US Pharm. 2013;38(11):HS8-HS14.
ABSTRACT: During the moderate and late stages of dementia, patients experience behavioral and psychological symptoms of dementia (BPSD), such as agitation, psychosis, and aggression. There is no FDA-approved treatment for BPSD, but antipsychotics are frequently prescribed off-label. Antipsychotics yield only modest improvements in BPSD and have been shown to increase death in elderly patients with dementia. It is recommended that, as a part of patient-centered care, nursing homes involve consultant pharmacists in the development and review of medication-safety procedures.
Dementia is a disorder characterized by cognitive and functional impairment. Alzheimer disease (AD), the most common form of chronic dementia, affected 4.7 million Americans in 2010. The number of patients with AD is expected to approach 13.8 million by 2050 and will cost the United States an estimated $1.1 trillion per year in drug and medical costs.1
In the moderate to late stages of AD, patients have difficulty communicating, completing complex tasks, and remembering new information. Additionally, behavioral and psychological symptoms of dementia (BPSD), such as delusions, wandering, aggression, and agitation, occur in 60% to 90% of patients with dementia.2 These symptoms are associated with greater functional impairment and frequently lead to nursing home placement.3
Currently, there is no FDA-approved treatment for BPSD. Antipsychotics have evidence of slight efficacy; however, reports of increased mortality, cerebrovascular events, and cardiovascular effects in elderly patients prompted the FDA to issue a black box warning for these agents.
Safety Concerns
Mortality: There is strong evidence that both atypical antipsychotics and typical antipsychotics (TABLE 1) increase mortality in elderly patients with dementia. An FDA meta-analysis of trials involving elderly patients with BPSD who were taking aripiprazole, olanzapine, quetiapine, or risperidone revealed a 4.5% incidence of death in treatment groups versus a 2.6% incidence in placebo groups. The relative risk—1.7—was consistent among all treatment groups.4 However, in a retrospective study, the highest mortality occurred with risperidone compared with olanzapine and quetiapine. The difference in risk between agents appeared to decrease after 2 months, despite risk persisting for up to 24 months. The most common causes of death were pulmonary infection and cardiovascular events.5,6 Overall, the Agency for Healthcare Research and Quality (AHRQ) estimates 1 death per 100 patients receiving an atypical antipsychotic for BPSD per 10-week period.7
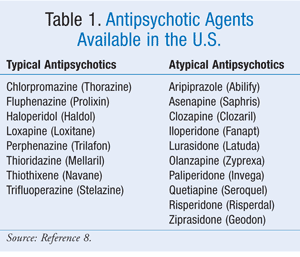
Prior to 2005, very little data existed regarding the safety of typical antipsychotics, leading some clinicians to believe that the older agents were safer. Data on comparative safety are conflicting. One study found a dose-dependent 37% increase in death in the first 6 months of treatment with typical agents.8 A large community cohort study also found higher mortality with typical agents (12.7%) than with atypical agents. In this study, the most frequently used typical and atypical antipsychotics were loxapine and quetiapine, respectively.9 Likewise, a small retrospective analysis found a statistically significant increase in mortality over 2 years in patients taking haloperidol compared with those taking risperidone or olanzapine (21.4% vs. 4.75%, P <.00001).10
Studies examining the risk of death from pneumonia with use of atypical agents versus typical agents also have produced conflicting results. Observational studies have shown a higher risk of pneumonia in community dwellers taking atypical agents, but in the nursing home setting, typical agents are associated with an increased risk of pneumonia.11,12 The likely cause of pneumonia is aspiration precipitated by excessive sedation and/or extrapyramidal side effects (EPS).
Cerebrovascular Events: Both classes of antipsychotics carry a black box warning for increased risk of cerebrovascular events. The risk of stroke or transient ischemic attack is estimated to be two times greater in antipsychotic users with dementia than in antipsychotic users without dementia.12 The risk, which appears to decline after 3 months, is greatest during the first week of treatment and in patients who are already at high risk for stroke.13 Risperidone was the first drug to receive a black box warning for cerebrovascular events, but subsequent trials have shown a similar incidence among all antipsychotics in both classes.6
Cardiovascular Events: The risk of cardiovascular events such as myocardial infarction, arrhythmias, and syncope are similar between both classes of antipsychotics. A meta-analysis from the AHRQ found that 1 in every 48 elderly patients taking olanzapine will have a cardiovascular event, compared with 1 in every 34 taking risperidone.7 Ziprasidone and the low-potency typical antipsychotics chlorpromazine and thioridazine appear to be associated with the greatest risk of QTc prolongation, which can lead to fatal arrhythmias.6 Periodic ECGs are recommended for patients with risk factors for arrhythmia who are taking an antipsychotic or who are taking medications that prolong the QTc interval.
Other Side Effects: Anticholinergicity, weight gain, and EPS (abnormal involuntary movements) are well-known side effects of antipsychotics. In the elderly, anticholinergic agents increase the risk of falls, constipation, and urinary retention. Olanzapine, clozapine, quetiapine, and low-potency typical antipsychotics have pronounced anticholinergic effects and can worsen cognition.7 Therefore, the anticholinergic load of all patient medications should be monitored.
