US Pharm. 2010;35(11):29-40.
About 64% of adults in the United States consumed alcohol in 2008.1 In that same year, approximately 3.8 billion prescription medications were dispensed.2 Despite this high prevalence, few physicians discuss alcohol use with their patients.3,4 In addition, most patients do not consider alcohol a drug capable of interacting with medications. Several studies have indicated benefits of moderate alcohol consumption in various disease states; however, the media often discuss these study results without mentioning relevant details about the type of alcoholic beverage used or the patient population involved.5 They also do not review possible interactions of alcohol with other diseases or medications. Patients misinterpret such information to rationalize their alcohol use without consulting their physicians. Alcohol has been reported as a risk factor for adverse drug-related hospital admissions.3
In light of the prevalence of alcohol consumption, it is essential that pharmacists enlighten their patients about drug-alcohol interactions. Community pharmacists are at the frontline to assist patients in understanding their medications and possible drug interactions. The objective of this review is to serve as a guide, especially for community pharmacists, in understanding various drug-alcohol interactions and explaining their clinical significance to patients, with a focus on three major drug classes—antibiotics, cardiovascular medications, and analgesics.
According to the most recent estimates, about 10% of adults in the U.S. consume alcohol on a daily basis.6 A standard drink of alcohol is any beverage containing about 0.5 oz (15 g) of pure alcohol; i.e., 12 oz of beer, 5 oz of wine, or 1.5 oz of 80-proof distilled spirits.5,6 Moderate alcohol consumption is defined by the U.S. Department of Health and Human Services as one standard drink per day for women and two standard drinks per day for men.5
Pharmacokinetics and Pharmacodynamics of Alcohol
To understand how alcohol can interact with certain medications, it is important to briefly identify the pharmacokinetic and pharmacodynamic interactions involved with alcohol consumption. When alcohol is consumed, about 10% undergoes first-pass metabolism in the stomach, intestines, and liver. One of the major enzymes involved in alcohol metabolism is alcohol dehydrogenase (ADH), which converts alcohol to acetaldehyde, a toxic compound that is subsequently metabolized by aldehyde dehydrogenase (ALDH) to acetate. Following first-pass metabolism, alcohol is distributed into various body tissues and exerts its effects. Alcohol is transported back to the liver for metabolism and elimination. In addition to further metabolism by ADH in the liver, alcohol is also metabolized by CYP450 enzymes, mainly CYP2E1.7-9
Alcohol is a substrate of CYP2E1, and depending on the frequency of alcohol intake, it can also be either an inducer or inhibitor of CYP2E1. Chronic, heavy alcohol consumption (³5 drinks/occasion) induces the activity of CYP2E1, while short-term heavy consumption inhibits CYP2E1’s activity by competing with other substrates.8
Alcohol intake can alter the pharmacokinetics of medications, including their absorption and metabolism. Conversely, alcohol pharmacokinetics can also be altered by medications. Examples of pharmacodynamic interactions involving alcohol and medications are an increased risk of adverse drug events or an increased susceptibility to the medications’ effects. Furthermore, alcohol intake can play a negative role in certain disease states such as diabetes mellitus.8,9
Methods
This review was conducted through a search of primary and tertiary literature to identify interactions of alcohol with medications in three therapeutic classes (antibiotics, cardiovasculars, and analgesics). Published studies were identified by searching PubMed for various keywords or headings including alcohol, ethanol, drug-alcohol interactions, alcohol and warfarin, and alcohol and hypertension. For the three drug classes, an overview of the available literature is presented and the clinical significance is discussed. The review is also summarized in TABLE 1. The information presented is intended to complement pharmacists’ clinical judgment when providing individualized patient care.
It is important to note that reliable studies on the various drug-alcohol interactions are scarce and not always updated. Many studies revealing an interaction between certain medications and alcohol use involve young healthy volunteers, are conducted in vitro, or involve animals. These factors make it difficult to extrapolate results from such studies to patients with certain disease states or to those taking multiple medications. In addition, the type of alcoholic beverage used varies among studies and includes beer, red wine, and others. Different alcoholic beverages have a variety of components that affect drug-metabolizing enzymes in different ways. Furthermore, much of the evidence for drug-alcohol interactions is based on case reports and not clinical trials.
