US Pharm. 2012;37(9):48-53.
According to the CDC’s Pregnancy Mortality Surveillance System, hypertensive disorders account for 11.1% of all causes of pregnancy-related death in the United States during the 2006–2007 period.1 Preeclampsia, in particular, represents a unique form of pregnancy-related hypertension. It is characterized by a series of multisystem abnormalities usually occurring after 20 weeks of gestation with an ensuing diagnosis of new-onset hypertension (blood pressure [BP] >140/90 mmHg) and proteinuria (a finding of protein in the urine >300 mg over a 24-hour period).2,3 In contrast, women who develop new-onset grand mal seizures in the presence of preeclampsia are deemed eclamptic and require immediate medical intervention.3
A PATHOPHYSIOLOGIC GUMBO
Despite decades of extensive clinical research and analysis, identifying the origin of preeclampsia remains an elusive target. Preeclampsia is a heterogeneous syndrome in which pathogenesis can be diverse among women with various risk factors. Pathologic variations in preeclampsia in nulliparous women may be different from those in women with preexisting cardiovascular disease, twin gestation, diabetes mellitus, chronic hypertension, or various thrombophilias.4,5 In addition, the pathophysiology of early-onset preeclampsia may be different from that of preeclampsia developing at term, during labor, or in the postpartum period. Disparities in the onset of preeclampsia might lead to different preventive and pharmacologic interventions.6 The causes of preeclampsia tend to reflect a pathophysiologic gumbo. Theories most commonly proposed for the etiology of preeclampsia include endothelial dysfunction, placental abnormalities, blood coagulation disorders, cardiovascular and immunologic maladaptation, genetic predisposition, and excessive inflammatory and oxidative stress, just to name a few.4,7-9
PREECLAMPSIA AT A GLANCE
Preeclampsia is a syndrome that embraces a wide spectrum of symptomatology. Traditionally, elevated BP and proteinuria served as primary indicators for confirmation of this disorder. Other signs and symptoms may also be present and can parallel expected symptoms seen throughout the pregnancy stages. In some cases, preeclampsia can be asymptomatic and only be discovered upon routine screening.10
Preeclampsia can be subdivided into two categories based on symptomatology—mild or severe. Mild preeclampsia by definition is the presence of hypertension (BP >140/90 mmHg) on two occasions at least 6 hours apart, but without evidence of end-organ damage in the patient.10 Severe preeclampsia, on the other hand, may include an array of signs and symptoms and significant laboratory findings. At least one of the signs or symptoms presented in TABLE 1 must coexist with the indicators of preeclampsia for the condition to be regarded as severe. Differentiating the characteristics of mild preeclampsia from those of severe preeclampsia is essential because this influences management.11
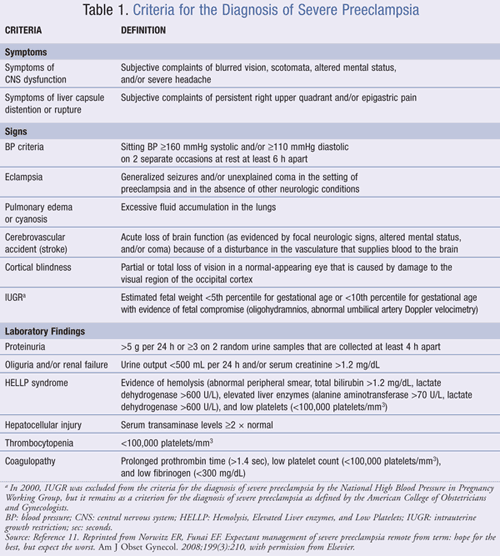
HIGH-RISK PREGNANCIES
Currently, there are no validated screening tools recommended to predict or identify the patient at risk for development of preeclampsia. Therefore, pregnant women should be informed and vigilant in monitoring for the signs and symptoms and communicating with their health care providers about any problems that could lead to unfavorable complications.3 Pregnancies considered most at risk are based on personal history, preexisting medical conditions, and family history. Examples of high-risk pregnancies include woman’s age ≥35 years, first pregnancy, preeclampsia in a previous pregnancy, chronic hypertension, gestational diabetes, multifetal gestations, antiphospholipid antibody syndrome, and renal disease.4,5
Obesity has been shown to have a strong correlation with the development of preeclampsia. In a study conducted in the United Kingdom, results indicated that 9% of extremely obese women experienced preeclampsia in comparison with 2% of matched controls.5,12 With the growing concerns about obesity worldwide, large studies are needed for confirmation of this plausible relationship; preventive measures will be crucial to thwart the potential impact upon the incidence of preeclampsia.
