randomized trials support the use of bone-modifying agents as palliative care for patients with metastatic bone disease. They have been also shown to reduce or delay SREs.
Bone metastases occur in most advanced cancers, especially cancers of the breast, prostate, and lung.1 Up to 70% of patients with advanced metastatic cancer of the breast and prostate experience debilitating cancer-related bone pain.2 Bone metastasis increases the risk of skeletal-related events (SREs), defined as pathologic fractures, spinal cord compression, bone pain requiring palliative radiotherapy, orthopedic surgery for pathologic fractures, or hypercalcemia of malignancy.
Pain arising from the bone is somatic and may be chronic or incidental in nature.3 Chronic pain may arise from stretching of the periosteum due to tumorous expansion of the bone, epidural spinal cord compression, or pathologic fractures. A significant contributor to bone-related pain is osteoclastic bone resorption. Briefly, tumor cells activate osteoclasts that degrade the bone matrix, thereby liberating inflammatory cytokines and growth factors harbored within the bone and causing continual stimulation of peripheral nociceptors as a consequence. Increased turnover of bone occurs, the proportion of immature, undermineralized bone increases, and thus the likelihood of pathologic axial (vertebral, rib, and pelvic) and appendicular (femoral and humeral) fractures increases. Pathologic fractures and microfractures may cause pain on movement, defined as incidental pain. Hypercalcemia, a comorbidity of bone metastasis, may also cause bone pain.
Management of SREs in patients with bone metastases can be a major cause of hospitalization and increased healthcare costs.4,5 Treatment is aimed at reducing pain, delaying or preventing SREs, and improving independence, mobility, and quality of life (QOL). A multidisciplinary approach in the management of bone pain includes pharmacologic and nonpharmacologic interventions, depending on the degree of bone damage.
PHARMACOLOGIC TREATMENT
Pharmacists have an important role in making decisions on the selection of drug management of cancer pain, including appropriate polypharmacy and patient medication counseling. Pharmacologic approaches to pain treatment include analgesics (acetaminophen, nonsteroidal anti-inflammatory drugs [NSAIDs], opiates), bone-modifying agents (bisphosphonates and denosumab), and corticosteroids.6
Analgesics
Acetaminophen, NSAIDs, and opiates are recommended in the basic approach to cancer pain associated with bone metastases.6 They should provide patients with adequate analgesia when used appropriately. The severity of a patient’s pain, rated on a scale of 1 to 10, is used to select pain medications. FIGURE 1 shows the stepwise approach to cancer pain management. Nonopioids such as acetaminophen or an NSAID such as ibuprofen, which constitute the basis of cancer pain treatment, are used frequently for mild-to-moderate pain (scale 1-3). They are also used as analgesic adjuvants to opioids.6,7
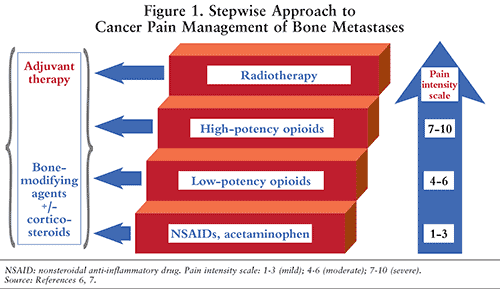
Low-potency opiate analgesics such as hydrocodone, oxycodone, codeine, and tramadol are used frequently for moderate-to-severe bone pain (scale 4-6). Severe pain (scale 7-10) requires moderate-to-high doses of high-potency opiates such as morphine, hydromorphone, hydrocodone, fentanyl, and methadone. A combination of controlled-release and immediate-release opiates can effectively control pain. They are available in different formulations and may be given by various routes of administration.6,7
NSAIDs inhibit prostaglandin synthesis via cyclooxygenases and reduce local edema and prostaglandin-induced sensitization. They are particularly useful for bone pain since many of the symptoms are related to local inflammation. The use of these agents may lead to renal insufficiency, hypertension, and peripheral edema in patients. Careful monitoring of renal function and blood pressure is advised in high-risk patients.6,7
Acetaminophen may prolong the duration of effect with opioids. Combination formulations with acetaminophen may provide better pain relief than opiates alone. Patients should be counseled about the risk of hepatic injury of using additional OTC acetaminophen for added pain control. Acetaminophen use should be closely monitored.6,7
Patients should be counseled about opiate-related side effects such as drowsiness, dizziness, constipation, nausea, and vomiting, and they should report if pain persists. Escalation of pain medication doses or adjuvant therapy may be indicated.6,7
Opiate-induced gastrointestinal side effects (e.g., abdominal pain, nausea, vomiting) are factors limiting their use when patients already have nausea and vomiting due to antineoplastic therapy, irradiation, or peritoneal metastases. Opiate-induced constipation can be avoided by dietary measures and the use of a stool softener and laxative.6,7
Factors limiting the use of NSAIDs or corticosteroids include patients with a low platelet count and the risk of gastrointestinal hemorrhage. Concurrent use of NSAIDS and corticosteroids should be avoided, or gastroprotective drugs should be administered concurrently.6,7
