US Pharm. 2014;39(3):39-43.
ABSTRACT: Pain is often challenging to manage in the geriatric population due to age-related changes that make drug selection difficult. In addition, geriatric patients often have comorbidities that can affect drug choice and lead to polypharmacy. Polypharmacy and age-related changes increase the risk of drug intolerance and drug interactions. The presence of dementia further complicates pain management because of difficulties with pain assessment and the increased risk of cognitive adverse events with commonly prescribed medications. Uncontrolled pain in this population can also contribute to behavioral issues that can be challenging for caregivers.
More than 36 million people worldwide are believed to suffer from dementia.1 The United States has the second largest population of people with dementia, about 4 million.1 A majority of cases of dementia occur after the age of 65 years.1 With rising growth rates in the elderly population, it is likely that the cases of dementia will increase accordingly.2
The prevalence of pain in U.S. adults 65 years and over is variable. There are reports suggesting the rates of pain in geriatric populations are around 21% (4 million people); most of this pain is classified as persistent.2,3 Other studies have shown that over 90% of older adults experience pain, predominantly musculoskeletal.4 The prevalence of pain in dementia varies, largely based upon the assessment tools utilized in the studies.4,5
Assessment of Pain
As dementia progresses, the ability to communicate and remember pain becomes increasingly difficult. These challenges cause problems with the proper assessment of pain in this population. It is widely acknowledged that pain is often undertreated in patients with dementia, especially as dementia progresses. Often pain will be displayed in the form of behavioral issues such as agitation, combativeness, and wandering (TABLE 1).4,6
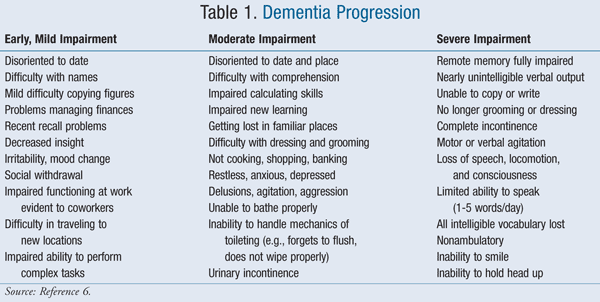
The current standard of pain assessment is patient self-report.5,7,8 Multiple pain scales exist that utilize the self-report such as the visual analog scales, Faces Pain Scale, and Numeric Rating Scale.4,5,9 Due to their simplicity, these scales are deemed appropriate for use in patients with mild-to-moderate dementia.7,9 Assessment becomes increasingly troublesome in advanced dementia. In some studies, self-report assessments have shown success. However, observation scales are more appropriate in patients who are not able to communicate, including the Pain Assessment Checklist for Seniors with Limited Ability to Communicate (PACS-LAC), Observational Pain Behavior Tool, Rating Pain in Dementia (RaPID), and Pain Assessment in Advanced Dementia (PAINAD).4,9 These scales are routinely recommended to be performed in concert with an attempt at self-report.4,8,9
Medication Management Principles
Pharmacokinetic and pharmacodynamic changes due to aging play a large role in the ability of this population to tolerate pain medication. With increasing age comes decline in renal and hepatic function. Reduced hepatic metabolism can lead to prolonged drug half-life, thus increasing retention in the blood stream. For drugs that are extensively metabolized via first-pass metabolism, there is a possibility for increased bioavailability. Because of decreased muscle mass and declining renal function, measures of serum creatinine are often underestimated in the elderly, sometimes leading to overestimated creatinine clearance values. Additionally, the half-life of renally eliminated drugs is often increased due to reduced renal blood flow, which can ultimately result in drug accumulation.7,10,11
Furthermore, volume of distribution is altered in patients with increases in body fat and decreases in total body water and lean muscle mass, which are commonly seen in older adults. Increases in volume of distribution can delay a drug’s onset of action and elimination rate, while reductions can cause increases in peak concentrations. Drugs that are highly protein-bound can also be affected by albumin levels, which can become low in elders with acute illnesses or those who are frail. Gastrointestinal (GI) absorption or function also changes during the normal aging process and can affect drug absorption in some patients. Finally, anticholinergic effects (e.g., confusion, constipation) are more prominent in the elderly.7,10,11 TABLE 2 outlines the impact of these changes in relation to selected analgesics.11,12
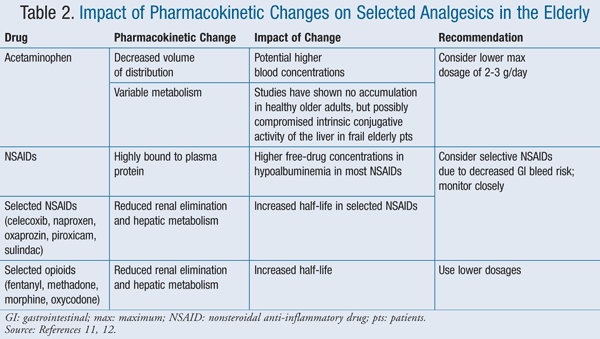
The elderly typically have higher rates of polypharmacy due to multiple comorbid conditions, which only increases the likelihood that this patient population will present with adverse reactions to these medications.10
Drug Selection Considerations
Considerations for drug selection should include type of pain, timing of pain (TABLE 3),7,9,13 and route of administration.7
