US Pharm. 2011;36(1):62-66.
While estimates vary depending on the definition of insomnia utilized, an estimated 70 million Americans live with a chronic sleep disorder, which is associated with an economic burden greater than $100 billion in direct and indirect costs every year in the United States.1 While only 10% of the adult population experiences chronic insomnia,2,3 in the past 8 years the number of individuals who sleep less than 6 hours a night has risen 13%.4 This increase is quite alarming, especially considering the myriad negative consequences associated with insomnia. Examples include increased use of health care resources, reduced quality of life, effects on physical health, impairment in family and social relationships, and the possibility of emergent psychiatric conditions.5,6 Due to the widespread impact of insomnia, adequate identification and management of this condition are critical. Health care providers must carefully assess those at greatest risk for insomnia, including the elderly, females, shift workers, individuals with comorbid medical and psychiatric conditions, and those with less education.2,3,5
Proper management of chronic insomnia includes the identification of the underlying medical, psychiatric, and psychosocial factors, as well as utilization of nonpharmacologic and pharmacologic treatment.7,8 Pharmacologic options will be discussed here, as well as concerns surrounding the use of these agents that must be considered when determining the optimal management for an individual with insomnia.
Pharmacologic Treatment
Benzodiazepines
Benzodiazepines are one of the most widely used drug classes for the short-term treatment of insomnia. These agents bind to gamma-aminobutyric acid (GABA) receptors in the central nervous system (CNS), causing inhibition of neuronal excitation. Benzodiazepines affect sleep by increasing total sleep time and shortening sleep latency.9,10
There are five benzodiazepines that are FDA approved for the treatment of insomnia, and all are schedule IV controlled substances. Recommended doses, onset and duration of action, half-life, and insomnia indication for these drugs are outlined in TABLE 1.9
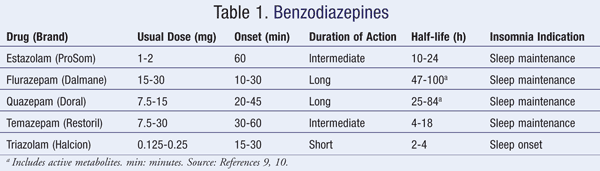
Benzodiazepines are safe and effective for the treatment of insomnia when used short-term at recommended doses; however, due to their mechanism of action, a number of adverse effects can occur.10 These may include changes in sleep architecture (mainly shortened time in both deep and rapid eye movement [REM] sleep and increased time in intermediate sleep), subsequent daytime hangover, anterograde amnesia, mental status changes, psychomotor impairment, rebound insomnia, and withdrawal symptoms upon discontinuation. Other common adverse effects include drowsiness, dizziness, and headache.9 Subsequent daytime hangover is particularly problematic when using benzodiazepines with long durations of action (e.g., flurazepam [Dalmane]). Long half-life agents also increase the risk for poor outcomes in elderly patients, including increased risk of drug accumulation, falls, and confusion.8,10 Since the side effects are mostly dose related, patients should be maintained on the lowest effective dose for the shortest period of time that is necessary to treat the insomnia complaints. The choice of the agent should be patient specific; benzodiazepines should be avoided in pregnancy (Category D or X), in those with a history of substance abuse, and in sleep apnea.8
Nonbenzodiazepines
Nonbenzodiazepines, also known as z-hypnotics, are a relatively new class of drugs for the treatment of insomnia. The mechanism of action of nonbenzodiazepines is similar to that of benzodiazepines but with less severe side effects. Similarly to benzodiazepines, z-hypnotics work through the potentiation of GABA, but they are more selective to the alpha1 subunit of the GABA receptor, which may be more specific for sedation and cause fewer adverse effects.9,10 Since the approval of the first z-hypnotic in 1993, the use of benzodiazepines for the management of insomnia has declined.11
There are three z-hypnotics that are FDA approved for the treatment of insomnia. Like benzodiazepines, they are schedule IV controlled substances. Recommended doses, onset and duration of action, half-life, and insomnia indication for various nonbenzodiazepines are outlined in TABLE 2.9,10 Compared to benzodiazepines, z-hypnotics are generally associated with less rebound insomnia and potential for abuse, as well as less risk of dependency, tolerance, and hangover effect.12 All three z-hypnotics have different pharmacologic properties, adverse effects, and clinical activity.