Bone-Modifying Agents
Treatment of bone pain should be initiated at the onset of pain, in conjunction with a bone-modifying agent such as a bisphosphonate or denosumab. When initiating therapy with bone-modifying agents, hypocalcemia should be corrected first with calcium and vitamin D supplementation.2
Bisphosphonates: Bisphosphonates are adjunctive therapy for cancer-related bone pain. In clinical trials, they have been shown to reduce pain scores, decrease analgesic use, reduce and/or delay SREs, and improve QOL. However, they have no impact on overall survival.8
Therapy with an IV bisphosphonate is recommended for patients with evidence of bone destruction due to bone metastases associated with solid tumors. Bisphosphonates have a high affinity for calcium and are absorbed primarily by the bones. Once integrated into the bone, these agents are potent inhibitors of osteoclast-mediated bone resorption and cancer-induced bone destruction. Bisphosphonates linger in the bone for an extended time and remain pharmacologically active.8
Two bisphosphonates currently approved by the FDA for the prevention of SREs in patients with breast cancer or other solid tumors are pamidronate (90 mg infused over no less than 2 hours every 3 to 4 weeks) and zoledronic acid (4 mg infused over no less than 15 minutes every 3 to 4 weeks). Insufficient evidence is available to demonstrate greater efficacy of one agent over another. Zoledronic acid has largely supplanted pamidronate in the outpatient clinical setting because infusion time is greatly reduced with zoledronic acid.8
Denosumab: Among other bone-modifying agents, denosumab is a human monoclonal antibody that targets the receptor activator of nuclear factor kappa- ligand (RANKL). RANKL inhibition with denosumab represents a new therapeutic approach to also reduce pain and reduce or delay SREs. Denosumab binds to RANKL, thereby inhibiting the formation, function, and survival of osteoclasts, which results in decreased bone resorption and increased bone mass and strength. The drug circulates in the body and is pharmacologically active for up to 6 months.8,9 Denosumab (120 mg SC every 4 weeks) may be considered in metastatic breast cancer patients who are candidates for bisphosphonates.10 Denosumab is recommended in patients with moderate-to-severe kidney failure who are not candidates for bisphosphonate therapy or who have failed bisphosphonate therapy.
In the United States, denosumab is marketed as both Xgeva and Prolia by Amgen (Thousand Oaks, CA).11,12 Only Xgeva is currently approved by the FDA for the prevention of SREs in patients with bone metastases from solid tumors but not multiple myeloma.10,11 Prolia is indicated for the treatment of postmenopausal women with osteoporosis at high risk for fracture or for treatment to increase bone mass in men with osteoporosis with high risk for fracture.12
Denosumab or zoledronic acid can be recommended in patients with bone metastases from castration-resistant prostate cancer who are at high risk for clinically relevant SREs.13 A randomized, controlled trial found that denosumab delayed SREs for longer than zoledronic acid. However, neither agent has been shown to prolong survival.14
Adverse Events: Nephrotoxicity is a serious adverse event associated with bisphosphonate therapy. It is reported to occur in approximately 7% of patients receiving either pamidronate or zoledronic acid.15 The risk of renal toxicity necessitates monitoring serum creatinine and maintaining adequate hydration before each infusion and reducing the dose of the bisphosphonate in renal insufficiency. Doses should be reduced in patients with a creatinine clearance (CrCl) ≤60 mL/min. Bisphosphonates are contraindicated in patients with a CrCl ≤30 mL/min.16
Denosumab is not excreted through the kidneys. The manufacturer does not recommend dosage adjustments in renal failure. Denosumab may be an option for patients with renal insufficiency.17
Osteonecrosis of the jaw (ONJ) is a serious but rare side effect of the bone-modifying agents. With bisphosphonates, the increased incidence of ONJ is associated with cumulative doses of drug.18 Before starting bone-modifying agents, all patients must receive a dental examination and complete appropriate preventive care, including dental surgery. Patients must achieve optimal periodontal health, maintain good oral hygiene, and be monitored for jaw or tooth pain. Data pooled from three major identical clinical trials comparing zoledronic acid to denosumab in the prevention of SREs demonstrated that ONJ occurred in 1.3% of patients receiving zoledronic acid compared with 1.8% of those treated with denosumab (P = .13).19
Other toxicities with IV bisphosphonate therapy may include acute-phase reactions. Patients should be advised about flulike symptoms, myalgias, arthralgias, and fever, which usually occur within 3 days and usually resolve within 3 days after onset. Acute-phase reactions are less frequent with denosumab.16
