US Pharm. 2015;40(5):HS35-HS42.
ABSTRACT: Fall risk in the elderly is a significant concern when considering treatment for insomnia. Falls are the leading cause of fatal and nonfatal injuries among adults aged ≥65 years. Clinicians have relied on short-term use of nonbenzodiazepine hypnotics (NBZDHs) to assist with sleep onset and quality. However, the American Geriatrics Society recommends nonpharmacologic alternatives to assist with insomnia in the elderly, identifying NBZDHs as high-risk medications. Elderly patients should be informed of nonpharmacologic options available for the treatment of insomnia, the risks associated with drugs used to treat insomnia, and fall prevention strategies to protect their health and well-being.
It has been observed that the prevalence of sleep-related disorders, such as insomnia, increases as the population ages.1 The estimated prevalence of insomnia in the elderly is 13% to 47%, affecting approximately 8 million seniors nationwide.2 This increase in prevalence of insomnia within our geriatric population is often the result of a higher presence of medical and/or psychosocial comorbidities; however, significant physiological changes begin to occur with age, affecting the daily sleep-wake cycle.1 To combat this growing issue, clinicians have relied on short-term use of nonbenzodiazepine hypnotics (NBZDHs) to assist with sleep onset as well as sleep quality. But are these medications truly a safe option for the elderly?
Fall Risk and Fractures
Falls and fractures can significantly impact the functional capacity and mortality of the elderly population. According to the CDC, falls are the leading cause of fatal and nonfatal injuries among adults aged ≥65 years.1 The incidence of insomnia can contribute to this statistic, serving as a risk factor for falls.3 In addition, fractures significantly impact an elderly patient’s quality of life; 1 out of 3 elderly adults who live independently prior to their hip fracture will remain in a nursing home for at least a year.4 Hip fractures are very serious injuries in the elderly, with 95% resulting from falls and 1 out of every 5 patients dying within a year of the injury.4
It is also noteworthy that many drugs that treat comorbid diseases, such as antidepressants and narcotics, and disease states that cause drowsiness, dizziness, and cognitive impairment, such as dementia, contribute to the risk of falls in this patient population, further increasing the risk for fracture.5,6
Fall risk in the elderly is a significant concern when contemplating treatment for insomnia in this population. Currently, the American Geriatrics Society (AGS) recommends nonpharmacologic alternatives to assist with insomnia in the elderly, and has identified the NBZDHs zolpidem (Ambien), eszopiclone (Lunesta), and zaleplon (Sonata) as “high-risk” medications.7 In addition, it has outlined other “potentially inappropriate medications” that can lead to falls or fractures in the elderly (TABLE 1).8
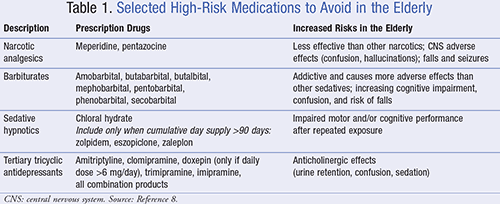
The Centers for Medicare and Medicaid Services (CMS) has incorporated the AGS recommendations into its Part D star ratings program. CMS has developed the Five-Star Quality Rating System designated for Medicare Advantage plans, with the purpose of helping beneficiaries identify health plans with higher quality of care. These plans are financially incentivized by CMS for achieving a five-star rating, benefiting both the plan and the beneficiary. There are 18 measures constituting the Part D star rating, of which five are medication-related and heavily weighted in terms of importance. Endorsed by the Pharmacy Quality Alliance (PQA) and adopted by CMS, these measures include: 1) high-risk medications (HRMs); 2) diabetes treatment and medication adherence; 3) diabetes medications; 4) hypertension medications; and 5) cholesterol medications. The HRM Part D star rating measure states that the star rating will be negatively impacted if people aged ≥65 years have received a prescription fill for zolpidem, eszopiclone, or zaleplon with the cumulative supply for those fills >90 days. The rationale for this measure’s restrictions is the fact that these agents should be used only in the short-term and can cause impaired motor and/or cognitive performance after repeated exposure.8,9
Medicare has taken a stance regarding NBZDHs, yet these medications continue to be prescribed. This raises the question: Are NBZDHs a viable treatment choice in the elderly? The intent of this article is to review the effectiveness of NBZDHs and evaluate their risk for falls and fractures, specifically in the elderly population, based on recent studies.
