US Pharm.
2008;33(2)38-42.
Last
year saw the introduction of a direct renin antagonist (RA), the first new
class of antihypertensive medications to enter the market in more than a
decade. Not since the release of angiotensin receptor blockers (ARBs) and
T-subtype calcium channel blockers (CCBs) have practitioners had a new choice
in controlling one of the most common and potentially devastating diseases in
the United States. ARBs have slowly progressed to become a common, if
expensive, therapeutic alternative, while T-subtype CCBs had a market run cut
short by unforeseen fatal toxicities. This review will focus on the first
agent of the RA class, aliskiren (Tekturna), reviewing its pharmacology,
clinical trials, and pertinent patient-counseling information while trying to
put into context its current and potential place in therapy.
Pharmacology
Renin, secreted by the kidneys in
response to poor renal perfusion and low serum sodium levels, starts the chain
of events that characterize the renin-angiotensin system (RAS), ultimately
leading to an increase in active angiotensin II. Angiotensin II acts by
increasing vasoconstriction and sodium reabsorption, increasing blood pressure
(BP). Aliskiren acts as a direct renin antagonist, blocking the conversion of
angiotensinogen to angiotensin I. While this may seem similar to
angiotensin-converting enzyme (ACE) inhibitors and ARBs, a mechanism unique to
RAs is the ability to blunt the plasma renin activity (PRA) that is increased
when these other agents are used. The clinical relevance of this blunted PRA
is unclear at this time.1 An overview of the RAS and agents that
work upon it are presented in FIGURE 1.
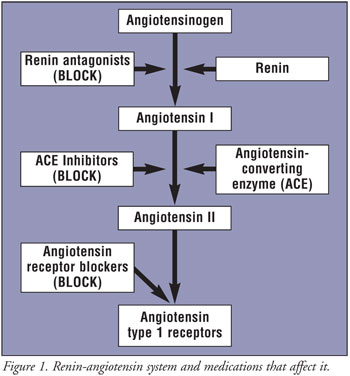
Aliskiren exhibits minimal
oral absorption (~2.5%) and has a plasma half-life of approximately 24 hours.
A majority of the dose is hepatically metabolized by the CYP3A4 pathway, with
25% excreted unchanged in the urine.1
Dosing and Drug
Interactions
Aliskiren is
currently indicated for the treatment of hypertension, either alone or in
combination with other agents. Its use has not been studied in combination
with maximal doses of ACE inhibitors, and this type of combination should only
be used cautiously.1
Aliskiren should be taken once
daily, preferably at the same time each day. Patients will typically be
started at 150 mg and titrated up to 300 mg per day as needed. Doses above 300
mg per day do not result in additional BP reduction and are not approved.
1
Aliskiren does not appear to
inhibit or induce known hepatic metabolic pathways. Through an unknown
mechanism, the medication seems to reduce the blood levels of furosemide by
approximately 50%. Both ketoconazole and atorvastatin increased aliskiren
levels, while irbesartan appears to decrease aliskiren levels. The clinical
significance of these interactions is unknown. Aliskiren has not been studied
in patients receiving warfarin therapy.1
Practical Considerations
and Patient Counseling
Aliskiren shares
many of the same practical considerations for pharmacists as other classes of
medications that act on the RAS. As with ACE inhibitors and ARBs, RAs may
cause severe angioedema or subcutaneous swelling. This toxicity can involve
any part of the body and can occur at any point of drug therapy. In the event
that the larynx, tongue, or mouth is involved, this reaction can be life
threatening and patients should seek immediate emergency medical care.
Increases in serum potassium are also common to ACE inhibitors, ARBs, and RAs.
Combinations of any of these agents will increase potassium levels more than
any agent used alone.1
Pregnancy is a
contraindication to the use of any agent working directly on the RAS. The
fetal toxicities with ACE inhibitors and ARBs are thought to occur in the
second and third trimesters, and the same is assumed to be true with the RAs.
1
Aliskiren has been shown to
significantly reduce the blood levels of furosemide. Patients receiving these
two agents concurrently should be monitored closely for loss of furosemide
efficacy. Aliskiren does not appear to cause rebound hypertension when
discontinued abruptly. Although patients should be counseled for compliance,
noncompliance may not be as dangerous with this class as with agents that
cause rebound hypertension (i.e., beta-blockers and direct vasodilators). The
most common adverse effect of aliskiren is mild diarrhea, which usually
resolves within the first few weeks of therapy.1
Clinical Trials
The efficacy of
aliskiren has been tested in various clinical trials, including
placebo-controlled studies, comparator studies with other antihypertensives,
and in combination with other antihypertensives.1 Aliskiren has
been compared to or combined with other antihypertensive agents including
hydrochlorothiazide (HCTZ) diuretics, ARBs (valsartan, irbesartan, and
losartan), ACE inhibitors (ramipril and enalapril), and dihydropyridine CCBs
(amlodipine) in patients with uncomplicated hypertension.1-6 There
are no comparative or combination trials with beta-blockers, loop diuretics,
alpha-blockers, alpha-agonists, or direct vasodilators. HCTZ diuretics, ACE
inhibitors, and ARBs each cause reactive rises in PRA. Therefore, it is
hypothesized that these drugs used in combination with aliskiren will inhibit
the rise in renin. Comparator trials will be discussed in this review.