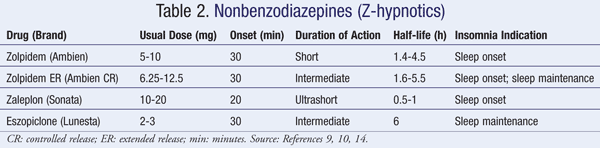
Zolpidem and Extended-Release Zolpidem: Zolpidem (Ambien) works mainly to induce sleep due to its relatively quick onset of action. Its duration of action may be as long as 8 hours in some patients and therefore may increase total sleep time as well.9 Extended-release (ER) zolpidem was FDA approved in 2005 for the treatment of insomnia that is characterized by difficulties in falling asleep and/or staying asleep. Zolpidem ER is a bilayered tablet with the first layer dissolving quickly to help with sleep onset. The second layer gradually releases active drug to help maintain sleep.13 An important difference is that the ER labeling does not restrict use to the short term, while FDA-approved labeling for immediate-release zolpidem is for short-term use only.14 These agents have not been shown to have deleterious effects on sleep architecture. The adverse effects of zolpidem and zolpidem ER are similar and include headache, drowsiness, fatigue, and dizziness. Postmarketing reports revealed that zolpidem may cause parasomnias such as sleep walking and sleep-related eating disorders.10,15
Zaleplon: Zaleplon (Sonata) has a rapid onset of action and a very short duration of action, making it an ideal drug for patients with insomnia characterized by difficulty with sleep onset. Due to the pharmacokinetic profile of zaleplon, there is virtually no next-day hangover effect.10 It can be taken within 4 to 5 hours of wake time, if needed, without the risk of next-day drowsiness,16 and should be taken on an empty stomach for optimal absorption.14 Zaleplon is not associated with tolerance or rebound insomnia and is relatively well tolerated, with the side-effect profile similar to that of placebo.10 Dose-dependent headache appears to be the most common complaint, although a small percentage of patients have reported CNS changes such as drowsiness, incoordination, hallucinations, dizziness, and ataxia.9
Eszopiclone: Eszopiclone (Lunesta) has a rapid onset that helps improved sleep onset, as well as an intermediate duration of action that helps with sleep maintenance. In contrast to other z-hypnotics, the dose can be adjusted to the desired effect. The 1- and 2-mg doses are used to improve sleep onset, while the 3-mg dose can be used for sleep maintenance.17 Eszopiclone is associated with minimal rebound insomnia, withdrawal, and tolerance.9 It is important to note that the eszopiclone labeling does not restrict it for short-term use. The most common adverse effects include unpleasant taste, headache, and dry mouth.17
Melatonin-Receptor Agonists
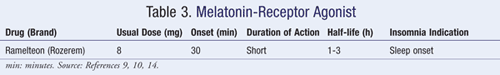
Ramelteon (Rozerem) is the only FDA-approved melatonin-receptor (MT) agonist. It works by binding to the MT1 and MT2 receptors. These receptors are involved in the maintenance of circadian rhythm, which help regulate the sleep-wake cycle. Ramelteon promotes sleep by agonizing the MT1 and MT2 receptors, therefore helping to regulate the sleep-wake cycle.9 Recommended doses, onset and duration of action, half-life, and insomnia indication for ramelteon are outlined in TABLE 3. Ramelteon does not bind to the GABA receptors; therefore, the side-effect profile differs from that of the benzodiazepines and nonbenzodiazepines. It is the only hypnotic sleep agent that is not classed as a controlled substance by the FDA, and it is well tolerated, with the most common side effects including headache, somnolence, dizziness, and fatigue. It is does not cause rebound insomnia or next-day hangover effect. Patients should be instructed to avoid taking ramelteon after a high-fat meal, as this may interfere with absorption.14 There is also a potential for drug interactions, because ramelteon is metabolized by CYP1A2, and it should avoided in patients with severe liver disease.18
Approaches to Treatment
The primary goals for the treatment of insomnia are to improve sleep quality and insomnia-related daytime impairments.19 The majority of studies evaluating treatment effectiveness have focused on improving sleep maintenance and decreasing sleep latency, but few have focused on long-term outcomes, including quality-of-life improvements, job performance, and interpersonal relationships. In addition, few studies have evaluated comparative therapy for the long-term treatment of chronic insomnia.
