US Pharm. 2011;36(5):41-44.
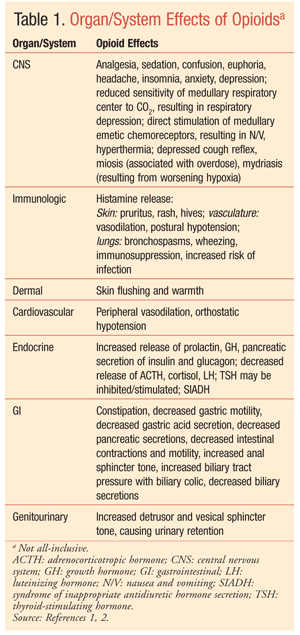
The rapid increase in the use of opioids for chronic noncancer pain (CNCP) over the past decade has exposed larger numbers of people to the wide-ranging side effects of opioid therapy. Several side effects—constipation, respiratory depression, and sedation—are well known. However, lesser-known side effects are occurring in more people; these deserve attention so that appropriate measures are taken to ameliorate them whenever possible. TABLE 1 lists many of the effects, desirable and undesirable, associated with opioid use.1,2
One condition that was considered rare until recently is narcotic bowel syndrome (NBS). NBS is one of several conditions in the broader category of opioid bowel dysfunction (OBD).3 OBD is most frequently thought of as constipation; however, the condition also involves nausea, bloating, ileus, reflux, and—less frequently—paradoxical worsening of stomach pain.4-8 NBS is distinguished from other OBDs by chronic or frequent stomach pain associated with opioid use that is worsened by increasing doses or continued use of opioids.9,10
This article differentiates NBS from other gastrointestinal (GI) conditions with similar presentation, suggests clues that can help medical professionals detect NBS in their patient populations, and reviews actions currently recommended for addressing NBS. The possibility of inappropriate treatment is also discussed.
NBS is notable for several reasons, including its impact on quality of life, the difficulty of recognizing its presence, and the possibility of treating stomach pain inappropriately. The issue of inappropriate therapy has been discussed in terms of increasing opioid use to alleviate pain; however, the inappropriateness of other types of therapies goes unrecognized. One category of medications that may be employed erroneously for NBS is acid-suppression therapy. The ease of access to OTC proton pump inhibitors (PPIs) and histamine2-receptor antagonists (H2-RAs), combined with strong advertising efforts by manufacturers, gives one cause to wonder whether acid-suppressive medications are being used to treat stomach pain related to NBS and are being overlooked as a clue to the presence of NBS.
Proposed Mechanisms for Development of NBS
It is posited that NBS is a form of hyperalgesia. Therefore, the mechanisms proposed for hyperalgesia are applicable to NBS. Several theories describe the paradoxical conditions of pain caused by opioid analgesics. The three theories presented here involve excitatory pathway activation; cholecystokinin (CCK) and release of dynorphin with subsequent antianalgesic effect; and direct and indirect effects on glial cells that result in opioid tolerance and expression of opioid-induced pain. All of these theories are quite complex. The following summaries are simplified explanations that demonstrate the relation of prolonged opioid use to the increased likelihood of hyperalgesia and NBS.
The first theory involves the discovery of the excitatory G-coupled opioid receptor GS. Opioid receptors contain both inhibitory G-coupled receptors (GI/GO) and excitatory GS-coupled receptors. The inhibitory GI/GO-coupled receptors are responsible for producing analgesia. In contrast, the GS-coupled receptors produce antianalgesia. It is proposed that, since tolerance to opioid analgesia develops over time, the antianalgesic effects of these GS-coupled receptors produce hyperalgesia.11,12
A second proposed mechanism for the development of hyperalgesia implicates the activity of CCK in the rostral ventromedial medulla (RVM). It is suggested that prolonged exposure to opioids may provoke adaptations in the brain and spinal cord known as neuroplastic changes. The increased activity of CCK in the RVM triggers a sequence of responses that result in activation of a descending pain-facilitation pathway, increased levels of spinal dynorphin, and release of excitatory transmitters.13,14 The end result is hyperalgesia.