Pharmacologic Options: Focus on NBZDHs
NBZDHs are frequently used to treat insomnia in the elderly due to their ability to cause sedation with less risk of side effects when compared to benzodiazepines (BZDs; e.g., alprazolam, lorazepam, diazepam).10 The most commonly prescribed NBZDHs include zolpidem and eszopiclone; zaleplon is effective for decreasing time to sleep onset but not for reducing nighttime awakening or for increasing total sleep time.10,11 Approximately 2.8 million Americans aged ≥60 years use a medication for the treatment of insomnia (common drugs, FDA-indicated or off-label, include NBZDHs, BZDs, barbiturates, doxepin, quetiapine, ramelteon, and trazodone).11 In 2010, roughly 2.5 million Americans used NBZDHs, with 87.5% taking zolpidem.11 It is worth noting that these prescription options are also Schedule IV controlled substances and may have restrictions in the state where they are dispensed.
The mechanism of action for NBZDHs is similar to that of BZDs. They are benzodiazepine receptor agonists that are more selective than BZDs for the gamma-aminobutyric acid type A (GABAA) receptors that contain the alpha1 subunit. Binding to the receptors, NBZDHs enhance GABA-induced ionic currents through the ligand-gated chloride channels, promoting inhibitory effects such as drowsiness.12
Zaleplon is effective for sleep-onset insomnia (i.e., it improves sleep latency), but it has not been shown to improve duration of sleep and number of awakenings due to its short half-life; it is FDA-approved for short-term use only.12 Zolpidem is effective for sleep-onset insomnia and sleep maintenance (the controlled-release dosage form) and has been shown to be effective for up to 6 months, but it is FDA-approved for only short-term use as well.12 In addition, in 2013 the FDA established new requirements for zolpidem-containing products, lowering the recommended initial dose to 5 mg for the immediate-release formulation and 6.25 mg for the extended-release formulation. Warnings for next-morning impairment of driving and other activities when using the 6.25- and 12.5-mg extended-release formulations of zolpidem were added as well.13 Eszopiclone is effective for sleep-latency and sleep maintenance, and it has shown to be effective for up to 12 months. Eszopiclone is the only NBZDH FDA-approved for long-term use.12 TABLE 2 provides a comparison of these agents.14,15

When comparing adverse effects of these agents in the elderly, zolpidem was associated with more somnolence than zaleplon (TABLE 3).15 The effects on sleep latency are similar in the elderly population. However, eszopiclone has shown to increase sleep duration more than zolpidem by about 30 minutes.14

Both untreated insomnia and medications used to treat insomnia have been associated with increased risk of falls in the elderly. According to a study that examined residents aged ≥65 years in nursing homes in Michigan, untreated insomnia and insomnia that was unresponsive to hypnotics treatment were associated with increased risk of falls.16 The adjusted odds ratio (OR) was 1.55 and 1.32, respectively. In contrast, the study did not show an increased risk of falls associated with hypnotics use.16 However, a study published in 2013 found that hypnotics (zolpidem, zopiclone, and others were included) were associated with falls in patients with a mean age of 64.7 years.17 In addition, zolpidem has been repeatedly observed to be associated with falls in the inpatient setting.17,18
Studies looking at NBZDHs and falls in the outpatient setting are scarce, but numerous studies in the inpatient setting show increased risk of falls when a patient is taking a hypnotic medication. Even when older patients stop taking the medication, they can experience impaired psychomotor functioning, which can persist for at least 6 months after withdrawal, placing the patient at risk for falls even after the drug has been discontinued.19
Fracture risk in the elderly is an important consideration when initiating pharmacologic therapy for any indication in this population. Fractures, especially hip fractures, can significantly decrease an elderly patient’s quality of life and increase the risk of mortality.4 In a retrospective study published in 2014 by Lin et al, patients who took zolpidem had a 2.23-fold higher hip fracture incidence than nonusers.20 The patients included within the study were aged 18 years; however, when comparing the elderly subpopulation, the elderly zolpidem users were twice as likely to experience a hip fracture when compared to elderly nonusers.20 This study coincides with a previous case control study in 2001 where zolpidem was associated with a nearly twofold increased risk of hip fracture in patients who were ³65 years of age.21 In addition, Berry and colleagues observed that NBZDH users in the nursing home setting, with an average age of 81 years, had an increased risk of fracture compared to nonusers (OR 1.66, 95% CI, 1.45-1.90).22
In a 2014 study conducted in Norway, anxiolytics (e.g., diazepam, oxazepam, alprazolam, hydroxyzine) and NBZDHs (zopiclone and zolpidem) were revealed to share comparable incidence ratios for hip fractures (1.4 and 1.2, respectively).23 This finding was similar to the results Finkle et al observed when trying to determine whether zolpidem was a safer alternative to BZDs; the risk of injury with zolpidem exceeded that of alprazolam and lorazepam but was similar to that with diazepam.24
NBZDHs are generally thought of as safer than BZDs; however, recent observational data paint a very similar picture to BZDs, which are considered potentially inappropriate medications in the elderly.8,17,18,20,21,23 As discussed earlier, hip fractures pose a serious threat to a patient’s morbidity and mortality, and any medication associated with an increase risk of hip fracture should be strongly discouraged unless the benefits outweigh the risks.