Most experts agree that all patients should be encouraged to practice good sleep hygiene, even though this alone is not a sufficient treatment for chronic insomnia.19,20 This includes establishing a regular bedtime routine; daily exercise; avoiding eating, drinking, or exercising before bed; and creating a comfortable, sleep-promoting environment.
There is good evidence to support starting with nonpharmacologic treatment alone or in combination with pharmacotherapy. Relaxation techniques and cognitive behavioral therapy (CBT) have proven beneficial for insomnia, and the benefits may be sustained for at least 6 to 24 months. These techniques have their limitations as well. They are often not effective in achieving immediate results, and thus patients abandon the practice in search of faster alternatives. Other limitations include high initial cost, few trained therapists, lack of insurance coverage, and decreased effectiveness in the elderly.20,21
Pharmacologic therapy is recommended, in combination with nonpharmacologic therapy, when a quick response is needed, when the insomnia produces impairment with daily living, when nonpharmacologic methods have not been effective, and when insomnia persists despite treatment of the underlying cause.21 The choice of agent should be based on patient-specific factors, including age, proposed length of treatment, sleep complaints, history of alcohol or drug abuse, psychiatric comorbidity, patient preference, and cost.19 For example, patients with complaints of sleep latency problems may benefit more from agents with a quick onset of action and a short duration of effect, such as zaleplon and temazepam. On the other hand, patients who have trouble maintaining sleep may benefit from agents that are longer acting, such as zolpidem and eszopiclone.
In general, the z-hypnotics (zolpidem, zaleplon, eszopiclone) should be selected for first-line pharmacologic therapy for most patients. Although there are no good studies directly comparing the benzodiazepines with z-hypnotics, indirect comparisons suggest similar efficacy. The incidence of adverse effects, as previously discussed, is lower with z-hypnotics, and these agents also have a lower potential for abuse.19,22 Two z-hypnotics, zolpidem ER and eszopiclone, are also approved for chronic insomnia, which is an advantage in patients with long-term symptoms. However, there is little evidence past the duration of 6 months, and additional studies directly looking at clinical outcomes in patients with chronic insomnia are needed.
Temazepam (Restoril), a short-to-intermediate–acting benzodiazepine, is also an initial treatment option,19 but it should only be used for short-term treatment (<2-4 weeks) due to the development of tolerance and dependence. It should also be avoided in patients with a history of drug or alcohol abuse.
If the initial selection is unsuccessful, it is reasonable to switch to an alternative first-line drug therapy or ramelteon.19 Ramelteon is also a good option for patients with a history of addiction, as it has a lower likelihood of abuse and physical dependence.21
The antidepressants amitriptyline, doxepin, trazodone, and mirtazapine have been shown to have beneficial effects on sleep architecture and may be an effective option in patients with concomitant insomnia and depression.21 Recent evidence suggests that there is no benefit from antidepressants for nondepressed patients with insomnia; however, doxepin, a tricyclic antidepressant (TCA), was recently FDA approved for sleep maintenance in patients without depression.23,24 While antidepressant agents may be useful, particularly in patients with comorbid depression, concerns with these agents include anticholinergic effects with TCAs, as well as orthostatic hypotension and hangover effects with trazodone.8
Anticholinergic effects and tolerance are also problematic with OTC antihistamine products, and these medications should not be taken by elderly patients.25 Some patients may feel that alternative therapies, such as use of melatonin, are more acceptable than prescription medications, but concerns of regulation, lack of long-term safety data, safety in pregnancy, and drug interactions make health care professionals hesitant to recommend these options in most cases.25 At this time, OTC antihistamines, herbal and dietary supplements, alcohol, barbiturates, and opiates should not be used to treat primary insomnia because of insufficient evidence as well as the potential for adverse effects.21
Conclusion
Prior to initiating treatment for insomnia, other medical or psychiatric conditions, as well as medications that may contribute to insomnia, need to be considered. If pharmacologic treatment is deemed appropriate for an individual with insomnia, nonpharmacologic interventions, including appropriate sleep hygiene and CBT, if available, should be initiated as well. Pharmacologic therapy should be utilized for the shortest duration possible, initiated in low doses, and titrated to the lowest effective dose to minimize concerns for dependence on controlled substances, as well as to reduce the risk for adverse events. Upon treatment discontinuation, medications should be withdrawn slowly to avoid rebound insomnia. Lastly, patient education regarding the expectations of treatment and possible adverse effects can help maximize treatment outcomes.22
REFERENCES
1. Fullerton DS. The economic impact of insomnia in managed care: a clearer picture emerges. Am J Manag Care. 2006;12(suppl 8):S246-S452.