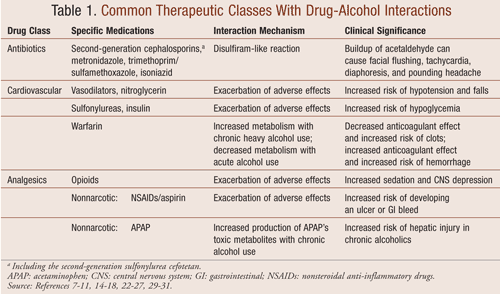
Antibiotics
Concomitant use of antibiotics and alcohol can cause or exacerbate adverse effects. Disulfiram-like reactions have been reported in patients who consume alcoholic preparations while being treated with certain antibiotics, including beta-lactam antibiotics such as the second-generation cephalosporin cefotetan10,11 and the third-generation cephalosporin ceftriaxone, although the interaction with ceftriaxone is extremely rare.12,13 The reaction is mediated by the inhibition of ADH, which in turn irreversibly inhibits the oxidation of acetaldehyde. Elevated concentrations of acetaldehyde cause facial flushing, nausea, vomiting, headache, tachycardia, hypotension, or a combination thereof. The mechanism is thought to involve the methyltetrazolethiol (MTT) side chain commonly found in cephalosporins associated with disulfiram-like reactions. The MTT structure resembles part of the disulfiram molecule.10
Based on several studies and case reports, metronidazole also causes disulfiram-like reactions when coadministered with alcohol. Tillonen et al conducted a study indicating that this reaction might involve ADH inhibition in the gastrointestinal (GI) tract, instead of in the liver as previously thought.14 A disulfiram-like reaction has also been reported with concomitant use of sulfamethoxazole/trimethoprim and alcohol.15
Isoniazid, an antitubercular drug, has been shown to be metabolized more quickly in chronic heavy alcohol users, which can lead to decreased drug effectiveness.16 In addition, the alcohol-isoniazid combination has been associated with an increased risk of isoniazid-associated hepatotoxicity as well as risk of disulfiram-like reactions.16,17
Therefore, patients should always be cautioned to avoid alcohol consumption during, and for several days after, antibiotic regimens known to interact with alcohol. Patients should be informed about unsuspected sources of alcohol and be advised to talk to their physicians regarding any alcohol use.
Cardiovascular Medications
Antihypertensives: Alcohol consumption interacts with some medications taken for hypertension and angina such as nitrates, hydralazine, and nitroglycerin. Concomitant use of alcohol with these medications may lead to enhanced orthostatic hypotension, putting patients at risk for injury or falls.8,9,18 Chronic alcohol use can lead to increased metabolism and decreased effectiveness of the beta-adrenergic blocking agent propranolol, and consumption of verapamil and alcohol has been shown to delay elimination of alcohol, which may lead to prolonged intoxication.7,8,18 Additionally, there is a positive association between alcohol consumption and blood pressure, and alcohol intake may aggravate hypertension or heart failure.8,9,19,20 Patients with cardiovascular conditions should consult their health care providers regarding alcohol use and potential drug interactions.
