US Pharm. 2009;34(9):HS-3-HS-6.
Clinical hypothyroidism is characterized by decreased production of thyroid hormones. Approximately five million Americans are affected by this disease state, which has a strong familial correlation; the condition is 10 times more prevalent in women than in men, occurring in approximately 1.5% to 2.0% of women and 0.2% of men.1,2 These estimates do not include cases of subclinical and congenital hypothyroidism. Subclinical hypothyroidism, characterized by normal thyroid hormone levels and marginally elevated levels of thyroid-stimulating hormone (TSH), affects approximately 20% of people over the age of 60 in the United States.3 Congenital hypothyroidism, a condition involving a lack of thyroid function at birth, occurs in 1 in 4,000 live births worldwide.4 This article attempts to provide a current understanding of hypothyroidism.
Synthesis and Function of Thyroid Hormones
Synthesis: The thyroid, a gland located on the ventral surface of the trachea, produces two hormones: thyroxine (T4) and triiodothyronine (T3). Hormone secretion is regulated by the hypothalamus and the pituitary gland. The hypothalamus secretes thyrotropin-releasing hormone (TRH), which in turn stimulates the anterior pituitary to release TSH. TSH binds to receptors on the thyroid gland to synthesize and release T4 and T3.
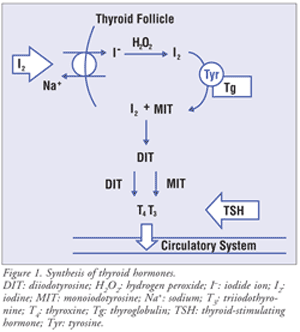
FIGURE 1 depicts the steps involved in the synthesis of thyroid hormones.5 The thyroid is designed to trap the rare element iodine, which is present in the earth's mantle. Iodine ingested through food is absorbed from the gastrointestinal tract and circulated in the blood. This circulated iodine is actively transported into the thyroid follicle, the smallest unit of thyroid gland, by sodium-iodide exchange. In the thyroid follicle, iodide ions are oxidized by hydrogen peroxide to form active iodine. Tyrosination of iodide ions is a critical step in the synthesis of thyroid hormones. Active iodine binds to tyrosine residues on thyroglobulin (Tg), a glycoprotein, to form monoiodotyrosine (MIT). Another iodine atom may bind to MIT to form diiodotyrosine (DIT). DIT combines with either MIT or DIT to form T3 or T4, respectively. Influenced by TSH, Tg undergoes hydrolysis, and the thyroid secretes T4 and T3 into circulation in a ratio of approximately 9 to 1. The enzyme deiodinase, present throughout the body, converts T4 to the more active hormone T3. It should be noted that T3 is 10 times more active than T4. In systemic circulation, T4 and T3 account for 80% and 20% of thyroid hormones, respectively.
Functions: Thyroid hormones act on almost every cell in the body and are extremely important for growth and development. Like steroid hormones, thyroid hormones are highly lipophilic and therefore can easily cross cell membranes. After their entry into the cell, thyroid hormones bind to thyroid receptors. Unlike steroid receptors, thyroid receptors are nuclear receptors (steroid receptors are mostly present in cytoplasm). After binding to thyroid hormones, thyroid receptors may form a homodimer or heterodimer that eventually binds to hormone response elements in the DNA. This results in the expression of several genes responsible for growth and development. In most tissues (the brain, spleen, and testes are exceptions), thyroid hormones stimulate the metabolic rate by increasing the number and size of mitochondria, activating the synthesis of enzymes in the respiratory chain and increasing membrane sodium-potassium adenosine triphosphatase (ATPase) concentration and membrane sodium and potassium permeability.5 There is evidence that thyroid hormones also may bind to membrane receptors, causing nongenomic effects including stimulation of sugar transport, calcium ATPase activity, and increased sodium transport in muscle; the receptors for these effects have not been identified, however.5
Types of Hypothyroidism
There are three types of hypothyroidism: primary, secondary, and tertiary. Primary hypothyroidism, which is quite common, results from abnormalities of the thyroid gland. Approximately 95% of hypothyroidism cases are diagnosed as primary hypothyroidism.3 Secondary and tertiary hypothyroidism result from malfunctions of the pituitary and the hypothalamus, respectively. Tertiary hypothyroidism is sometimes not distinguished from secondary hypothyroidism.6 Secondary and tertiary hypothyroidism are sometimes referred to as central hypothyroidism.