Weight gain appears to be less pronounced in older patients than in younger patients. Even so, the risk of hyperglycemia and hypercholesterolemia should be considered in patients with preexisting diabetes or heart disease. Classwide, atypical agents are more likely to cause metabolic disturbances, with clozapine, risperidone, and olanzapine conferring the greatest risk.7
EPS include dystonia, parkinsonism, and tardive dyskinesia. The AHRQ reports that olanzapine carries the greatest risk of inducing EPS in the elderly population; 1 out of every 10 patients taking olanzapine will experience EPS. Risperidone causes 1 case for every 20 patients treated.7 It is recommended that all facilities administer the Abnormal Involuntary Movements Scale at baseline and every 6 months thereafter (or sooner, if clinically indicated).
Reducing Inappropriate Use
Despite serious safety concerns and minimal overall effect on symptomatology, antipsychotics are frequently prescribed off-label for BPSD. In 2010, the Centers for Medicare and Medicaid Services (CMS) reported that 39% of U.S. nursing home residents were inappropriately prescribed an antipsychotic without having a clinical indication for treatment.14
According to CMS guidelines, it is appropriate to consider the use of an antipsychotic to treat delusions, hallucinations, and/or aggression in patients with dementia, but only if these symptoms are dangerous and/or severely distressing to the patient or others. Behaviors such as wandering, hoarding, and apathy are not considered appropriate treatment targets (TABLE 2).15 Additionally, modifiable causes of behavioral problems should always be definitively ruled out prior to beginning treatment with an antipsychotic. TABLE 3 lists conditions that can cause behavioral problems and/or mimic dementia.15,16


To thwart the practice of prescribing antipsychotics for the chief purpose of behavior control or sedation, the CMS issued a mandate in 2012—since renewed for 2013—that all nursing homes reduce unnecessary use of antipsychotics by at least 15%. CMS regulation F329 (commonly known as F-Tag 329) defines unnecessary use as excessive dose, excessive duration, inadequate monitoring, lack of clinical indication for use, and presence of adverse events.15
It is important to note that none of the regulations is meant to impede patient care; the CMS recognizes that the benefit may exceed the risk in patients with appropriate clinical indications.15 The risks and benefits of therapy, as well as the establishment of a reasonable expectation of symptom control, should be discussed with the patient and caregivers prior to initiation.
Gradual Dose Reduction
To be considered fully compliant with F-Tag 329, a facility must attempt a gradual dose reduction (GDR) of all antipsychotics—including those used for psychiatric diagnosis—twice within the first year of antipsychotic treatment. Attempts should be made in two separate quarters at least 1 month apart. After the first year, a GDR should be attempted annually. To avoid discontinuation syndrome, the dose should be reduced by no more than 50% every 2 weeks.15
However, not all patients are candidates for a dose reduction. GDR is contraindicated in patients with dementia if the patient’s symptoms worsened as a result of a prior GDR attempt or if the physician provides a specific rationale for why a GDR would harm the patient. For patients without dementia who are taking antipsychotics for a psychiatric condition, a GDR is contraindicated if the treatment is in accordance with current practice guidelines or if the patient’s symptoms had worsened after a previous GDR attempt. Reasons for contraindication should be documented in the medical record; otherwise, the facility could be cited as noncompliant.15
Even in the absence of a GDR, all patients should be given the lowest clinically effective dose for the shortest duration. Since many adverse events, particularly EPS and cardiovascular events, appear to be dose-related, the CMS has published maximum daily doses for antipsychotics used to treat BPSD (TABLE 4).15
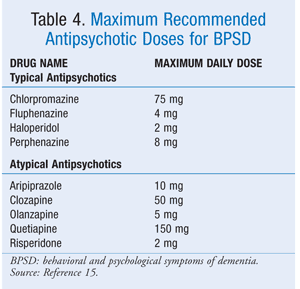
If the patient has not exhibited the expected treatment response by the time the dose approaches the upper threshold, modifiable causes of BPSD should be reevaluated and an alternative therapy should be considered.15
Efficacy
Initial antipsychotic drug selection should be guided by comparative-efficacy data. The Clinical Antipsychotic Trials of Intervention Effectiveness—Alzheimer’s Disease (CATIE-AD) evaluated median time to discontinuation in elderly AD subjects taking olanzapine (mean end-study dose 5.5 mg), quetiapine (56.5 mg), or risperidone (1 mg) compared with placebo.17 The median time to discontinuation due to lack of efficacy was 26.1 weeks for risperidone and 22 weeks for olanzapine; the median time for both quetiapine and placebo was 9 weeks (P = .002). There were no significant differences in clinical-improvement scales among the treatment groups, however. Discontinuation due to intolerability was greatest for olanzapine (24%); risperidone and quetiapine had similar discontinuation rates (18% and 16%, respectively), but placebo was favored, with a discontinuation rate of 5% (P = .009).17
The AHRQ conducted a comparative-efficacy review to examine the strength of evidence of efficacy of atypical agents in treating overall BPSD, agitation, and psychosis compared with placebo. Based on results of this review, risperidone is the only agent with strong evidence of efficacy for overall BPSD, agitation, and psychosis; aripiprazole also has strong evidence of efficacy for treating overall BPSD in dementia, whereas olanzapine and quetiapine have low evidence of efficacy. Risperidone is the only agent with strong efficacy data for treatment of psychosis in dementia; for agitation, olanzapine has proven efficacy. Because of a paucity of evidence supporting its use, quetiapine is not recommended unless other agents have failed. The AHRQ noted, however, that despite strong evidence of efficacy, the magnitude of treatment effect on clinical improvement was small and did not reach statistical significance.17
The efficacy of typical agents used to treat BPSD has not been studied as extensively as that of the newer agents. Of these agents, haloperidol has the most clinical data for agitation in dementia.18 However, its utility is diminished by reports of high mortality and risk of EPS.