Cost of Therapy: From an economic perspective denosumab is more costly than bisphosphonates.16
Duration of Therapy: Although duration of therapy is not specifically established, therapy with bone-modifying agents should continue until evidence of substantial decline in a patient’s general performance status. Long-term safety data are unavailable since additional benefit beyond 2 years of treatment has not been tested.16
The OPTIMIZE-2 trial, a phase III randomized, blinded study, determined the efficacy and safety of continued zoledronic acid every 4 weeks versus every 12 weeks over 1 year in women with bone metastases from breast cancer in 412 patients who had already received at least 9 doses of zoledronic acid.20 Less frequent treatment was not inferior to monthly treatment in reducing SREs, and the investigators concluded that women with breast cancer and bone metastasis who have received at least 9 doses of zoledronic acid over the previous year can safely scale back dosing from every 4 weeks to every 12 weeks. Furthermore, there were no differences in pain levels and use of pain medications between the two treatment schedules. Lower cumulative doses also may decrease the risk of adverse events associated with zoledronic acid.20
In contrast, less frequent dosing of denosumab (Xgeva) in reducing SREs has not been studied. Denosumab works by a different mechanism and is pharmacologically active for a shorter duration than bisphosphonates.16
Corticosteroids
Anti-inflammatory adjuvants such as corticosteroids may control pain by reducing inflammation around the tumor mass, curtailing prostaglandin and leukotriene production via inhibition of cyclooxygenase and phospholipase A2, but they do not affect the tumor mass itself. They may reduce peritumoral edema and help alleviate tumor compression or infiltration. Additionally, corticosteroids may stabilize neuronal membranes, decreasing the incidence of spontaneous electrical discharges that may cause nerve pain.6
Dexamethasone is the corticosteroid of choice for advanced uncontrolled bone pain. It has a long half-life (>36 hours) and causes less fluid retention than other steroids because it has minimal mineralocorticoid effect.21
The recommended daily dose of dexamethasone for spinal cord compression is 16 to 32 mg daily in two divided doses. In bone pain the dose is 4 to 8 mg daily. Doses are tapered over 1 to 3 months. Unrelated to pain management, corticosteroids may lead to improvement in appetite, mood, energy levels, and control of nausea and emesis.21
Concurrent treatment with corticosteroid therapy may be recommended in patients receiving palliative radiation for bone pain. The dose should be tapered slowly as the radiotherapy nears completion.6 Rapid or premature tapering of corticosteroids may be associated with worsening of the underlying pain. Immediate adverse effects associated with corticosteroids include steroid psychosis, immunosuppression, and hyperglycemia. Long-term side effects include gastric ulceration, myopathy, Cushing syndrome, and osteoporosis.21
TABLE 1 summarizes drug therapy for metastatic bone pain.6-8,21

NONPHARMACOLOGIC TREATMENT
External Beam Radiation
Patients often benefit from opiates and external beam radiation when bone pain exists without structural insufficiency or fractures. Radiotherapy is thought to act by reducing local inflammation and by tumor shrinkage.22
Surgery
Surgery is sometimes indicated for spinal tumors, including histologic diagnosis, tumor local control or oncologic cure, pain relief, spinal cord decompression, and restoration of neurologic function, restoration of spine stability, and rectification of deformity.23,24
Direct decompressive surgery combined with postoperative radiotherapy may provide better outcomes than treatment with radiotherapy alone for patients with spinal cord compression due to metastatic cancer.25
Radionuclides
Bone-seeking injectable radionuclides may provide effective palliation to widespread bone metastases in advanced breast and prostate cancer. Myelosuppression would be expected to be greater in extensive bone metastases and multiple cycles of chemotherapy and radiotherapy. The long-term loss of bone marrow reserve is a limitation of radionuclide therapy in patients who are candidates for extended chemotherapy.26
SUMMARY
Metastatic bone cancer leads to painful SREs. Management of cancer-related bone pain in patients with advanced solid tumor cancers includes palliative care with analgesics and bone-modifying agents such as bisphosphonates or denosumab. Bone-modifying agents not only help to control pain but have been shown to reduce or delay SREs. Adjuvant therapy with corticosteroids, radiotherapy, and bone surgery is also used as palliative care.
REFERENCES
1. Li S, Peng Y, Weinhandl ED, et al. Estimated number of prevalent cases of metastatic bone disease in the US adult population. Clin Epidemiol. 2012;4:87-93.2. Loftus LS, Edwards-Bennett S, Sokol GH. Systemic therapy for bone metastases. Cancer Control. 2012;19(2):145-153.