Nonpharmacologic Options for Treating Insomnia
Cognitive behavioral therapy for insomnia (CBT-I) is a comprehensive restructuring of a patient’s sleeping behavior.1 CBT-I includes sleep hygiene instruction, stimulus control, sleep restriction, and cognitive restructuring (education on sleep and motivation to increase adherence).1 CBT-I has been shown to decrease total wake time by 55 to 60 minutes, increase slow-wave sleep (stages 3 and 4) by 17 to 21 minutes, and increase sleep efficiency (time asleep/time in bed) by 9% to 11%.25 The benefits of CBT-I have been shown to be sustained for up to 2 years.1 However, a literature search was not able to find information on CBT-I and its effects on falls and fractures in the elderly when compared to pharmacotherapy. This would be an interesting topic for further research.
While effective, CBT-I may not be available for all patients due to limitations on geographic area or insurance coverage. Access to a doctoral-level psychologist or other American Board of Sleep Medicine (ABSM)–certified specialists for CBT-I is inadequate for millions of Americans, and most primary care physicians are not sufficiently trained to perform CBT-I.26 CBT-I is covered by Medicare and TRICARE as a mental health service, but coverage under private insurances will be widely varied based on individual plan benefits.27
When comparing CBT-I to zopiclone (the racemic mixture opposed to eszopiclone, which is available in the United States, and the S-isomer), CBT-I decreased total wake time and increased sleep efficiency more than zopiclone when measured by polysomnography and use of a sleep diary (TABLE 4).28 In addition, CBT-I increased the amount of time in slow-wave sleep, while zopiclone decreased the amount of time in slow-wave sleep.25 It is believed that the lack of slow-wave sleep may be to blame for the presence of daytime impairment in patients experiencing insomnia.24 The efficacy of zolpidem was also less than CBT-I when measured by polysomnography according to an 8-week study published in 2004.28 The task of comparing adverse effects of CBT-I and NBZDHs is difficult due to the fact that there are very limited data for CBT-I. Clinical studies that evaluate CBT-I do not report adverse effects frequently. In addition, the head-to-head trials of CBT-I to zolpidem and CBT-I to zopiclone did not report any participant withdrawals due to side effects, with the exception of one participant who was taking zopiclone.28

Other nonpharmacologic options to combat insomnia include proper sleep hygiene.29 Avoiding caffeine or other stimulants, eating a light snack, setting the thermostat on a cool and comfortable temperature, and ensuring a relaxed bedroom atmosphere prior to bedtime are all recommended sleep hygiene practices. In addition, progressive muscle relaxation, stimulus control, and paradoxical intention all meet the American Academy of Sleep Medicine criteria for empirically supported psychological treatments for insomnia.29
Fall Prevention Strategies
The AGS has released a clinical practice guideline specifically for healthcare professionals regarding fall prevention strategies in older persons.30 Recommendations include a multifactorial fall risk assessment, followed by direct interventions tailored to the identified risk factors, coupled with an appropriate exercise program; an exercise program incorporating gait, balance, and strength training, with flexibility and endurance training offered as supplements, for fall-risk patients; and adaptation or modification of the home environment.30 It is imperative for healthcare professionals to review these strategies in addition to addressing the patient’s insomnia to avoid potentially dangerous fall-risk situations.