MANAGEMENT OF PREECLAMPSIA
The clinical course of preeclampsia can lead to devastating effects for both the mother and the fetus. Characteristics such as gestational age and disease severity are key components in identifying the most appropriate methods for managing patients at risk.10 Goals of management should emphasis safety of the mother and delivery of a healthy infant, while minimizing risk to both. The American College of Obstetricians and Gynecologists (ACOG) recommends that management of women with severe preeclampsia remote from term be monitored in a tertiary care setting or with the advice of an obstetrician-gynecologist who specializes in high-risk pregnancies.3
In women with severe preeclampsia where HELLP (evidence of Hemolysis, Elevated Liver enzymes, and Low Platelets) syndrome is present, delivery is the management of choice, despite gestational age.3,5 HELLP is usually characterized by progressive and sometimes sudden deterioration in both maternal and fetal condition. Maternal complications can include eclampsia, abruptio placentae, acute renal failure, and postpartum bleeding, whereas the unborn child may experience intrauterine growth restriction (IUGR), thrombocytopenia, or even perinatal death; thus, the management of HELLP syndrome is highly controversial.5,11,13,14 Based on the weeks of gestation, some clinicians may administer corticosteroids in the attempt to accelerate fetal lung maturity followed by delivery after 24 hours, while others may opt to prolong pregnancy until maternal or fetal indications dictate delivery. In the latter case, expectant management is instituted through the following measures: bed rest, antihypertensive agents, antithrombotic agents, plasma volume expanders, and corticosteroids.5,11 Current recommendations state that expectant management should only be considered in those with mild preeclampsia.3
THERAPEUTIC MODALITIES
The therapeutic options presented below are the most common drugs used in the management of seizures and stabilization of BP in severe preeclampsia.
Prophylaxis and Treatment of Seizures
Magnesium Sulfate: Magnesium sulfate has been utilized for seizure control since the 1920s, but it received its first confirmation as an efficacious treatment for severe preeclampsia and eclampsia in 1995 through the Collaborative Eclampsia Trial.15 This was a multicenter, international, randomized, placebo-controlled study that included over 10,000 women. Those treated with magnesium sulfate had a 52% and 67% lower recurrence of convulsions than those treated with diazepam and phenytoin, respectively. In patients with severe preeclampsia, the use of magnesium reduced the risk of progression to eclampsia by more than half and reduced maternal mortality.15 A 2-year outcome follow-up trial was also conducted and revealed that women receiving magnesium sulfate therapy had a 58% lower risk of eclampsia (95% CI, 40%-71%) than women receiving placebo (0.8% risk in patients receiving magnesium sulfate versus 1.9% in patients receiving placebo). The results of this trial suggest that magnesium sulfate therapy prevents eclampsia and may reduce the risk of maternal death.16
Magnesium sulfate is considered first-line treatment for the prevention of primary and recurrent eclamptic seizures. It is also utilized for prophylactic treatment in all patients with severe preeclampsia.15,16 The mechanism of action of magnesium sulfate is thought to trigger cerebral vasodilation, thus reducing ischemia generated by cerebral vasospasm during an eclamptic event. The substance also acts competitively in blocking the entry of calcium into synaptic endings, thereby altering neuromuscular transmission. The recommended regimen of magnesium sulfate is a loading dose of 4 to 6 g given over 15 to 20 minutes, followed by a maintenance dose of 2 g/h as a continuous IV solution.3,17-19 Magnesium sulfate is initiated at the beginning of the observation period and then continued during labor and for at least 24 hours postpartum. In those with abnormal renal function (oliguria or serum creatinine ≥1.2 mg/dL), the dose of magnesium sulfate should be reduced and even discontinued.5,20 The therapeutic serum magnesium concentration range is considered to be 4 to 8 mg/dL. Signs of toxicity start with the loss of patellar deep tendon reflexes, weakness, double vision, and dysarthria. Respiratory depression/or arrest can occur with levels >14 mg/dL.17
Controlling Elevated Blood Pressure
Treating acute severe hypertension in preeclampsia is crucial to preventing cerebrovascular and cardiovascular events as well as maternal death. Antihypertensive therapies should be utilized in women with systolic BP values of 160 to 180 mmHg or higher19 and diastolic BP values of 105 to 110 mmHg or higher.3,19 TABLE 2 summarizes these agents.