Primary hypothyroidism is associated with decreased thyroid hormone and increased TSH levels. These abnormal levels may result from treatment for hyperthyroidism, iodine deficiency, thyroid cancer, or autoimmune disorders such as Hashimoto's thyroiditis (HT). In the U.S. and most of the Western world, where iodine deficiency is uncommon, HT is the predominant cause of hypothyroidism.7 In HT, autoantibodies are produced against thyroid peroxidase (TPO) and Tg.7 TPO oxidizes iodide to form Tg, which subsequently forms MIT upon further iodination (FIGURE 1). Autoantibodies against TPO destroy these macromolecules, resulting in MIT deficiency and overall thyroid hormone production.
Secondary hypothyroidism is characterized by decreased secretion of TSH, most commonly owing to pituitary failure.6 Sometimes, decreased TRH production by the hypothalamus results in decreased TSH production by the pituitary (tertiary hypothyroidism).8 In some patients, TSH is mildly elevated when levels of free thyroid hormones are normal (subclinical hypothyroidism). This situation indicates a possibility of the initial stage of thyroid failure.
Causes, Signs, and Symptoms
Hypothyroidism can be caused by several different conditions. In the U.S., the most common cause is destruction of the thyroid gland by the immune system (HT). Too little iodine in the diet is a rare cause of hypothyroidism in the U.S. Another exogenous cause is treatment for hyperthyroidism, including radiation therapy (RT), surgery, and drugs that alter the function and synthesis of thyroid hormones. Bromine, fluorine, and lithium are commonly used to treat hyperthyroidism. RT of the neck and lithium block iodide transport into the thyroid, and large doses of lithium inhibit the secretion of thyroid hormones.2
Since thyroid hormones play a major role in the body's overall metabolic activity, the signs and symptoms of hypothyroidism include a host of metabolic dysfunctions. Early signs and symptoms include cold intolerance, weakness, fatigue, and constipation; late signs and symptoms include decreased sense of smell and taste, hoarseness, and menstrual disorders. Complaints of muscle cramps, myalgia, fatigue, depression, and loss of energy are common.
Diagnosis
Measuring TSH and T4 concentrations in the blood is the most efficient way to diagnose hypothyroidism. Increased TSH concentrations and decreased thyroid hormone concentrations indicate primary hypothyroidism. Normal serum TSH and T4 values vary slightly between laboratories. An individual with no symptoms of abnormal thyroid function is represented by serum TSH in the range of 0.4 to 4.0 mU/L.9 Serum TSH values greater than this indicate primary hypothyroidism. Certain drugs (e.g., lithium, antithyroid medications, dopamine) may interfere with serum TSH. A typical normal serum T4 value is in the range of 4.5 to 11.2 mcg/dL. Lower-than-normal serum T4 indicates hypothyroidism. Certain drugs may interfere with serum T4; for example, birth control pills, clofibrate, estrogen, and the like are known to increase T4; lithium, propranolol, androgens, and so on are known to decrease T4. The cost-effectiveness of hypothyroidism screening is more favorable for women and the elderly population; this is strongly influenced by the cost of TSH measurement. The American Thyroid Association recommends routine screening in men and women aged 35 years and over.10
As mentioned previously, HT is an autoimmune disorder in which autoantibodies are produced against TPO and Tg; therefore, it is diagnosed by additional tests that identify the presence of these autoantibodies. The normal range for TPO autoantibodies is between 42 and 100 IU/mL, whereas the normal range for Tg autoantibodies is between 67 and 115 IU/mL. Values greater than these are considered positive.7
Treatment
Hypothyroidism is best treated with hormone replacement therapy (HRT). The desired clinical outcome of therapy is normal TSH levels. Levothyroxine, a synthetic product routinely used to treat hypothyroidism, is available under various brand names (e.g., Levothroid, Levoxyl, Synthroid, Unithroid).11 In adults, a daily dose of 1.6 mcg/kg of levothyroxine is recommended. In patients with ischemic heart disease, a low initial thyroxine dose is recommended (12.5-50 mcg/day) to avoid exacerbating angina.12 The half-life of levothyroxine is 1 week. Therefore, after 3 to 5 weeks, dose adjustment may be necessary. In most cases, symptoms of hypothyroidism start to improve within a week after initiation of HRT. Levothyroxine is an FDA pregnancy category A medication, which means that it is safe for use during pregnancy, although a dose increase may be necessary. Levothyroxine also is safe for use in breastfeeding women (the drug passes into breast milk, but is not expected to harm the nursing infant).