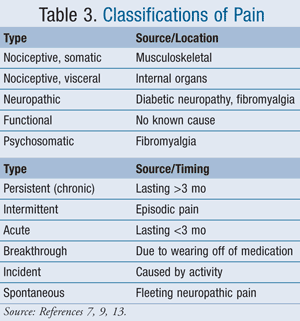
Timing: Extended-release products can be utilized in patients with higher short-acting medication demand to potentially improve pain control and reduce pill burden. These patients should have a short-acting product available for breakthrough pain. However, because they may not be able to ask for them, patients with dementia should not regularly be given pain medications on an as-needed basis. If as-needed pain control is used, frequent pain assessment should be performed. Finally, intensity of incident pain can be reduced with prior scheduled pain medication (i.e., giving pain medication 30 minutes preceding physical therapy).7
Route of Administration: It is important to consider dosage forms when making recommendations for patients with dementia. For those who have behavioral issues that make medication management difficult (e.g., agitation, refusing to take medications), consideration should be given to transdermal dosage forms placed in locations from which they cannot easily be removed. For patients with difficulty swallowing, mucosal, rectal, and transdermal dosage forms are available.7
Pharmacotherapy
Acute pain should be managed with short-acting, rapid-onset analgesics. For pain that is persistent, consideration should be given to initial treatment with nonopioid analgesics. Opioid analgesics should be used for pain nonresponsive to nonopioid agents. Adjuvant analgesics are recommended for neuropathic pain. The Beers criteria and the Consensus Guidelines for Oral Dosing of Primarily Renally Cleared Medications in Older Adults are references that can be used to assist in determining appropriate drug selection and dosing of medications in older adults.14,15
Nonopioid Analgesics: Acetaminophen is used in this population as initial treatment because of its safety profile and efficacy.7 It is important to note that there have been many changes in the last few years regarding dosing for acetaminophen. In 2011, McNeil Consumer Healthcare (Tylenol’s manufacturer) reduced the maximum daily dose of extra-strength acetaminophen to 3,000 mg when used for self-care in response to the FDA recommendation regarding the sole utilization of the 325-mg dose in combination products.16,17 A maximum daily dose of 4,000 mg is still allowable for regular-strength OTC acetaminophen products or use of acetaminophen under the care of a healthcare provider; however, due to previously mentioned pharmacokinetic changes in the elderly, a maximum dose of 2,000 to 3,000 mg is recommended.12,18
Nonsteroidal anti-inflammatory drugs (NSAIDs) should be used with extreme caution because of a higher association with GI bleeding in the geriatric population.7,14 NSAIDs are most commonly used when inflammation is present, for which steroids may be alternatively used with caution. If NSAIDs are used, adjunctive therapy for GI protection with a proton pump inhibitor (PPI) is warranted. The use of topical NSAIDs at approved doses appears to be safe for periods of <4 weeks because of seemingly low systemic absorption. It is important to keep in mind that NSAIDs may also potentiate heart failure, hypertension, and renal dysfunction.7
Opioid Analgesics: Treatment with opioids has shown efficacy in many types of pain, but these medications are not without adverse effects.7 These medications require monitoring and typically treatment for associated complications. The majority of these agents are considered appropriate in elderly patients, with the exception of meperidine, which should be avoided due to high rates of neurotoxicity in patients with renal dysfunction (TABLE 4).7,14

Adverse effects include constipation, sedation, cognitive impairment, delirium, and respiratory depression. Constipation is one of the more easily preventable adverse events associated with opioid use. Upon initiation of an opioid pain regimen, patients can be scheduled on an osmotic or stimulant laxative plus a stool softener such as docusate. Patients should also be counseled on maintaining adequate hydration. Because opioids decrease motility, secretions, and blood flow in the GI tract, docusate alone is unlikely to be effective. In addition, constipation does not lessen during treatment; thus, duration of laxative treatment should parallel that of opioid use.7,13,19 If constipation is severe, consideration can be given to the use of transdermal fentanyl or buprenorphrine.11,19
While the amount of sedation may decrease with prolonged opioid use, cognitive impairment is seen typically at drug initiation or upward titration.19 It is reasonable to evaluate baseline cognitive assessment prior to the initiation or upward titration of opioids in patients with dementia. Care should be taken to limit or avoid concurrent use of medications with anticholinergic or sedating side effects to prevent worsening of sedation, cognitive function, or delirium.