Antidiabetics: The use of alcohol in patients with diabetes presents several health issues. Alcohol suppresses gluconeogenesis, which may increase risk for hypoglycemia in diabetic patients. The risk is further increased if the patient uses insulin or oral hypoglycemics.8,9 In addition, heavy alcohol use by diabetics may increase risk for diabetic complications including diabetic neuropathy and retinopathy.8,21 For patients on sulfonylureas, literature suggests an increased risk of hypoglycemia, especially with the longer-acting first generation sulfonylureas (tolbutamide and chlorpropamide).7,8,18 There may also be increased risk in second-generation sulfonylureas.22,23 Burge et al examined the effect of alcohol consumption in fasting elderly diabetic patients on glyburide therapy. They reported that small amounts of alcohol consumption (1-2 beverages) may increase patients’ risk of hypoglycemia.22 Hartling et al reported no significant difference in blood glucose levels in healthy subjects receiving glipizide alone or in combination with alcohol. However, there was a delay in blood glucose levels returning to normal when glipizide was taken with alcohol, suggesting a prolonged hypoglycemic effect in the presence of alcohol.23
Disulfiram-like reactions have been reported with concomitant use of alcohol and chlorpropamide. However, it is unclear whether concomitant use of alcohol and either glipizide or glyburide produces the same reaction.18 Diabetic patients on metformin are at an increased risk for lactic acidosis if they consume alcohol heavily.9
The American Diabetes Association (ADA) recommends that patients with diabetes inform their health care providers about alcohol consumption. In addition, the ADA recommends avoiding alcohol on an empty stomach and limiting daily alcohol consumption to one standard drink for women and two standard drinks for men.8,21
Anticoagulants: Alcohol interacts with warfarin, and the consequences of this interaction can lead to significant patient harm. Depending on the frequency of alcohol consumption, alcohol can lead to an increase or decrease in the anticoagulant effect of warfarin. When alcohol is chronically consumed by patients, there is increased metabolism of warfarin, decreasing the drug’s effect and increasing the risk of clot formation. On the other hand, acute alcohol ingestion appears to decrease warfarin’s metabolism, which can greatly increase the risk for a hemorrhage.8,9 The exact mechanism is not well elucidated in the literature and may involve impaired hepatic metabolism as well as other patient-specific factors. Due to the unpredictable effects of alcohol’s interaction with warfarin, patients should be advised to inform their health care providers of any alcohol intake so that the international normalized ratio (INR) may be monitored and warfarin’s dose adjusted as necessary.24,25
Analgesics (Nonnarcotic)
Nonsteroidal Anti-inflammatory Drugs (NSAIDs) and Aspirin: Both NSAIDs and high-dose aspirin are associated with an increased risk of GI bleeding.8,26,27 Epidemiologic data suggest a three- to fivefold increase in the risk of developing a major GI bleed or an ulcer when NSAIDs are used.27 Alcohol consumption can irritate the GI mucosa and increase gastric acid secretions, which may lead to gastritis. Concomitant use of NSAIDs or aspirin and alcohol can further increase risk of a major GI bleed; however, no randomized controlled studies have been conducted to verify such interaction.9
Most evidence for this interaction is based on case-control studies. A large case-control study conducted in the U.S. and Sweden indicated a strong positive association between NSAID or aspirin and alcohol use in increasing risk for an upper GI bleed. The highest incidence of upper GI bleed was in patients who were heavy drinkers (³3 drinks/day) and used aspirin (>325 mg/day) and/or ibuprofen.26 However, this study did not provide enough information on other NSAIDs, including celecoxib. Nevertheless, the package insert for celecoxib states an increased risk of developing an ulcer or GI bleed when NSAIDs are used with alcohol.28
Acetaminophen: This drug is one of the most frequently used analgesics in the U.S. In addition to its availability as an OTC product, acetaminophen is also found in combination with many narcotic medications and is among the most commonly prescribed analgesics.29 In acute overdoses, acetaminophen is associated with hepatotoxicity. There have been numerous case reports suggesting severe liver injury when therapeutic doses of acetaminophen (<4 g/day) are taken in conjunction with chronic alcohol use. However, few, if any, well-designed clinical studies verify the validity of this interaction.7,29 To understand the possible interaction of acetaminophen with alcohol use, a brief explanation of the complex metabolism of acetaminophen is essential.
