US Pharm. 2010;35(5):HS8-HS15.
An estimated 4 million people suffer from neuropathic pain in the United States.1 Neuropathic pain, as defined by the International Association for the Study of Pain, is “pain initiated or caused by a primary lesion or dysfunction in the nervous system.”2 Dysfunction of the nervous system can lead to numbness, weakness, loss of deep tendon reflexes, and spontaneous and stimulus-evoked pain.3 Pain is most often described as burning, shooting, stabbing, or shocklike, and many patients experience allodynia and hyperalgesia. Neuropathic pain also affects patients’ mood, quality of life, activities of daily living, and work performance.3 Underlying causes of neuropathic pain include infection, trauma, inflammation, cancer, metabolic abnormalities, neurotoxins, and nerve compression.4 Neuropathic pain is further classified as diabetic neuropathy, postherpetic neuralgia, trigeminal neuralgia, cancer-related neuropathy, and phantom limb pain, to name a few. This review will focus on diabetic neuropathy.
Diabetic neuropathy, a microvascular complication of diabetes, is one of the most common forms of neuropathic pain. It develops in 28% to 55% of patients with diabetes mellitus.5 Patients with type 1 or type 2 diabetes with prolonged hyperglycemia are at greatest risk of developing diabetic neuropathy. In addition to hyperglycemia, patients with existing comorbidities such as dyslipidemia, hypertension, and cardiovascular disease are also at increased risk.6
Pathophysiology
Alterations in peripheral nerves, the brain, or the spinal cord lead to changes that result in pain transmission. Many of these changes include upregulation or downregulation of neuropeptides and neurotransmitters.7 Following damage to the nervous system, transcription and axonal trafficking of sodium channels are increased, and potassium channels are decreased at the site of injury.3 This leads to hyperexcitable neurons and generation of ectopic activity, and potentially to the generation of sudden and spontaneous sensations of pain. Voltage-gated calcium channels also increase in number following damage to the nerves.3 Calcium entry leads to the release of substance P and glutamate from injured peripheral nerves.3 Increased expression of the alpha-2-delta subunit of voltage-gated calcium channels within the dorsal root ganglion leads to allodynia.3 Peripheral nerve injury results in many other changes that could explain neuropathic pain. It results in hypertrophy and activation of glial cells and microglia within the gray matter of the spinal cord.3 Upon activation by adenosine triphosphate (ATP), microglia release interleukin-1 (IL-1), tumor necrosis factor-alpha (TNF-alpha) and neurotrophins, including brain-derived neurotrophic factor, which exacerbate nociceptive transmission and contribute to the maintenance of neuropathic pain.3
Clinical Symptoms
Focal and diffuse neuropathies are the two major categories.5 While focal neuropathies such as Bell’s palsy have an increased incidence in patients with diabetes, diffuse neuropathy is the most common. Diabetic neuropathy is the most common distal form of neuropathy. Uncontrolled diabetes leads to progressive demyelination, resulting in hyperesthesia and allodynia, and burning, stabbing, or shooting pains. These symptoms are often worse at night, which in turn can lead to loss of sleep. This disorder affects sensory function and is predominately distal and symmetric.5 While up to 32% of diabetic patients with peripheral neuropathy experience painful symptoms, many remain asymptomatic.8 Typically, small fiber neuropathy develops with burning feet prior to large fiber neuropathy, which results in numbness, weakness, or gait disturbances. As one moves from the toes proximally, a reduction in pinprick sensation is the most identifiable sign of neuropathy. A reduction in temperature, sensation, light touch, proprioception, and vibration may also be present.5 Loss of sensation predisposes patients to foot ulcers, which could lead to serious infections.