While the risk of respiratory depression increases with age, dose, and the presence of underlying pulmonary conditions, tolerance does develop with continued use. Options for prevention include low initial dosing with conservative titrations.11,19
Adjuvant Analgesics: Adjuvant analgesics are strongly recommended for use in neuropathic pain, but can be used for other types of pain as well. They are sometimes combined with other analgesics for increased pain control. The term adjuvant drugs is used to group the antidepressant and anticonvulsant drug classes in pain management.7 While tricyclic antidepressants (TCAs) have shown efficacy in neuropathic pain reduction, use of these agents in the elderly is not recommended due to their strong anticholinergic properties and incidence of sedation and orthostasis.7,14
Serotonin-norepinephrine reuptake inhibitors (SNRIs; venlafaxine, duloxetine, milnacipran) are preferred above the TCAs for neuropathic pain control in the elderly because of milder adverse-event profiles. Their efficacy is thought to be related to the norepinephrine reuptake inhibition. It is important to note that venlafaxine only exhibits norepinephrine reuptake at higher doses. Doses of 150 mg per day or greater have shown efficacy for neuropathic pain.20,21
The anticonvulsants gabapentin and pregabalin are also used commonly for neuropathic pain control. Gabapentin can cause sedation that lessens with drug use; thus, it should be started at a low dose, such as 300 mg in the evening, and titrated slowly to effect.4,7 It can take several weeks to show improvement in pain control with gabapentin and around 1 week with pregabalin.7,22
Other Drugs: Other medications that can be considered for treatment of pain in elderly demented patients include corticosteroids, calcitonin, bisphosphonates, and topical analgesics.7 Those with inflammatory disorders may experience some additional relief with corticosteroids. For patients with bone-related pain, there is some evidence that calcitonin may lessen pain in compression and pelvic fractures. Pain associated with multiple myeloma and breast or prostate cancer may be reduced with the use of bisphosphonates. When considering topical analgesics, it is important to recognize that the majority of supportive evidence for the lidocaine patch is in neuropathic pain, although it appears to be less efficacious than the adjuvant analgesics. In addition, while effective for neuropathic and nonneuropathic pain, topical capsaicin is not well tolerated because of the burning sensation associated with its use.7
There are other drugs that have been used to treat pain that may not be appropriate for use in the elderly. Muscle relaxants can increase the risk of anticholinergic-related adverse events as well as sedation and should be avoided in the geriatric population. Benzodiazepines should also be avoided due to their limited efficacy in pain management and the increased risk of cognitive impairment, delirium, and falls in older adults and those with dementia.7,14 Cannabinoids are not used often in geriatric patients because of the associated dysphoria.7
Nonpharmacologic Therapy
There is limited evidence available for nonpharmacologic options for pain management in older adults. Therapies with supportive evidence include exercise, music therapy, and acupuncture. Other therapies that are commonly used include heat and ice therapy, transcutaneous electric nerve stimulation, and cognitive-behavioral therapy.23
Conclusion
The management of pain in patients with dementia can be complicated due to comorbidities, polypharmacy, age-related pharmacodynamic and pharmacokinetic changes that affect drug choice, and difficulty in pain assessment, especially in advanced dementia. Even so, there are many agents available for use in this population. The regimen of each patient should be individually tailored. Medications should be started at the lowest dosage and slowly titrated. Renal function should be regularly assessed with medication change as necessary. Examination of compliance, monitoring for adverse events, and prevention of drug interactions should occur at every opportunity, as should determination of the patient’s ability to report pain and request as-needed medication.7,10 Regular assessment of a pain regimen can help to ensure better pain control in elderly patients with dementia.
REFERENCES
1. World Health Organization (WHO). Dementia: A Public Health Priority.
Geneva, Switzerland: WHO; 2012.
http://apps.who.int/iris/bitstream/10665/75263/1/9789241564458_eng.pdf.
Accessed November 23, 2013.
2. Werner CA. 2010 Census Brief C2010BR-09: The Older Population: 2010. Suitland, MD: U.S. Census Bureau; November 2011. www.census.gov/prod/cen2010/briefs/c2010br-09.pdf. Accessed September 6, 2013.