Aliskiren Monotherapy:
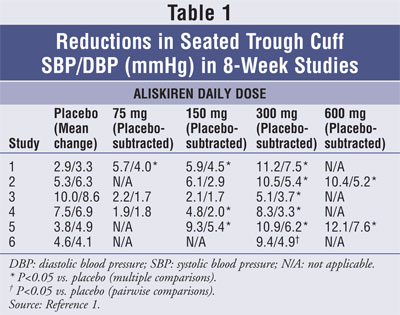
Antihypertensive effects of aliskiren have been displayed in six randomized,
double-blind, placebo-controlled, eight-week clinical trials in patients with
mild-to-moderate hypertension (TABLE 1). The studies included 2,730
patients given aliskiren doses of 75 to 600 mg; 1,231 patients received
placebo. All aliskiren doses decreased BP within two weeks of treatment, with
reasonable efficacy seen at 150 to 300 mg, and no clear further efficacy at
600 mg. Patients in the placebo-controlled trials continued open-label
aliskiren for up to one year. A statistically significant difference between
patients kept on aliskiren and those randomized to placebo was demonstrated by
a withdrawal study in which patients were randomized to continue either the
drug or placebo. With treatment cessation, BP gradually returned toward
baseline levels over a period of several weeks with no evidence of rebound
hypertension. BP was lowered in all demographic subgroups. African-American
patients tended to have smaller reductions compared with Caucasians and
Asians, as observed with ACE inhibitors and ARBs.1
Aliskiren Monotherapy/in
Combination with HCTZ:
Varying doses of aliskiren (75, 150, or 300 mg) and HCTZ (6.25, 12.5, or 25
mg) were studied alone and in combination in an eight-week, randomized,
placebo-controlled trial of 2,776 patients with mild-to-moderate hypertension.
The primary objectives were to assess the changes in mean sitting diastolic
blood pressure (MSDBP ?95 mmHg) and mean sitting systolic blood pressure
(MSSBP 110 mmHg) at baseline. The effects on PRA were also assessed. BP
reductions were greater with the combinations than with monotherapy.
Responders were defined as achieving a MSDBP 90 mmHg and/or a >10 mmHg
reduction from baseline. BP control was defined as a MSDBP 90 mmHg and a MSSBP
140 mmHg. Superior responder rates occurred with aliskiren 300 mg (63.9%; P
=0.0005), HCTZ 12.5 and 25 mg (60.6% and 59.0%, respectively; both P
0.02), and all combination doses compared with placebo. Better control and
responder rates for all combinations of aliskiren (75 to 300 mg) with HCTZ 25
mg and aliskiren 300 mg/HCTZ 12.5 mg were superior to both monotherapies (P
0.05). Aliskiren reduced PRA by up to 65% from baseline. PRA decreased in all
of the combination groups; however, HCTZ alone increased PRA by up to 72%.
Aliskiren monotherapy provided significant BP lowering, which was greater when
combined with HCTZ. The compensatory rise in PRA induced by HCTZ can be
neutralized by renin inhibition with aliskiren.2
Aliskiren Monotherapy/in
Combination with Valsartan:
Aliskiren and valsartan were investigated as monotherapy and in combination
in an eight-week, randomized, placebo-controlled, dose-escalation trial of
1,797 patients with uncomplicated hypertension.1,3 Patients
received either aliskiren 150 mg, valsartan 160 mg, the combination of
aliskiren 150 mg/valsartan 160 mg, or placebo. After four weeks, all
medication doses were doubled and therapy continued for an additional four
weeks. Seated trough cuff BP was measured at baseline, four weeks, and eight
weeks. BP reductions with the combinations were greater than the reductions
with both monotherapies. The aliskiren 300 mg/valsartan 320 mg combination had
statistically superior reductions in MSSBP and MSDBP compared with aliskiren
300 mg alone and valsartan 320 mg alone at week 8. Aliskiren used in
combination with valsartan may provide additive and tolerable BP-lowering
effects.4
Aliskiren versus and in
Combination with Ramipril:
In an eight-week, double-blind, multicenter study, 837 patients with
hypertension (MSDBP ?95 mmHg and 110 mmHg) and type 1 or type 2 diabetes
mellitus were randomized to one of three treatment groups: aliskiren 150 mg,
ramipril 5 mg, or aliskiren 150 mg plus ramipril 5 mg. The doses of aliskiren
and ramipril were doubled after four weeks. The primary end point was the
change from baseline to end point (week 8) in MSDBP. The combination of
aliskiren and ramipril resulted in statistically significant decreases in
MSDBP from baseline to end point (-12.78 mmHg) compared with aliskiren alone
(-11.32 mmHg) and ramipril alone (-10.71 mmHg). The aliskiren/ramipril
combination also resulted in statistically superior reduction in MSSBP from
baseline to end point compared with ramipril monotherapy (-4.63 mmHg). After
eight weeks, the proportion of patients classified as responders was
statistically superior in the aliskiren/ramipril group (73.12%) compared with
ramipril (65.82%), but was similar compared with aliskiren alone (74.09%).