Conclusion
Insomnia is an important issue in the elderly population because the medications used to treat it are associated with significant adverse effects; however, the disease state itself produces similar negative effects. Falls and hip fractures need to be prevented in the elderly population because of their significant effects on morbidity, mortality, and quality of life. NBZDHs are associated with both increased risk of falls and increased risk of hip fractures in the elderly population.
The efficacy of NBZDHs is not superior to CBT-I, and the adverse effects of NBZDHs compound the increased risk of falls and fractures. Currently, there is no study identifying whether CBT-I is superior to NBZDHs in terms of fall rates; however, due to the low risk of adverse events, CBT-I should be considered as a first-line treatment option in the elderly population. Yet despite evidence supporting CBT-I’s effectiveness, many primary care physicians may not be aware of it or know how to refer patients for treatment.25 In addition, the availability of CBT-I is limited by the small number of qualified behavioral sleep medicine specialists and restricted to their geographic region; for Medicare and TRICARE recipients, coverage is not a limitation.27,31 Regardless of CBT-I’s purported efficacy, NBZDHs are highly prescribed in the elderly, even with risk for falls and hip fractures comparable to that of BZDs.
As part of a comprehensive counseling session, elderly patients should be informed of nonpharmacologic options available for the treatment of insomnia, the risks associated with the medications used to treat insomnia, and fall prevention strategies to protect their health and well-being. Further research is needed to evaluate the adverse events and fall and fracture risk for CBT-I when compared to pharmacotherapy.
REFERENCES
1. Bloom HG, Ahmed I, Alessi CA, et al. Evidence-based recommendations for the assessment and management of sleep disorders in older persons. J Am Geriatr Soc. 2009;57(5):761-789.
2. Doghramji K. The epidemiology and diagnosis of insomnia. Am J Manag C. 2006;12:S214-S220.
3. Rodrigues IG, Fraga GP, Barros MB. Falls among the elderly: risk factors in a population-based study. Rev Bras Epidemiol. 2014;17(3):705-718.
4. CDC. Hip fractures among older adults. www.cdc.gov/homeandrecreationalsafety/falls/adulthipfx.html. Accessed November 3, 2014.
5. Emblem Health. Falls in the elderly: drugs associated with increased fall risk. June 2009. www.emblemhealth.com/pdf/HIP_PR_FLY_805_Falls_Drugs_final091010.pdf. Accessed November 3, 2014.
6. Jørgensen TS, Hansen AH, Sahlberg M, et al. Nationwide time trends and risk factors for in-hospital falls-related major injuries. Int J Clin Pract. 2015 Feb 24 [Epub ahead of print].
7. Green AR, Lee SJ. Ask the geriatrician: sedative-hypnotic drugs and related medications. HealthinAging.org. February 2013. www.healthinaging.org/resources/resource:ask-the-geriatrician-sedative-hypnotic-drugs-and-related-medications. Accessed November 3, 2014.
8. American Geriatrics Society 2012 Beers Criteria Update Expert Panel. American Geriatrics Society updated Beers Criteria for potentially inappropriate medication use in older adults. J Am Geriatr Soc. 2012;60(4):616-631. www.americangeriatrics.org/files/documents/beers/2012BeersCriteria_JAGS.pdf. Accessed:November 3, 2014.
9. Pharmacy Quality Alliance 2013. Use of high-risk medications in the elderly (HRM). http://pqaalliance.org/images/uploads/files/HRM%20Measure%202013website.pdf. Accessed November 3, 2014.
10. Dopp JM, Phillips BG, Dopp JM, et al. Chapter 55. Sleep disorders. In: DiPiro JT, Talbert RL, Yee GC, et al, eds. Pharmacotherapy: A Pathophysiologic Approach. 9th ed. New York, NY: McGraw-Hill; 2014. http://accesspharmacy.mhmedical.com/content.aspx?bookid=689&Sectionid=45310506. Accessed November 3, 2014.
11. Bertisch SM, Herzig SJ, Winkelman JW, Buettner C. National use of prescription medications for insomnia: NHANES 1999-2010. Sleep. 2014;37(2):343-349.
12. Mihic S, Harris R, Mihic S, et al. Chapter 17. Hypnotics and sedatives. In: Brunton LL, Chabner BA, Knollmann BC, et al, eds. Goodman & Gilman’s The Pharmacological Basis of Therapeutics. 12th ed. New York, NY: McGraw-Hill; 2011. http://accesspharmacy.mhmedical.com/content.aspx?bookid=374&Sectionid=41266223. Accessed November 3, 2014.