Acetaminophen is metabolized primarily through glucuronidation or sulfation (90%-96%) and also through CYP2E1 (4%-10%), the same enzyme involved in alcohol metabolism. When acetaminophen is metabolized through CYP2E1, a hepatotoxic metabolite, N-acetyl-para-
The possible interaction of alcohol with acetaminophen use depends on the manner in which alcohol is consumed. In chronic alcohol users, the activity of CYP2E1 is induced, theoretically leading to increased NAPQI production. In addition, published studies suggest that decreased levels of glutathione in alcoholics further increase risk for liver failure.7 Although there is an ongoing controversy in the literature concerning use of acetaminophen in alcoholics, based on current studies, there is no increased risk of hepatotoxicity in alcoholic patients taking therapeutic doses of acetaminophen (<4 g/day) in the absence of other risk factors.29,30
The FDA recommends that patients who drink more than three alcoholic drinks per day should consult their physician prior to any OTC pain reliever use.30 In addition, there is tremendous variability in how patients consume alcohol and in their medication compliance, so patients should always be warned about the increased risk of liver injury when acetaminophen-containing products are taken with alcohol.
Analgesics (Narcotic)
Opioid medications such as methadone and codeine derivatives are well known for central nervous system (CNS) effects, including sedation and respiratory depression.18 The mechanism of interaction between opioid analgesics and alcohol is pharmacodynamic in nature. Alcohol increases the risk of adverse effects and CNS depression.8,18
Few studies are available illustrating this interaction, and warnings regarding concomitant use of alcohol and opioids are based on package inserts of the various opioid medications, including codeine and its derivatives, morphine, fentantyl, and methadone.18 However, after the hydromorphone extended-release capsule Palladone was removed from the market due to a possible lethal interaction with alcohol, it became evident that there is a need for more investigation into alcohol’s interaction with opioids, especially extended-release formulations.7,31 The FDA requested the removal of Palladone in 2005 after a pharmacokinetic study revealed a “dose-dumping” effect when the drug and alcohol were used concomitantly. The average peak hydromorphone concentration rose two- to sixfold when it was administered with alcoholic beverages.7,32 Thus, patients should inform their health care providers about any alcohol use prior to starting analgesic therapy.
Conclusion
This review summarizes drug-alcohol interactions among three major therapeutic drug classes (antibiotics, cardiovascular medications, and analgesics). Pharmacists play a key role in educating patients about medications, their adverse effects, and possible interactions. Pharmacists should also counsel patients regarding any drug-alcohol interactions. Although this review is meant to be an overview of alcohol’s interactions with common therapeutic classes, it is not all-inclusive. This review may be used to complement pharmacists’ clinical judgment when providing individualized patient care.
REFERENCES
1. Centers for Disease Control and Prevention. Alcohol use. www.cdc.gov/nchs/fastats/
2. IMS National Prescription Audit PLUS. 2008 Top therapeutic classes by U.S. dispensed prescriptions. IMS Health. www.imshealth.com/
3. Onder G, Pedone C, Landi F, et al. Adverse drug reactions as cause of hospital admissions: results from the Italian Group of Pharmacoepidemiology in the Elderly (GIFA). J Am Geriatr Soc. 2002;50:1962-1968.
4. Brown RL, Dimond AR, Hulisz D, et al. Pharmacoepidemiology of potential alcohol-prescription drug interactions among primary care patients with alcohol-use disorders. J Am Pharm Assoc. 2007;47:135-139.
5. Ferreira MP, Weems MK. Alcohol consumption by aging adults in the United States: health benefits and detriments. J Am Diet Assoc. 2008;108:1668-1676.
6. National Institute on Alcohol Abuse and Alcoholism. Percent distribution of days of alcohol use in past month by demographic characteristics: NSDUH (NHSDA), 1994–2002. www.niaaa.nih.gov/Resources/
7. Jang GR, Harris RZ. Drug interactions involving ethanol and alcoholic beverages. Expert Opin Drug Metab Toxicol. 2007;3:719-731.
8. Moore AA, Whiteman EJ, Ward KT. Risks of combined alcohol/medication use in older adults. Am J Geriatr Pharmacother. 2007;5:64-74.