Recently, there has been much interest in examining the potential advantage of combined therapy of T3 and T4. Joffe et al reviewed nine such studies and concluded that the variability and limitations of the study designs made it difficult to determine any clinical recommendations; they further stated that additional studies are required to draw any conclusion about the clinical benefits of combination therapy over monotherapy.13 Currently, no concrete evidence supports an advantage for combination therapy.
Hypothyroid Conditions and Their Treatment
Gestational Hypothyroidism: This disorder occurs in approximately 2.5% of pregnant women.14 The fetus relies on the mother for thyroid hormones because development of the thyroid gland begins after the first trimester and is not complete until birth.15 Uncontrolled thyroid dysfunction in pregnancy is associated with adverse fetal complications because thyroid hormones are essential for the neurologic development and overall growth of the fetus.16 Untreated hypothyroidism during pregnancy may result in fetal distress in labor, preterm birth, low birthweight, congenital malformations, decreased neurologic function, and even fetal or perinatal death.
Maternal complications may also be serious and include anemia, postpartum hemorrhage, cardiac dysfunction, preeclampsia, and placental abruption.17 Therefore, it is essential to monitor and treat hypothyroidism during pregnancy effectively and appropriately. Recently, the American Association of Clinical Endocrinologists recommended that all women considering conception and all pregnant women in the first trimester be screened for thyroid dysfunction; however, the American College of Obstetricians and Gynecologists and the United States Preventive Services Task Force have not endorsed these recommendations.18
Maternal hypothyroidism generally necessitates levothyroxine intervention treatment. Several studies have confirmed that levothyroxine requirements in most women with existing hypothyroidism increase substantially during pregnancy.14 Prenatal vitamins should be carefully checked because they contain iron and calcium, both of which inhibit the absorption of levothyroxine.19
Congenital Hypothyroidism: One of the most common infantile endocrine disorders, congenital hypothyroidism has been detected at the rate of 1 in 3,000 to 4,000 live births worldwide.20 Routine screening for this disorder should be conducted when the infant is 3 to 4 days old. Screening is usually performed by measuring T4 and TSH levels. A T4 less than 6.5 mcg/dL and a TSH greater than 20 mIU/L indicate abnormal values necessitating treatment with levothyroxine at a dose of 10 to 15 mcg/kg/day.20 Treatment should be initiated as early as possible, since delayed treatment increases the risk of mental retardation.
Recent cohort analyses estimate that approximately 2% of thyroid dysgenesis cases are familial. The candidate genes associated with primary congenital hypothyroidism can be divided into two main groups: those causing thyroid dysgenesis and those associated with defects in iodide organification, leading to dyshormonogenesis. Genes associated with thyroid dysgenesis include TSH receptor, TITF-1, TITF-2, PAX8, and Gsa; those associated with dyshormonogenesis include TPO, Tg, sodium-iodide symporter, pendrin, and thyroid oxidase 2.21
Subclinical Hypothyroidism: This form of hypothyroidism is characterized by slight elevations of TSH and normal levels of T4 and T3. The risk of overt hypothyroidism in these patients is greater; 2% to 5% of those with signs of subclinical hypothyroidism eventually develop overt hypothyroidism.22 Levothyroxine treatment has been shown to be beneficial in patients exhibiting symptoms of hypothyroidism; on the other hand, levothyroxine treatment in asymptomatic patients with TSH levels between 4.5 and 10 mIU/L may not be beneficial.23
The advantages of early detection and treatment of subclinical hypothyroidism are not well established. Reports indicate an association between subclinical hypothyroidism and poor outcomes of pregnancy, as well as dyslipidemias, atherogenesis, and increased mortality in the long term. There are no published trials assessing the benefits of levothyroxine therapy in pregnant women with subclinical hypothyroidism, but the potential benefits of treatment for a thyroid hormone-deficient mother and fetus compared with the risks of treatment for a euthyroid mother and fetus justify its use.