The third theory is an extension of the RVM hypothesis. This theory focuses on spinal cord glial cells and the release of dynorphin. Data from a study conducted by Gardell et al support the concept of a morphine-induced RVM cascade resulting in elevated spinal dynorphin.15 The study further demonstrated that dynorphin is involved in the release of excitatory transmitters from primary afferent fibers.15
All of these theories support the long-recognized relationship between long-term opioid use and hyperalgesia. It is also recognized that NBS is a form of hyperalgesia.10
Identification and Prevalence of NBS
NBS has been recognized in published papers for decades.16 Despite alerts that NBS is “clinically important and frequently unrecognized”16 or “often overlooked,”9 the disorder remains seldom diagnosed. The difficulty in identifying NBS in practice is related to the predictable and well-known actions of opioids on the GI system and the similarities between NBS symptoms and more common conditions, such as irritable bowel syndrome or gastritis. For example, a patient with NBS may experience vomiting, weight loss, bloating, or constipation, all of which are commonly associated with opioid use.4,8,17 TABLE 2 compares the symptoms of several GI disorders with those of NBS.10,18-20 The distinguishing symptom of NBS is abdominal pain that is associated with continued opioid use or dose increases.9,10

Diagnosing NBS can be time-consuming. When a patient receiving chronic opioid therapy presents with stomach pain, NBS may be considered if the following conditions are ruled out: peptic ulcer, gastritis, gallstones, pancreatitis, intermittent partial mechanical obstruction, abdominal angina, renal calculi, uterine fibroids, and ovarian cysts.9 Criteria for diagnosis include chronic or acute high-dose opioid (HDO) use, pain that worsens in duration and intensity with increased opioid doses, and pain frequency that increases with continued opioid use. A final criterion is that other conditions are absent or cannot adequately explain the pain.10 A confounding symptom that adds to the difficulty of diagnosing NBS is “soar and crash,” which is characterized by worsened pain when the opioid effect fades and improvement when the medication is reintroduced.10 The time required to rule out conditions and make a diagnosis of NBS may contribute to lack of recognition.
Only recently is the incidence of NBS being determined. Most reports describing side effects related to chronic opioid use focus on constipation as the only significant GI side effect; however, the incidence of NBS is estimated to be between 4.2% and 6.4%.3,21 In a retrospective study by Choung et al, self-report questionnaires were mailed to residents of a community in Minnesota; respondents were assessed for GI complaints, and the data were stratified according to use and nonuse of opioids.3 Four percent of respondents were chronic opioid users; of these, 4.2% were identified as “probably experiencing NBS.” Tuteja et al studied CNCP patients treated at a university-based pain-management clinic.21 In this population, 58.2% of respondents reported chronic abdominal pain and 6.4% met the criteria for NBS.
Although NBS was previously considered rare, these studies imply that 1 in 20 chronic opioid users may be at risk for this disorder. Therefore, it is important for physicians, nurses, and pharmacists to be alert to symptoms so that appropriate action may be taken to address the pain caused by opioids. In particular, pharmacists in the community and hospital settings may be in a unique position to discover patients with possible NBS.
CNCP Patients, NBS, and the Pharmacist
Articles describing NBS indicate that prescribers who are unaware of the syndrome attempt to alleviate patients’ pain by increasing the opioid dose, which results in increased pain.10,22 These efforts often result in an escalating cycle of dose increases followed by increased pain, not to mention frustration for the patient and the prescriber. The published articles discussing NBS do not address whether acid-suppression therapy is employed to treat the GI pain. The idea that this may be occurring led to an effort to ascertain whether acid-suppressive medications are, in fact, being used.
To investigate this possibility, preliminary data mining at a pharmacy benefit management (PBM) company specializing in workers’ compensation was conducted over a 3-month period (October 1–December 31, 2010).23 Prescription activity was collected for all patients receiving high-dose opioid (HDO) therapy (³150 mg morphine equivalent daily). The report listed all medications submitted through patients’ workers’ compensation claims and yielded 1,524 patients on HDO therapy. Medication lists for all patients were reviewed for the use of nonsteroidal anti-inflammatory drugs (NSAIDs), including oral, topical, nonselective, and selective cyclo-oxygenase-II agents, aspirin, warfarin, and PPIs and/or H2-RAs. Patients receiving HDO therapy with transactions for PPIs and/or H2-RAs but without NSAID, aspirin, or warfarin therapy were considered potential NBS candidates. It was determined that 118 patients (7.7% of total number of patients on daily HDOs) met these criteria. Further analysis of the data is in progress to investigate the intended use of these acid-suppression therapies. It is suspected that at least some of these patients are experiencing stomach pain and perhaps NBS, and are using PPIs and/or H2-RAs in an attempt to alleviate the pain.