2. Roth T, Roehrs T. Insomnia: epidemiology, characteristics, and consequences. Clin Cornerstone. 2003;5:5-15.
3. Walsh JK. Clinical and socioeconomic correlates of insomnia. J Clin Psychiatry. 2004;65(suppl 8):13-9.
4. National Sleep Foundation. 2009 Sleep in America Poll highlights and key findings. March 2, 2009. www.sleepfoundation.org/sites/
5. Buscemi N, Vandermeer B, Friesen C, et al. Manifestations and Management of Chronic Insomnia in Adults. Evidence Report/Technology Assessment No. 125. Rockville, MD: Agency for Healthcare Research and Quality; June 2005. AHRQ pub no. 05-E021-2.
6. Ohayon MM, Reynolds CF. Epidemiological and clinical relevance of insomnia diagnosis algorithms according to the DSM-IV and the International Classification of Sleep Disorders (ICSD). Sleep Med. 2009;10:952-960.
7. Sateia MJ, Doghramji K, Hauri PJ, Morin CM. Evaluation of chronic insomnia. Sleep. 2000;23:1-39.
8. Dopp JM, Phillips BG. Sleep disorders. In: DiPiro JT, Talbert RL, Yee GC, et al, eds. Pharmacotherapy: A Pathophysiologic Approach. 7th ed. New York, NY: McGraw-Hill; 2008.
9. Morin AK, Jarvis CI, Lynch AM. Therapeutic options for sleep-maintenance and sleep-onset insomnia. Pharmacotherapy. 2007;27:89-110.
10. Passarella S, Duong MT. Diagnosis and treatment of insomnia. Am J Health Syst Pharm. 2008;65:927-934.
11. Walsh JK, Schweitzer PK. Ten-year trends in the pharmacological treatment of insomnia. Sleep. 1999;22:371-375.
12. Winkelman J, Pies R. Current patterns and future directions in the treatment of insomnia. Ann Clin Psychiatry. 2005;17:31-40.
13. Ambien CR (zolpidem extended release) package insert. Bridgewater, NJ: Sanofi-Aventis; December 2007.
14. Clinical Pharmacology [database online]. Tampa, FL: Gold Standard, Inc; 2010. Updated September 2010. www.clinicalpharmacology.com. Accessed October 1, 2010.
15. Lee YJ. Overview of the therapeutic management of insomnia with zolpidem. CNS Drugs. 2004;18(suppl 1):17-24.
16. Walsh JK, Pollak CP, Scharf MB, et al. Lack of residual sedation following middle-of the-night zaleplon administration in sleep maintenance insomnia. Clin Neuropharmacol. 2000;23:17-21.
17. Lunesta (eszopiclone) package insert. Marlborough, MA: Sepracor Inc; March 2009.
18. Rozerem (ramelteon) package insert. Deerfield, IL: Takeda Pharmaceuticals America, Inc; October 2008.
19. Schutte-Rodin S, Broch L, Buysse D, et al. Clinical guideline for the evaluation and management of chronic insomnia in adults. J Clin Sleep Med. 2008;4:487-504.
20. Summers MO, Crisostomo MI, Stepanski EJ. Recent developments in the classification, evaluation, and treatment of insomnia. Chest. 2006;130:276-286.
21. Ramakrishnan K, Scheid DC. Treatment options for insomnia. Am Fam Physician. 2007;76:517-526.
22. Budur K, Rodriguez C, Foldvary-Schaefer N. Advances in treating insomnia. Cleve Clin J Med. 2007;74:251-266.
23. Mendelson WB. A review of the evidence for the efficacy and safety of trazodone in insomnia. J Clin Psychiatry. 2005;66:469-476.
24. Silenor (doxepin) package insert. San Diego, CA: Somaxon Pharmaceuticals, Inc.; March 2010.
25. Kirkwood CK, Melton ST. Insomnia. In: Berardi RR, Ferreri SP, Hume AL, et al, eds. Handbook of Nonprescription Drugs: An Interactive Approach to Self-Care. 16th ed. Washington, DC: American Pharmacists Association; 2009.
To comment on this article, contact rdavidson@uspharmacist.com.