Conclusion
Hypothyroidism is a chronic condition that afflicts millions of Americans. Proper and timely diagnosis, along with correct treatment, can go a long way toward reducing the morbidity and mortality associated with this disease state. Measurement of TSH and T4 blood concentrations remains the standard method of diagnosis. Levothyroxine is the current treatment of choice; when used correctly, it alleviates the signs and symptoms of hypothyroidism.
Acknowledgment: The authors acknowledge the assistance of Ms. Sue Annunnziata and Ms. Jenna Redmond in literature search.
REFERENCES
1. Pratt M, Levy EG. Hypothyroidism: An Essential Guide for the Newly Diagnosed. New York, NY: Marlowe & Co; 2003:xviii.
2. Dipiro JT, Talbert RL, Yee GC, et al, eds. Pharmacotherapy: A Pathophysiologic Approach. 7th ed. New York, NY: McGraw-Hill Medical; 2008:1254-1256.
3. AACE Thyroid Task Force. American Association of Clinical Endocrinologists medical guidelines for clinical practice for the evaluation and treatment of hyperthyroidism and hypothyroidism. Endocr Pract. 2002;8:458-469.
4. Vaidya B, Pearce SH. Management of hypothyroidism in adults. BMJ. 2008;337:a801.
5. Nussey SS, Whitehead SA. Endocrinology: An Integrated Approach. London, England: Taylor & Francis; 2001:71-111.
6. Brunton L, Lazo J, Parker K, eds. Goodman & Gilman's The Pharmacological Basis of Therapeutics.
7. Pearce EN, Farwell AP, Braverman LE. Thyroiditis. N Engl J Med. 2003;348:2646-2655.
8. Roberts C, Ladenson PW. Hypothyroidism. Lancet. 2004;363:793-803.
9. Harel L, Prais D, Uziel Y, et al. Increased prevalence of antithyroid antibodies and subclinical hypothyroidism in children with juvenile idiopathic arthritis. J Rheumatol. 2006;33:164-166.
10. Ladenson PW, Singer PA, Ain KB, et al. American Thyroid Association guidelines for detection of thyroid dysfunction. Arch Intern Med. 2000;160:1573-1575.
11. Singer PA, Cooper DS, Levy EG, et al. Treatment guidelines for patients with hyperthyroidism and hypothyroidism. JAMA. 1995;273:808-812.
12. Topliss DJ, Eastman CJ. 5: Diagnosis and management of hyperthyroidism and hypothyroidism. Med J Aust. 2004;180:186-193.
13. Joffe RT, Brimacombe M, Levitt AJ, Stagnaro-Green A. Treatment of clinical hypothyroidism with thyroxine and triiodothyronine: a literature review and metaanalysis. Psychosomatics. 2007;48:379-384.
14. Rashid M, Rashid MH. Obstetric management of thyroid disease. Obstet Gynecol Surv. 2007;62:680-688.
15. Smallridge RC, Ladenson PW. Hypothyroidism in pregnancy: consequences to neonatal health. J Clin Endocrinol Metab. 2001;86:2349-2353.
16. Casey BM, Dashe JS, Wells CE, et al. Subclinical hypothyroidism and pregnancy outcomes. Obstet Gynecol. 2005;105:239-245.
17. Poppe K, Glinoer D. Thyroid autoimmunity and hypothyroidism before and during pregnancy. Hum Reprod Update. 2003;9:149-161.
18. Wier FA, Farley CL. Clinical controversies in screening women for thyroid disorders during pregnancy. J Midwifery Womens Health. 2006;51:152-158.
19. Campbell NA, Reece JB. Biology. 7th ed. San Francisco, CA: Pearson Education; 2005:953-955.
20. Jain V, Agarwal R, Deorari AK, Paul VK. Congenital hypothyroidism. Indian J Pediatr. 2008;75:363-367.
21. Park SM, Chatterjee VK. Genetics of congenital hypothyroidism. J Med Genet. 2005;42:379-389.
22. Surks MI, Ortiz E, Daniels GH, et al. Subclinical thyroid disease: scientific review and guidelines for diagnosis and management. JAMA. 2004;291:228-238.
23. Ibay AD, Bascelli LM, Nashelsky J. Management of subclinical hypothyroidism. Am Fam Physician. 2005;71:1763-1764. 11th ed. New York, NY: McGraw-Hill Professional; 2005:1511-1527.