Although acid-suppressive medications are considered quite safe, inappropriate acid-suppression therapy is a concern.24,25 OTCs’ availability and perceived safety by patients and medical professionals may foster unnecessary use of these medications, which may be problematic. Further, the clinical significance of opioid-induced reduction of gastric acid secretion paired with acid-suppression therapy with PPIs or H2-RAs is unknown. For these reasons, the use of PPIs and H2-RAs deserves closer attention and assurance of appropriate use in patients taking chronic opioids.
Recommendations to Pharmacists
Pharmacists practicing in the hospital setting are in an excellent position to identify potential NBS. A CNCP patient may be suspected of having stomach pain if, upon admission, his or her home medications do not include NSAIDs, but include HDOs and PPIs or H2-RAs. A review of medications against diagnoses may reveal a case of stomach pain or NBS. If NBS is underdiagnosed, as suggested, further investigation to rule out NBS may be appropriate.21 Educating the prescriber about NBS may be beneficial to the patient. Questions the pharmacist may pose to the prescriber include: Does the patient continue to experience GI pain despite acid-suppression therapy? Does the pain worsen when the opioid dose is increased? If other conditions are present, could they be the source of the pain?
Pharmacists practicing in the community setting also have the opportunity to identify CNCP patients with stomach pain. If the pharmacist knows the patient and the patient purchases OTC PPIs or H2-RAs or has a prescription for one, the pharmacist is in a position to speak with the patient. The questions noted above may assist the pharmacist in identifying a condition requiring further evaluation by the patient’s physician. If the pharmacist is engaged in providing medication therapy management, sending a follow-up written observation to the prescriber may be in order.
Recommendations to Prescribers
Underdiagnosis of NBS may persist for a number of reasons. Because opioids affect the GI system in many ways, stomach pain may be attributed to any one of several GI effects. Additionally, NBS has been considered rare and has received little attention in the literature. However, in the past several years, reports of increased NBS incidence have heightened awareness of this condition. Finally, it may be a time-consuming process to rule out other conditions and determine the likelihood of NBS.
Prescribers may want to consider a diagnosis of NBS in their patients receiving chronic opioid therapy. An assessment of the correlation between opioid dose and pain intensity may help identify the presence or absence of NBS. Diagnosis is confirmed when stomach-pain intensity is not associated with other GI disorders, but is correlated to opioid dose increases.10 The prescriber also should question the patient about OTC acid-suppressive medication use and whether the stomach pain eases when these products are used. If NBS is believed to be present, appropriate steps should be taken to alleviate the pain.
To date, the only recommended treatment for NBS is opioid dose reduction or discontinuation of opioid therapy.10,26 Nonopioid analgesic therapy, antidepressant therapy, and nonpharmacologic therapies may be needed to address chronic pain and improve the patient’s quality of life.22 Grunkemeier et al have published a comprehensive review of NBS, including guidance for narcotic withdrawal and the use of pharmacotherapies and psychological support to facilitate the transition from opioid therapy.10
Conclusion
Recent data suggest that NBS is more prevalent than was previously thought. Better recognition of NBS may lead to appropriate addressing of this condition and improved quality of life for the patient. More studies are needed to determine whether patients with unrecognized NBS are being unnecessarily treated with acid-suppressive medications. Preliminary data from a PBM company’s database are intriguing. Further investigation is underway that may contribute to a better understanding of the prevalence of NBS and how unnecessary treatment may be avoided.
More needs to be discovered about the effects of long-term opioid use. Questions that deserve further consideration include: Who is at risk for developing NBS? What is the clinical significance of acid suppression that occurs from opioid use alone? Does the concomitant use of acid-suppressive medications expose the patient to unnecessary risks?27 After centuries of use, opioid therapy remains a field of intense interest and abundant research opportunities.
REFERENCES
1. Gutstein HB, Akil H. Opioid analgesics. In: Brunton LL, Lazo JS, Parker KL, eds. Goodman & Gilman’s The Pharmacological Basis of Therapeutics. 11th ed. New York, NY: McGraw-Hill; 2006:547-590.
2. MS Contin (morphine sulfate) package insert. Stamford, CT: Purdue Pharma L.P.; March 2009.
3. Choung RS, Locke GR III, Zinsmeister AR, et al. Opioid bowel dysfunction and narcotic bowel syndrome: a population-based study. Am J Gastroenterol. 2009;104:1199-1204.
4. Kurz A, Sessler DI. Opioid-induced bowel dysfunction: pathophysiology and potential new therapies. Drugs. 2003;63:649-671.