Elevated potassium levels (>5.5 mmol/L) were noted for more patients in the
aliskiren/ramipril combination group (5.5%) compared with aliskiren (2.2%) and
ramipril monotherapy (2.6%).5
Aliskiren and HCTZ
versus Combination Therapy:
In a double-blind, multicenter trial, 560 obese adults (BMI ?30 kg/m2
) with arterial hypertension (MSDBP ?95 mmHg and 110 mmHg at baseline)
received HCTZ 25 mg daily for four weeks. Nonresponders (MSDBP ?90 mmHg and
110 mmHg) were randomized to four-week, double-blind treatment with aliskiren
150 mg, irbesartan 150 mg, amlodipine 5 mg, or placebo as HCTZ was continued.
After four weeks, the doses of aliskiren, irbesartan, and amlodipine were
doubled and therapy continued for an additional four weeks. The primary
objective was to compare the change in MSDBP from baseline to end point (week
8). A total of 489 out of the 560 patients who received four-week treatment
with HCTZ 25 mg were randomly assigned to double-blind aliskiren/HCTZ (n=122),
irbesartan/HCTZ (n=119), amlodipine/HCTZ (n=126), or placebo/HCTZ (n=122).
Aliskiren 300 mg in combination with HCTZ 25 mg resulted in greater reductions
in MSDBP and MSSBP compared with HCTZ 25 mg alone at week 8 (P0.0001).
Doubling the aliskiren dose from 150 to 300 mg (in addition to HCTZ 25 mg)
resulted in greater BP reductions at week 4 versus week 8. It was concluded
that combination treatment with aliskiren and a thiazide diuretic is an
effective and well-tolerated therapeutic option for obese patients with
hypertension who fail to achieve BP control with first-line thiazide diuretic
treatment alone.
Cost
Aliskiren is an expensive choice for
an antihypertensive. A
month's supply of the 150-mg dose costs approximately $70, and the 300-mg dose
is $90.7
Place in Therapy
Current hypertension guidelines
do not mention the role of RAs in the management of hypertension or
hypertension-induced diseases. Those patients most likely to benefit from the
reduction of renin activity (i.e., patients with diabetes, diabetes-induced
renal disease, and heart failure) were excluded from many of the currently
available trials evaluating aliskiren. At this point, the rationale for the
appropriate use of aliskiren is extremely limited.
The combination product of aliskiren and HCTZ was approved by the FDA in
January 2008 for the treatment of hypertension, but not as initial drug
therapy.8
Conclusions
A
liskiren is the first representative of the RA class of antihypertensives. In
all of the reviewed studies, combination therapy with aliskiren plus another
antihypertensive agent was significantly more effective in decreasing BP than
both agents alone. Comparative trial data show that aliskiren monotherapy has
effects on potassium concentrations similar to ACE inhibitors and additive
effects when used in combination. Further studies are needed to determine if
aliskiren is as beneficial as ACE inhibitors or ARBs in patients with heart
failure, diabetes, chronic kidney disease, and high risk of c
oronary disease.
REFERENCES
1. Tekturna (aliskiren) package insert. East Hanover, NJ: Novartis; March 2007.
2. Villamil A, Chrysant SG, Calhoun D, et al. Renin inhibition with aliskiren provides additive antihypertensive efficacy when used in combination with HCTZ. J Hypertens. 2007; 25:217-226.
3. Tekturna (aliskiren) Formulary Submission Dossier. Prepared by Novartis US Clinical Development and Medical Affairs. Release date: March 16, 2007.
4. Pool JL, Schmieder RE, Azizi M, et al. Aliskiren, an orally effective renin inhibitor, provides antihypertensive efficacy alone and in combination with valsartan. Am J Hypertens. 2007;20:11-20.
5. Uresin Y, Taylor A, Kilo C, et al. Aliskiren, a novel renin inhibitor, has greater BP lowering than ramipril and additional BP lowering when combined with ramipril in patients with diabetes and hypertension. J Hypertens. 2006;24(suppl 4):S82.
6. Jordan J, Engeli S, Boye SW, et al. Direct renin inhibition with aliskiren in obese patients with arterial hypertension. Hypertension. 2007;49:1047-1055.
7. Prescription price. Tekturna. Available at: www.drugstore.com. Accessed December 14, 2007.
8. Tekturna HCT (aliskiren and
hydrochlorothiazide) package insert. East Hanover, NJ: Novartis; January 2008.
To comment on
this article, contact editor@uspharmacist.com.