Treatment
Currently, there are no treatment options that have been shown to reverse nerve injury. However, tight glycemic control slows progression of diabetic neuropathy and reduces the development of symptoms.5 The available treatments of diabetic neuropathy are palliative, with the goal of reducing pain.8 Antidepressants, anticonvulsants, opioids, and topical agents such as lidocaine and capsaicin are used in the treatment of neuropathic pain (TABLE 1). Currently, duloxetine and pregabalin are the only agents that have FDA approval for the treatment of painful diabetic neuropathy.4
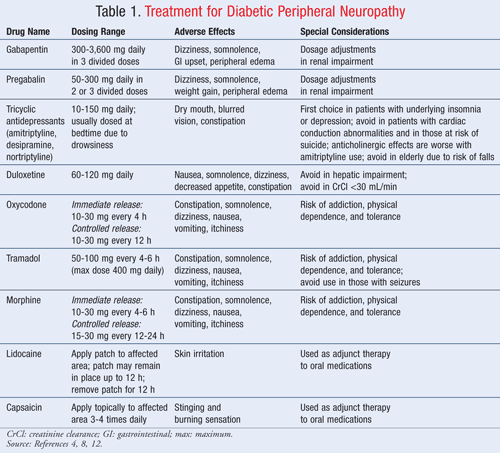
Anticonvulsants
Gabapentin, an alpha-2-delta subunit voltage-gated calcium channel antagonist, lessens depolarization-induced calcium influx at nerve terminals.9,10 This leads to decreased release of excitatory neurotransmitters. Intra- and intersubject variability in the absorption of gabapentin may occur because gabapentin is absorbed from the gastrointestinal (GI) tract through a saturable amino acid active transport system.11 In addition, as the dose of gabapentin increases, bioavailability decreases.11 Gabapentin is eliminated renally as an unchanged drug and does not inhibit or induce CYP450 isoenzymes. It is typically well tolerated, with the most common adverse effects being dizziness, somnolence, and peripheral edema.12
A meta-analysis by Finnerup et al reviewed randomized, placebo-controlled, double-blind studies of treatments for neuropathic pain that were published in peer-reviewed journals through April 2005.13 One hundred and five studies met the inclusion and exclusion criteria, 39 of which looked at anticonvulsants in neuropathic pain. The principal effect measure was number needed to treat (NNT). NNT was defined as the number of patients that need to be treated with a certain drug in order for one patient to achieve a reduction in pain by 50%. The number needed to harm (NNH) was also computed and was defined as the number of patients that need to be treated for one patient to drop out due to adverse effects. For gaba-pentin, the overall NNT in neuropathic pain, including high as well as low doses, was calculated to be 5.1.13 Excluding one study that used only 900 mg/day of gabapentin and including only the high dose of 2,400 mg in a study that looked at 1,800 to 2,400 mg of gabapentin, a combined NNT of 3.8 was calculated.13 The NNH for gabapentin was 26.1.13
A Cochrane systematic review conducted through November 2004 calculated the NNT for gabapentin based on type of neuropathic pain.14 Based on four placebo-controlled trials that looked at gabapentin in patients with diabetic neuropathy, the NNT for effectiveness was 2.9.
Pregabalin, the S-enantiomer of 3-aminomethyl-5-methylhexanoic acid, has been termed a successor to gabapentin. Like gabapentin, it is an alpha-2-delta subunit voltage-gated calcium channel antagonist; however, pregabalin is six times more potent than gabapentin because of its stronger binding affinity for the alpha-2-delta subunit.15 Pharmacokinetics of gabapentin and pregabalin differ, as pregabalin shows linear pharmacokinetics across the dose range of 150 to 600 mg/day.11 Pregabalin has an oral bioavailability ≥90% and is rapidly absorbed from the GI tract.11 Pregabalin is eliminated unchanged in the urine and does not inhibit or induce CYP450 isoenzymes. As with gabapentin, dizziness, somnolence, and peripheral edema are the most common reported adverse events.16 Because pregabalin received ratings of “good drug effect” and “high” similar to diazepam in a study of 15 recreational sedative/hypnotic users, pregabalin is classified as a schedule V controlled substance by the Drug Enforcement Administration (DEA).16 Abrupt or rapid discontinuation of pregabalin has been associated with insomnia, nausea, headache, and diarrhea—symptoms that could be associated with physical dependence.16 The aforementioned meta-analysis by Finnerup et al also included studies of pregabalin and calculated an NNT of 4.2 for pregabalin 150 to 600 mg in postherpetic neuralgia and painful diabetic neuropathy.13 The NNH was 11.7.