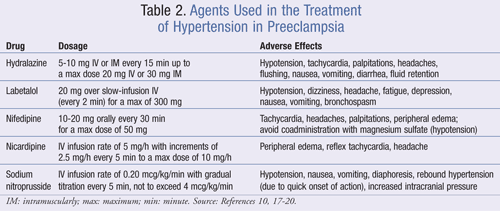
Hydralazine: Hydralazine is an arteriolar vasodilator that has long been used for lowering BP in severe hypertension and preeclampsia in pregnancy. It is administered as a 5- to 10-mg dose IV or IM every 15 minutes up to a maximum dose of 20 mg IV or 30 mg IM. The onset of action is 10 to 20 minutes. BP is recorded every 15 minutes during therapy and every hour once the desired values are achieved. If hydralazine does not lower BP adequately or if maternal side effects such as tachycardia or headaches develop, another agent should be considered.10,17-20
Labetalol: Labetalol is a nonselective, competitive beta-adrenergic and a selective, competitive alpha1-adrenergic blocking agent. The mechanism of action is exerted by reduction of peripheral vascular resistance without compromising blood flow to the brain and peripheral, coronary, or renal systems. The recommended dose of labetalol is 20 mg over slow-infusion IV (every 2 minutes) for a maximum dosage of 300 mg.19,20 Potential benefits of labetalol over hydralazine include quicker onset of action and less risk for reflex tachycardia. Labetalol, however, should be avoided in patients with moderate-to-severe asthma, bradycardia (heart rate <60 beats per minute), or congestive heart failure.17-20
Nifedipine: Nifedipine is an oral, type 2 calcium channel blocker that inhibits the inward flow of calcium across slow channels of cellular membranes. It reduces BP without compromise to placental blood flow. Compared with hydralazine, nifedipine has a quicker onset of action, as well as the added advantage of oral administration. Nifedipine should only be given as an oral short-acting preparation in an initial dose of 10 to 20 mg orally every 30 minutes for a maximum dosage of 50 mg.20 Common side effects include tachycardia, headaches, and palpitations. Marked hypotension with magnesium sulfate and nifedipine has been reported; therefore, concomitant use should be avoided.10,20
Nicardipine: Nicardipine hydrochloride is a calcium channel blocker that may be an alternative to the agents previously discussed. It causes less tachycardia than nifedipine and provides BP-lowering effects within an average of 15 minutes following IV administration. Nicardipine is initiated as an IV infusion at a rate of 5 mg/h with increments of 2.5 mg/h every 5 minutes to a maximum dosage of 10 mg/h or until the mean arterial pressure is reduced by 15%.17,20
Sodium Nitroprusside: When the aforementioned agents fail to lower the BP in severe preeclampsia, sodium nitroprusside may be given. It is a vasodilatory agent that works by releasing nitrous oxide, which reduces both preload and afterload. The onset of action is very quick and can result in severe rebound hypertension. Additionally, the patient may experience headaches, palpitations, and cyanide poisoning. If severe hypertensive emergency necessitates the use of sodium nitroprusside, the dosage should be initiated as an IV infusion with a rate of 0.20 mcg/kg/min with gradual titration every 5 minutes, not to exceed 4 mcg/kg/min.17,20
CONCLUSION
Without clear national consensus guidelines on the origin of preeclampsia, the clinician is faced with management challenges and maternal and fetal risk-versus-benefit issues. The key to prevention lies in identifying those patients most at risk and closely monitoring their clinical and laboratory progress. The main objectives of treating preeclampsia include avoiding complications of severe hypertension, preventing the development of eclampsia, and decreasing the morbidity and mortality of the mother and unborn child. Pharmacists can play an integral role in providing early education to those expectant mothers at high risk for developing preeclampsia.