3. Paice JA, Ferrell B. The management of cancer pain. CA Cancer J Clin. 2011;61:157-182.
4. Hagiwara M, Delea TE, Saville MW, Chung K. Healthcare utilization and costs associated with skeletal-related events in prostate cancer patients with bone metastases. Prostate Cancer Prostatic Dis. 2013;16(1):23-27.
5. Hagiwara M, Delea TE, Chung K. Healthcare costs associated with skeletal-related events in breast cancer patients with bone metastases. J Med Econ. 2014;17(3):223-230.
6. Slatkin N. Cancer-related pain and its pharmacologic management in the patient with bone metastasis. J Support Oncol. 2006;4(2 suppl 1):15-21.
7. National Comprehensive Cancer Network (NCCN). Adult Cancer Pain. V.1.2015. www.nccn.org. Accessed April 12, 2015.
8. Van Poznak CH, Temin S, Yee GC, et al. ASCO clinical practice guideline update on the role of bone-modifying agents in metastatic breast cancer. J Clin Oncol. 2011;29(9):1221-1227.
9. Hsu H, Lacey DL, Dunstan CR, et al. Tumor necrosis factor receptor family member RANK mediates osteoclast differentiation and activation induced by osteoprotegerin ligand. Proc Natl Acad Sci U S A. 1999;96:3540-3545.
10. National Comprehensive Cancer Network (NCCN). Clinical practical guidelines in oncology: breast cancer V.2.2015. www.nccn.org. Accessed April 12, 2015
11. Prolia (denosumab) package insert. Thousand Oaks, CA: Amgen Inc; September 2012.
12. Xgeva (denosumab) package insert. Thousand Oaks, CA: Amgen Inc; June 2013.
13. National Comprehensive Cancer Network (NCCN). Clinical practical guidelines in oncology: prostate cancer V.1.2015. www.nccn.org. Accessed April 12, 2015
14. Fizazi K, Carducci M, Smith M, et al. Denosumab versus zoledronic acid for treatment of bone metastases in men with castration-resistant prostate cancer: a randomised, double-blind study. Lancet. 2011;377:813-822.
15. Rosen LS, Gordon D, Kaminski M, et al. Zoledronic acid versus pamidronate in the treatment of skeletal metastases in patients with breast cancer or osteolytic lesions of multiple myeloma: a phase III, double-blind, comparative trial. Cancer J. 2001;7(5):377-387.
16. Gralow J, Biermann JS, Farcooki A, et al. NCCN Task Force Report: bone health in cancer care. JNCCN. 2013;11(suppl 3):S1-S51.
17. Aapro M, Saad F, Costo L. Optimizing clinical benefits of bisphosphonates in cancer patients with bone metastases. Oncologist. 2010;15:1147-1158.
18. Wilkinson GS, Kuo Y-F, Freeman JL, Goodwin JS. Intravenous bisphosphonate therapy and inflammatory conditions or surgery of the jaw: a population based analysis. J Natl Cancer Inst. 2007;99:1016-1024.
19. Saad F, Brown JE, Van Poznak C, et al. Incidence, risk factors, and outcomes of osteonecrosis of the jaw: integrated analysis from three blinded active-controlled phase III trials in cancer patients with bone metastases. Ann Oncol. 2012;23(5):1341-1347.
20. Hortobagyi GN, Lipton A, Chew HK, et al. Efficacy and safety of continued zoledronic acid every 4 weeks versus every 12 weeks in women with bone metastases from breast cancer: results of the OPTIMIZE-2 trial. J Clin Oncol. 2014;32:5(suppl): LBA9500.
21. Leppert W, Buss T. The role of corticosteroids in the treatment of pain in cancer patients. Curr Pain Headache Rep. 2012;16(4):307-313.
22. Johnstone C, Lutz ST. External beam radio-therapy and bone metastases. Ann Palliat Med. 2014;3(2):114-122.
23. Ribas ES, Schiff D. Spinal cord compression. Curr Treat Options Neurol. 2012;13(4):391-401.
24. Patchell RA, Tibbs PA, Regine WF, et al. Direct decompressive surgical resection in the treatment of spinal cord compression caused by metastatic cancer: a randomised trial. Lancet. 2005;366:643-648.
25. Furlan JC, Chan KK, Sandoval GA, et al. The combined use of surgery and radiotherapy to treat patients with epidural cord compression due to metastatic disease: a cost-utility analysis. Neuro Oncol. 2012;14(5):631-640.
26. Tomblyn M. The role of bone-seeking radio-nuclides in the palliative treatment of patients with painful osteoblastic skeletal metastases. Cancer Control. 2012;19(2):137-144.
To comment on this article, contact rdavidson@uspharmacist.com.