9. Fraser AG. Pharmacokinetic interactions between alcohol and other drugs. Clin Pharmacokine.
10. Kline SS, Mauro VF, Forney RB, et al. Cefotetan-induced disulfiram-type reactions and hypoprothrombinemia. Antimicrob Agents Chemother. 1987;31:1328-1331.
11. Norrby SR. Adverse reactions and interactions with newer cephalosporin and cephamycin antibiotics. Med Toxicol. 1986;1:32-46.
12. Eron LJ, Park CH, Hixon DL, et al. Ceftriaxone therapy of bone and soft tissue infections in hospital and outpatient settings. Antimicrob Agents Chemother. 1983;23:731-737.
13. Billstein SA, Sudol TE. Disulfiram-like reactions rare with ceftriaxone. Geriatrics. 1992;47:70.
14. Tillonen J, Vakevainen S, Salaspuro V, et al. Metronidazole increases intracolonic but not peripheral blood acetaldehyde in chronic ethanol-treated rats. Alcohol Clin Exp Res. 2000;24:570-575.
15. Heelon MW, White M. Disulfiram-cotrimoxazole reaction. Pharmacotherapy. 1998;869-870.
16. Baciewicz AM, Self TH. Isoniazid interactions. South Med J. 1985;78:714-718.
17. Isoniazid package insert. Eatontown, NJ: West-Ward Pharmaceutical Corp; March 2008.
18. Drug interactions. Thomson Micromedex. Greenwood Village, CO. www.thomsonhc.com. Accessed October 9, 2010.
19. Zilkens RR, Burke V, Hodgson JM, et al. Red wine and beer elevate blood pressure in normotensive men. Hypertension. 2005;45:874-879.
20. Bau PF, Bau CH, Naujorks AA, Rosito GA. Early and late effects of alcohol ingestion on blood pressure and endothelial function. Alcohol. 2005;37:53-58.
21. Alcohol. American Diabetes Association. www.diabetes.org/type-1- 1997;33:79-90.
22. Burge MR, Zeise TM, Sobhy TA, et al. Low-dose ethanol predisposes elderly fasted patients with type 2 diabetes to sulfonylurea-induced low blood glucose. Diabetes Care. 1999;22:2037-2043.
23. Hartling SG, Faber OK, Wegmann ML, et al. Interaction of ethanol and glipizide in humans. Diabetes Care. 1987;10:683-686.
24. Hylek EM, Heiman H, Skates SJ, et al. Acetaminophen and other risk factors for excessive warfarin anticoagulation. JAMA. 1998;279:657-662.
25. Havrda DE, Mai T, Chonlahan J. Enhanced antithrombotic effect of warfarin associated with low-dose alcohol consumption. Pharmacotherapy. 2005;25:303-307.
26. Kaufman DW, Kelly JP, Wiholm BE, et al. The risk of acute major upper gastrointestinal bleeding among users of aspirin and ibuprofen at various levels of alcohol consumption. Am J Gastroenterol.
27. Pfau PR, Lichenstein GR. NSAIDS and alcohol: never the twain shall mix? Am J Gastroenterol.
28. Celebrex (celecoxib) package insert. New York, NY: Pfizer, Inc; June 2009.
29. Kuffner EK, Green JL, Bogdan GM, et al. The effect of acetaminophen (four grams a day for three consecutive days) on hepatic tests in alcoholic patients—a multicenter randomized study. BMC Med.
30. FDA proposes alcohol warning for all OTC pain relievers. U.S. Department of Health and Human Services. November 14, 1997. http://archive.hhs.gov/news/ 1999;94:3189-3196. 1999;94:3098-3101. 2007;5:13.
31. Walden M, Nicholls FA, Smith KJ, Tucker GT. The effect of ethanol on the release of opioids from oral prolonged-release preparations. Drug Dev Ind Pharm. 2007;33:1101-1111.
32. Information for healthcare professionals: hydromorphone hydrochloride extended-release capsules (Palladone). FDA alert. July 2005. www.fda.gov/Drugs/DrugSafety/
To comment on this article, contact rdavidson@uspharmacist.com.