Antidepressants
Tricyclic antidepressants (TCAs) should be used as first-line therapy in patients with underlying insomnia or depression. However, TCAs should be avoided in patients who are suicidal and in those at risk for cardiac conduction abnormalities. TCAs inhibit norepinephrine and serotonin, which contribute to pain relief. In addition, they inhibit calcium and sodium channels, adenosine receptors, and NMDA receptors, which lead to suppression of neuronal hyperexcitability. Desipramine and nortriptyline exhibit fewer anticholinergic adverse effects such as dry mouth, blurred vision, and constipation.8 The Finnerup meta-analysis calculated an NNT for TCAs of 2 to 3.13 The NNH was 14.7.
Serotonin-norepinephrine reuptake inhibitors (SNRIs), such as duloxetine, have shown to be effective for the treatment of painful diabetic neuropathy.4 Similar to the TCAs, SNRIs produce pain relief through serotonin and norepinephrine reuptake. Adverse effects include nausea, somnolence, dizziness, decreased appetite, and constipation. Duloxetine should be avoided in patients with renal and hepatic impairment.4 A meta-analysis conducted by Sultan and colleagues calculated the NNT for at least 50% pain relief at 12 to 13 weeks with duloxetine for painful diabetic neuropathy.17 The NNT for duloxetine 60 mg was 5.8 and for duloxetine 120 mg was 5.7. The NNH was 15.
Opioids
While opioids are frequently used in the treatment of neuropathic pain, their long-term efficacy remains uncertain. Opioids such as tramadol, oxycodone, and morphine are not considered first-line therapy for patients with diabetic neuropathy. However, these agents have been studied to support their efficacy and safety in the treatment of neuropathic pain. Oxycodone, tramadol, and morphine are opioid agonists at the mu receptors. Common adverse effects include constipation, drowsiness, nausea, headache, and dizziness.18 Physical dependence and withdrawal symptoms may sometimes limit the use of opioids. Opioids should be avoided in patients with a history of drug and alcohol abuse. The Finnerup meta-analysis calculated an NNT of 2.6 for oxycodone, 2.5 for morphine, and 3.9 for tramadol.13 The combined NNH for tramadol was 9. However, it was nonsignificant for oxycodone and morphine.
Topical Agents
Capsaicin and lidocaine are the two main topical agents used for diabetic neuropathy. Capsaicin is an alkaloid derived from chili peppers that works mainly on sensory C fibers to deplete substance P. This causes desensitization to afferent sensory nerves resulting in pain relief.8,18 Adverse effects include burning, stinging, and erythema.8 A disadvantage to capsaicin use is that it must be applied 3 to 4 times daily for up to 8 weeks for optimal pain relief to occur.18 The Finnerup meta-analysis calculated an NNT of 6.7 for capsaicin; however, it is important to note that capsaicin relieved pain in only three out of the five studies. The NNH was 11.5.13
In contrast, lidocaine works by inhibiting the voltage-gated sodium channels in the damaged nerves. The most common adverse effect associated with lidocaine use is skin irritation. While case reports have supported the use of lidocaine patches, no randomized controlled trials have been conducted specifically related to diabetic neuropathy.18
Combination Therapy
An adequate trial of a single medication followed by titration to alleviate pain should be the first step in managing diabetic neuropathy. Managing adverse effects should be a continuous process throughout treatment. Drug tapering or discontinuation may be required if the adverse effects are intolerable. If a single drug therapy is ineffective, utilizing combination therapy may be the next step. This has been shown in the recent article published by Gilron et al.19 The authors concluded that gabapentin in combination with nortriptyline was more efficacious than either drug alone for the treatment of neuropathic pain. This article recommends the use of combination therapy with gabapentin and nortriptyline for patients who show a partial response to either drug alone.19 While further studies are needed to evaluate the use of combination therapy for treatment of neuropathic pain, the use of combination therapy is more widely acceptable by clinicians.