5. Bell TJ, Panchal SJ, Miaskowski C, et al. The prevalence, severity, and impact of opioid-induced bowel dysfunction: results of a US and European patient survey (PROBE 1). Pain Med. 2009;10:35-42.
6. Panchal SJ, Müller-Schwefe P, Wurzelmann JI. Opioid-induced bowel dysfunction: prevalence, pathophysiology and burden. Int J Clin Pract. 2007;61:1181-1187.
7. Furlan AD, Sandoval JA, Mailis-Gagnon A, Tunks E. Opioids for chronic noncancer pain: a meta-analysis of effectiveness and side effects. CMAJ. 2006;174:1589-1594.
8. Pappagallo M. Incidence, prevalence, and management of opioid bowel dysfunction. Am J Surg.
9. Rogers M, Cerda JJ. The narcotic bowel syndrome. J Clin Gastroenterol. 1989;11:132-135.
10. Grunkemeier DM, Cassara JE, Dalton CB, Drossman DA. The narcotic bowel syndrome: clinical features, pathophysiology, and management. Clin Gastroenterol Hepatol. 2007;5:1126-1139.
11. Crain SM, Shen KF. Antagonists of excitatory opioid receptor functions enhance morphine’s analgesic potency and attenuate opioid tolerance/dependence liability. Pain. 2000;84:121-131.
12. Gintzler A, Chakrabarti S. Opioid tolerance and the emergence of new opioid receptor-coupled signaling. Mol Neurobiol. 2000;21:21-33.
13. Ossipov MH, Lai J, King T, et al. Underlying mechanisms of pronociceptive consequences of prolonged morphine exposure. Biopolymers. 2005;80:319-324.
14. Vanderah TW, Gardell LR, Burgess SE, et al. Dynorphin promotes abnormal pain and spinal opioid antinociceptive tolerance. J Neurosci. 2000;20:7074-7079.
15. Gardell LR, Wang R, Burgess SE, et al. Sustained morphine exposures induces a spinal dynorphin-dependent enhancement of excitatory transmitter release from primary afferent fibers. J Neurosci.
16. Sandgren JE, McPhee MS, Greenberger NJ. Narcotic bowel syndrome treated with clonidine: resolution of abdominal pain and intestinal pseudo-obstruction. Ann Intern Med. 1984;101:331-334.
17. Mehendale SR, Yuan CS. Opioid-induced gastrointestinal dysfunction. Dig Dis. 2006;24:105-112. 2001;182:11S-18S. 2002;22:6747-6755.
18. Irritable bowel syndrome—symptoms. Mayo Clinic Health Information.
www.mayoclinic.com/health/
irritable-bowel-syndrome/
DS00106/DSECTION=symptoms. Accessed December 27, 2010.
19. Gastritis—symptoms. Mayo Clinic Health Information.
www.mayoclinic.com/health/
gastritis/DS00488/DSECTION=
symptoms. Accessed December 27, 2010.
20. Chronic pancreatitis. The Merck Manuals Online Medical Library—Home Edition.
www.merckmanuals.com/home/
sec09/ch124/ch124c.html#sec09-
ch124-ch124c-443. Accessed December 27, 2010.
21. Tuteja AK, Biskupiak J, Stoddard GJ, Lipman AG. Opioid-induced bowel disorders and narcotic bowel syndrome in patients with chronic non-cancer pain. Neurogastroenterol Motil. 2010;22:424-430.
22. Drossman DA. Beyond tricyclics: new ideas for treating patients with painful and refractory functional gastrointestinal symptoms. Am J Gastroenterol. 2009;104:2897-2902.
23. Vertice [database online]. Tampa, FL: Healthesystems; 2011. Updated January 1, 2011.
24. Ali T, Roberts DN, Tierney WM. Long-term safety concerns with proton pump inhibitors. Am J Med.
25. Thomson AB, Sauve MD, Kassam N, Kamitakahara H. Safety of the long-term use of proton pump inhibitors. World J Gastroenterol.
26. Drossman DA. Severe and refractory chronic abdominal pain: treatment strategies. Clin Gastroenterol Hepatol. 2008;6:978-982. 2009;122:896-903.
27. Lin KM, Ueda RK, Hinder RA, et al. Etiology and importance of alkaline esophageal reflux. Am J Surg. 1991;162:553-557.
To comment on this article, contact rdavidson@uspharmacist.com.