REFERENCES
1. Pregnancy-related mortality in the United States. CDC
Reproductive Health.
www.cdc.gov/reproductivehealth/MaternalInfantHealth/Pregnancy-relatedMortality.htm#data.
Accessed May 1, 2012.
2. Report of the National High Blood Pressure Education Program Working Group on High Blood Pressure in Pregnancy. Am J Obstet Gynecol. 2000;183(suppl 1):S1-S22.
3. American College of Obstetricians and Gynecologists.
Diagnosis and management of preeclampsia and eclampsia. ACOG Practice
Bulletin No. 33. Am J Obstet Gynecol. 2002;99:159-167.
4. Cudihy D, Lee RV. The pathophysiology of pre-eclampsia: current clinical concepts. J Obstet Gynaecol. 2009;29:576-582.
5. Sibai BM. Hypertension. In: Gabbe SG, Niebyl JR, Simpson JL, et al, eds. Obstetrics: Normal and Problem Pregnancies. 6th ed. Philadelphia, PA: Saunders; 2012:779-823.
6. Valensise B, Vasapollo B, Gagliardi G, Novelli GP.
Early and late preeclampsia: two different maternal hemodynamic states
in the latent phase of the disease. Hypertension. 2008;52:873-880.
7. Alladin AA, Harrison M. Preeclampsia: systemic
endothelial damage leading to increased activation of the blood
coagulation cascade. J Biotech Res. 2012;4:26-43.
8. Vatten L, Skjaerven R. Is pre-eclampsia more than one disease? BJOG. 2004;111:298-302.
9. Ahn H, Park J, Gilman-Sachs A, Kwak-Kim J. Immunologic characteristics of preeclampsia, a comprehensive review. Am J Reprod Immunol. 2011;65:377-394.
10. Lim KH, Erogul M, Sayah A, et al. Preeclampsia. Medscape Reference. http://emedicine.medscape.com/article/1476919-overview. Accessed April 23, 2012.
11. Norwitz ER, Funai EF. Expectant management of severe
preeclampsia remote from term: hope for the best, but expect the worst. Am J Obstet Gynecol. 2008;199:209-212.
12. Knight M, Kurinczuk JJ, Spark P, Brocklehurst P. Extreme obesity in pregnancy in the United Kingdom. Obstet Gynecol. 2010;115:989-997.
13. Harman K, Svendesn E, Abildgaard U. The HELLP syndrome: clinical issues and management. A review. BMC Pregnancy Childbirth. 2009;9:8:1-15 www.biomedcentral.com/1471-2393/9/8/prepub. Accessed April 23, 2012.
14. Sibai BM, Barton JR. Expectant management of severe
preeclampsia remote from term: patient selection, treatment, and
delivery indications. Am J Obstet Gynecol. 2007;196:514.e1-514.e9.
15. The Eclampsia Trial Collaborative Group. Which
anticonvulsant for women with eclampsia? Evidence from the Collaborative
Eclampsia Trial. Lancet. 1995;345:1455-1463.
16. Altman D, Carroli G, Duley L, et al. Do women with
pre-eclampsia, and their babies, benefit from magnesium sulphate? The
Magpie Trial: a randomised placebo-controlled trial. Lancet. 2002;359:1877-1890.
17. McCoy S, Baldwin K. Pharmacotherapeutic options for the treatment of preeclampsia. Am J Health Syst Pharm. 2009;66:337-344.
18. Lindheimer MD, Taler SJ, Cunningham FG. ASH position paper: hypertension in pregnancy. J Clin Hypertens. 2009;11:214-225.
19. Wagner LK. Diagnosis and management of preeclampsia. Am Fam Physician. 2004;70:2317-2324.
20. Drug information. Micromedex Healthcare Series
[subscription required]. www.thomsonhc.com/micromedex2/librarian.
Accessed May 21, 2012.
To comment on this article, contact rdavidson@uspharmacist.com.