13. FDA drug safety communication. FDA approves new label changes and dosing for zolpidem products and a recommendation to avoid driving the day after using Ambien CR. www.fda.gov/drugs/drugsafety/ucm352085.htm. Accessed March 5, 2015.
14. Carson S, McDonagh MS, Thakurta S, Yen PY. Drug Class Review: Newer Drugs for Insomnia. Final Report Update 2. Portland, OR: Oregon Health & Science University; October 2008. www.ncbi.nlm.nih.gov/pubmedhealth/PMH0008716/. Accessed November 3, 2014.
15. LexiComp Online. www.lexi.com. Accessed November 3, 2014.
16. Avidan AY, Fries BE, James ML, et al. Insomnia and hypnotic use, recorded in the minimum data set, as predictors of falls and hip fractures in Michigan nursing homes. J Am Geriatr Soc. 2005;53(6):955-962.
17. Obayashi K, Araki T, Nakamura K, et al. Risk of falling and hypnotic drugs: retrospective study of inpatients. Drugs R D. 2013;13(2):159-164.
18. Kolla BP, Lovely JK, Mansukhani MP, Morgenthaler TI. Zolpidem is independently associated with increased risk of inpatient falls. J Hosp Med. 2013;8(1):1-6.
19. Puustinen J, Lähteenmäki R, Polo-Kantola P, et al. Effect of withdrawal from long-term use of temazepam, zopiclone, or zolpidem as hypnotic agents on cognition in older adults. Eur J Clin Pharmacol. 2014;70(3):319-329.
20. Lin FY, Chen PC, Liao CH, et al. Retrospective population cohort study on hip fracture risk associated with zolpidem medication. Sleep. 2014;37(4):673-679.
21. Wang PS, Bohn RL, Glynn RJ, et al. Zolpidem use and hip fractures in older people. J Am Geriatr Soc. 2001;49(12): 1685-1690.
22. Berry SD, Lee Y, Cai S, Dore DD. Nonbenzodiazepine sleep medication use and hip fractures in nursing home residents. JAMA Intern Med. 2013;173(9):754-761.
23. Bakken MS, Engeland A, Engesaeter LB, et al. Risk of hip fracture among older people using anxiolytic and hypnotic drugs: a nationwide prospective cohort study. Eur J Clin Pharmacol. 2014;70(7):879-880.
24. Finkle WD, Der JS, Greenland S, et al. Risk of fractures requiring hospitalization after an initial prescription for zolpidem, alprazolam, lorazepam, or diazepam in older adults. J Am Geriatr Soc. 2011;59(10):1883-1890.
25. Sivertsen B, Omvik S, Pallesen S, et al. Cognitive behavioral therapy vs. zopiclone for treatment of chronic primary insomnia in older adults. JAMA. 2006;295(24):2851-2858.26. Fields BG, Schutte-Rodin S, Perlis ML, Myers M. Master’s-level practitioners as cognitive behavioral therapy for insomnia providers: an underutilized resource. J Clin Sleep Med. 2013;9(10):1093-1096.
27. Integrative Insomnia and Sleep Health Center. Frequently asked questions. www.integrativesleep.com/patient-frequently-asked-questions.html. Accessed March 9, 2015.
28. Mitchell MD, Gehrman P, Perlis M, Umscheid CA. Comparative effectiveness of cognitive behavioral therapy for insomnia: a systematic review. BMC Fam Pract. 2012;13:40.
29. McCall WV. Sleep in the elderly: burden, diagnosis and treatment. Prim Care Companion J Clin Psychiatry. 2004;6(1): 9-20.
30. American Geriatrics Society. AGS/BGS clinical practice guideline: prevention of falls in older persons. www.americangeriatrics.org/health_care_professionals/clinical_practice/clinical_guidelines_recommendations/prevention_of_falls_summary_of_recommendations. Accessed March 9, 2015
31. Siebern AT, Manber R. New developments in cognitive behavioral therapy as the first-line treatment of insomnia. Psychol Res Behav Manag. 2011;4:21-28.
32. Schutte-Rodin S, Broch L, Buysse D, et al. Clinical guideline for the evaluation and management of chronic insomnia in adults. J Clin Sleep Med. 2008;4(5):487-504.
To comment on this article, contact rdavidson@uspharmacist.com.