Role of the Pharmacist
The Nursing Home Reform Act, which is part of the Omnibus Budget Reconciliation Act of 1987, requires facilities to provide patient-centered care to all residents.15 As part of its quality-improvement plan, the CMS recommends that all facilities include a consultant pharmacist on their quality-care committees, particularly to help develop and review procedures for medication safety.7 Patient-centered care of the elderly is possible when a team-based approach including representatives from all healthcare professions is utilized.
REFERENCES
1. Hebert LE, Weuve J, Scherr PA, Evans DA. Alzheimer disease in the United States (2010-2050) estimated using the 2010 census. Neurology. 2013;80:1778-1783.
2. Ernst RL, Hay JW. The US economic and social costs of Alzheimer’s disease revisited. Am J Public Health. 1994;84:1261-1264.
3. Scarmeas N, Brandt J, Blacker D, et al. Disruptive behavior as a predictor in Alzheimer disease. Arch Neurol. 2007;64:1755-1761.
4. Singh S, Wooltorton E. Increased mortality among elderly patients with dementia using atypical antipsychotics. CMAJ. 2005;173:252.
5. Kales HC, Kim HM, Zivin K, et al. Risk of mortality among individual antipsychotics in patients with dementia. Am J Psychiatry. 2012;169:71-79.
6. Steinberg M, Lyketsos CG. Atypical antipsychotic use in patients with dementia: managing safety concerns. Am J Psychiatry. 2012;169:901-906.
7. Maglione M, Maher AR, Hu J, et al. Off-label use of atypical
antipsychotics: an update [Internet]. Rockville, MD: Agency for
Healthcare Research and Quality; September 2011. Comparative
Effectiveness Reviews, No. 43.
www.effectivehealthcare.ahrq.gov/offlabelantipsych.cfm. Accessed July
28, 2013.
8. Wang PS, Schneeweiss, Avorn J, et al. Risk of death in elderly users of conventional vs. atypical antipsychotic medications. N Engl J Med. 2005;353:2335-2341.
9. Setoguchi S, Wang PS, Alan Brookhart M, et al. Potential causes of
higher mortality in elderly users of conventional and atypical
antipsychotic medications. Am J Geriatr Soc. 2008;56:1644-1650.
10. Nasrallah HA, White T, Nasrallah AT. Lower mortality in geriatric
patients receiving risperidone and olanzapine versus haloperidol. Am J Geriatr Psychiatry. 2004;12:437-439.
11. Aparasu RR, Chatterjee S, Chen H. Risk of pneumonia in elderly nursing home residents using typical versus atypical agents. Ann Pharmacother. 2013;47:464-474.
12. Huybrechts KF, Schneeweiss S, Gerhard T, et al. Comparative safety of antipsychotic medications in nursing home residents. J Am Geriatr Soc. 2012;60:420-429.
13. Douglas IJ, Smeeth L. Exposure to antipsychotics and risk of stroke: self controlled case series study. BMJ. 2008;337:a1227.
14. Department of Health and Human Services Office of the Inspector
General. Medicare atypical antipsychotic drug claims for elderly nursing
home residents. May 2011.
http://oig.hhs.gov/oei/reports/oei-07-08-00150.pdf. Accessed July 28,
2013.
15. Centers for Medicare and Medicaid Services. Dementia care in
nursing homes: clarification to Appendix P State Operations Manual (SOM)
and Appendix PP in the SOM for F309—quality of care and
F329—unnecessary drugs.
www.cms.gov/Medicare/Provider.../Survey-and-Cert-Letter-13-35.pdf.
Accessed July 28, 2013.
16. Downing LJ, Caprio TV, Lyness JM. Geriatric psychiatry review:
differential diagnosis and treatment of the 3 D’s—delirium, dementia,
and depression. Curr Psychiatry Rep. 2013;15:365-375.
17. Schneider LS, Tariot PN, Dagerman KS, et al. Effectiveness of
atypical antipsychotic drugs in patients with Alzheimer’s disease. N Engl J Med. 2006;355:1525-1538.
18. Chahine LM, Acar D, Chemali Z. The elderly safety imperative and antipsychotic usage. Harv Rev Psychiatry. 2010;18:158-172.
To comment on this article, contact rdavidson@uspharmacist.com.