3. National Center for Health Statistics (NCHS). Health, United States, 2006 With Chartbook on Trends in the Health of Americans. Hyattsville, MD: NCHS; 2006. www.cdc.gov/nchs/data/hus/hus06.pdf. Accessed November 23, 2013.
4. Scherder E, Plooij B. Assessment and management of
pain, with particular emphasis on central neuropathic pain, in moderate
to severe dementia. Drugs Aging. 2012;29:701-706.
5. McAuliffe L, Brown D, Fetherstonhaugh D. Pain and dementia: an overview of the literature. Int J Older People Nurs. 2012;7:219-226.
6. A guide to dementia diagnosis and treatment. American
Geriatrics Society. http://dementia.americangeriatrics.org. Accessed
November 19, 2013.
7. AGS Panel on Pharmacological Management of Persistent
Pain in Older Persons. Pharmacological management of persistent pain in
older persons. JAGS. 2009;57:1331-1346.
8. Herr K, Coyne PJ, McCaffery M, et al. Pain assessment
in the patient unable to self-report: position statement with clinical
practice recommendations. Pain Manag Nurs. 2011;12:230-250.
9. Achterberg WP, Peiper M, van Dalen-Kok AH, et al. Pain management in patients with dementia. Clin Interv Aging. 2013;8:1471-1482.
10. Petrovic M, van der Cammen T, Onder G. Adverse drug reactions in older people. Drugs Aging. 2012;29:453-462.
11. Pergolizzi J, Böger RH, Budd K, et al. Opioids and the
management of chronic severe pain in the elderly: consensus statement
of an International Expert Panel with focus on the six clinically most
often used World Health Organization step III opioids (buprenorphine,
fentanyl, hydromorphone, methadone, morphine, oxycodone). Pain Pract. 2008;8:287-313.
12. O’Neil C, Hanlon J, Marcum Z. Adverse effects of
analgesics commonly used by older adults with osteoarthritis: focus on
non-opioid and opioid analgesics. Am J Geriatr Pharmacother. 2012;10:331-342.
13. Reuben DB, Herr KA, Pacala JT, et al. Geriatrics At Your Fingertips: 2011. 13th ed. New York, NY: The American Geriatrics Society; 2011.
14. The American Geriatrics Society 2012 Beers Criteria
Update Expert Panel. American Geriatrics Society updated Beers Criteria
for potentially inappropriate medication use in older adults. J Am Geriatr Soc. 2012;16:616-631.
15. Hanlon JT, Aspinall SL, Selma TP, et al. Consensus
guidelines for oral dosing of primarily renally cleared medications in
older adults. J Am Geriatr Soc. 2009;57:335-340.
16. FDA drug safety communication: prescription
acetaminophen products to be limited to 325 mg per dosage unit; boxed
warning will highlight potential for severe liver failure. January 13,
2011. www.fda.gov/Drugs/DrugSafety/ucm239821.htm. Accessed December 11,
2013.
17. Kuffner EK. Tylenol (acetaminophen) for healthcare
professionals [letter]. McNeil Consumer Healthcare Division of
McNeil-PPC, Inc. July 28, 2011.
www.tylenolprofessional.com/tylenol-dosage-letter-to-hcps.html. Accessed
December 11, 2013.
18. Shega JW, Morrissey MB, Reid MC. From Publication
to Practice: An Interdisciplinary Look at the Labeling Changes for
Acetaminophen and the Implications for Patient Care. Washington, DC:
Gerontological Society of America; 2011.
www.tylenolprofessional.com/assets/v4/gsa-report-acetaminophen.pdf.
Accessed December 11, 2013.
19. Martin CM, Forrester CS. Anticipating and managing opioid side effects in the elderly. Consult Pharm. 2013;28:150-159.
20. Redrobe JP, Bourin M, Colombel MC, et al.
Dose-dependent noradrenergic and serotonergic properties of venlafaxine
in animal models indicative of antidepressant activity. Psychopharmacology. 1998;138:1-8.
21. Dworkin RH, O’Connor AB, Backonja M, et al. Pharmacologic management of neuropathic pain: evidence-based recommendations. Pain. 2007;132:237-251.
22. Freeman R, Durso-DeCruz, Emir B. Efficacy, safety, and
tolerability of pregabalin treatment for painful diabetic peripheral
neuropathy. Diabetes Care. 2008;31:1448-1454.
23. Park J, Hughes AK. Nonpharmacological approaches to
the management of chronic pain in community-dwelling older adults: a
review of empirical evidence. JAGS. 2012;60:555-568.
To comment on this article, contact rdavidson@uspharmacist.com.