Conclusion
Several factors should be considered when individualizing therapy for neuropathic pain, including analgesia, tolerability, adverse effects, cost-effectiveness, and improvement of sleep, mood, and quality of life.3 Few direct comparator trials exist in the area of neuropathic pain. The European Medicines Agency has published guidelines for neuropathic pain studies in hopes of making future data more comparable.20 When treating neuropathic pain, it is important to remember that treatment must be individualized, as there is not one drug that works for all types of neuropathic pain.3
REFERENCES
1. Dworkin RH, Backonja M, Rowbotham MC, et al. Advances in neuropathic pain: diagnosis, mechanisms, and treatment recommendations. Arch Neurol. 2003;60:1524-1534.
2. IASP pain terminology. Neuropathic pain. International Association for the Study of Pain. www.iasp-pain.org. Accessed April 9, 2010.
3. Gilron I, Watson CP, Cahill CM, Moulin DE. Neuropathic pain: a practical guide for the clinician. CMAJ.
2006;175:265-275.
4. Burzynski J, Strassels S. Persistent pain. In: Linn WD, Wofford MR, O’Keefe ME, Posey LM, eds. Pharmacotherapy in Primary Care. New York, NY: McGraw-Hill Publishing, Inc; 2009:203-217.
5. Rutkove SB, Discussant MD. A 52-year-old woman with disabling peripheral neuropathy: review of diabetic polyneuropathy. JAMA. 2009;302:1451-1458.
6. Tesfaye S, Chaturvedi N, Eaton SE, et al. Vascular risk factors and diabetic neuropathy. N Engl J Med. 2005;352:341-350.
7. Chen H, Lamer TJ, Rho RH, et al. Contemporary management of neuropathic pain for the primary care physician. Mayo Clinic Proc. 2004;79:1533-1545.
8. Zin CS, Nissen LM, Smith MT, et al. An update on the pharmacological management of post-herpetic neuralgia and painful diabetic neuropathy. CNS Drugs. 2008;22:417-442.
9. Cheng JK, Chiou LC. Mechanisms of the antinociceptive action of gabapentin. J Pharmacol Sci. 2006;100:471-486.
10. Rose MA, Kam PC. Gabapentin: pharmacology and its use in pain management. Anaesthesia. 2002;57:451-462.
11. Bialer M, Johannessen SI, Kupferberg HJ, et al. Progress report on new antiepileptic drugs: a summary of the Seventh Eilat Conference (EILAT VII). Epilepsy Res. 2004;61:1-48.
12. Neurontin (gabapentin) package insert. New York, NY: Parke-Davis Division of Pfizer; January 2007.
13. Finnerup NB, Otto M, McQuay HJ, et al. Algorithm for neuropathic pain treatment: an evidence based proposal. Pain. 2005;118:289-305.
14. Wiffen PJ, McQuay HJ, Edwards JE, Moore RA. Gabapentin for acute and chronic pain. Cochrane Database Syst Rev. 2005;(3):CD005452.
15. Sabatowski R, Galvez R, Cherry D, et al. Pregabalin reduces pain and improves sleep and mood disturbances in patients with post-herpetic neuralgia: results of a randomised, placebo-controlled trial. Pain. 2004;109:26-35.
16. Lyrica (pregabalin) package insert. New York, NY: Parke-Davis Division of Pfizer; November 2006.
17. Sultan A, Gaskell H, Derry S, Moore RA. Duloxetine for painful diabetic neuropathy and fibromyalgia pain: systematic review of randomised trials. BMC Neurol. 2008;8:29.
18. Chong MS, Hester J. Diabetic peripheral neuropathy: current and future treatment options. Drugs. 2007;67:569-585.
19. Gilron I, Bailey J, Tu D, et al. Nortriptyline and gabapentin, alone and in combination for neuropathic pain: a double-blind, randomised controlled crossover trial. Lancet. 2009;374:1252-1261.
20. Hempenstall K, Nurmikko TJ, Johnson RW, et al. Analgesic therapy in postherpetic neuralgia: a quantitative systematic review. PLoS Med. 2005;2:e164.
To comment on this article, contact rdavidson@uspharmacist.com.





