US Pharm. 2009;34(11)(Oncology suppl):3-12.
ABSTRACT: The National Cancer Institute reported that 10.8 million Americans were alive in 2004 with a documented history of cancer, either actively being treated for cancer or in a state of remission.1 In 2008 alone, it was estimated that 1.4 million individuals were newly diagnosed with some form of cancer. This figure excluded carcinoma in situ, of any site other than the bladder, and basal and squamous cell skin cancers.1 Pain associated with cancer and its treatment is one of the principal concerns and fears amongst individuals afflicted with cancer and their families.2 The goals of pain management are very much patient specific; treatment, therefore, must be individualized accordingly.
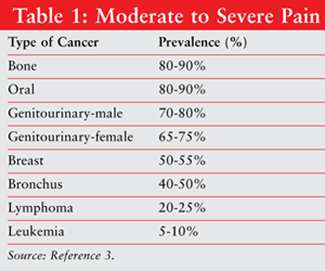
Thirty percent of patients complain of pain at the time of their cancer diagnosis, and between 65% and 85% of patients experience pain as their disease progresses.3 In addition, it is suggested that one-third to one-fourth of patients with cancer undergoing treatment and three-quarters of patients with advanced disease experience pain.4 Pain symptoms are highly variable and individualized, but some generalizations exist regarding certain types of cancer and their associated pain intensity (TABLE 1).3 Bone pain is associated with the highest prevalence and leukemia with the lowest prevalence of moderate to severe pain.
Eighty-five to 95% of cancer patients’ pain symptoms can be adequately managed when medication is appropriately selected and optimized; however, despite the fact that pain can be effectively managed, it often is not.5 Deandrea et al determined that nearly one in two patients with cancer pain may actually be undertreated.6 Deandrea et al showed that 43% of cancer patients had a negative Pain Management Index, which estimated the adequacy of pain control by comparing the patient’s reported level of pain and the appropriateness of analgesic therapy.5 The problem of ineffective pain management is multifactorial and includes personal biases, miscommunication, and lack of information and education of health care providers, patients, caregivers, and society in general.2 The above information is sufficiently compelling and should provide the impetus for pharmacists and other health care professionals to get engaged and advocate appropriate management and treatment of cancer-related pain.
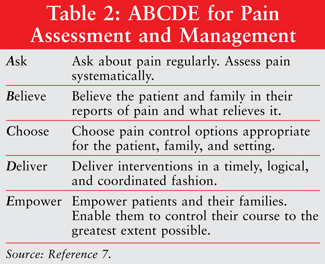
The patient with cancer experiencing pain requires ongoing comprehensive evaluations, histories, physical examinations, assessments, and reassessments of their pain.4,7,8 The location of their pain, the characteristics of their pain—both intensity and quality—and the patient’s response to treatment must be completely assessed and documented.4,7,8 The health care professional must thoroughly understand the source of the underlying malignancy, any metastases if applicable, as well as any pertinent laboratory findings as they become available.8 All of these factors may affect the drug-selection process. The Agency for Health Care Policy and Research developed the ABCDE pain assessment and management mnemonic (TABLE 2), which health care professionals have found to be very useful in their provision of optimal patient care and selection of drug therapies.7 As part of the assessment and management process, the quality of the patient’s pain should ideally be assessed at baseline and following therapy changes.
The most valuable measure of pain intensity is the patient’s self report.3 The mnemonic PQRST has been developed to assist health care professionals in their query of the patient and patient’s family so that pertinent contributory factors are not unintentionally omitted or overlooked in the assessment and management process (TABLE 3).9,10

Pain symptoms are very subjective and are often difficult to communicate appropriately to health care professionals. To improve communications between patients and health care providers, the medical community tries to quantify one’s complaint of pain. It is generally accepted that a pain score of 0 is equivalent to no pain, and a pain score of 10 is equivalent to severe pain. When a patient is unable to quantify pain in terms of a numeric system or rating scale, pictures of faces experiencing varying degrees of pain may be useful. Many tools and aids are available to assist health care professionals and patients in communicating a description of the intensity of their pain including 0-10 numeric, categorical, visual analog, and faces rating scales.11,12
Pain assessment becomes more challenging and often difficult with older patients, nonverbal patients, non-English-speaking persons, and patients with varying levels of cognitive impairment. It is mistakenly believed that older adults are less sensitive to pain, more tolerant to pain, and less tolerant to opioids, resulting in undertreatment of cancer pain.12,13 In addition, older individuals may be more reluctant to complain of pain specifically but feel more comfortable acknowledging discomfort, hurting, or aching symptoms.12 Thus, the manner in which pain-related questions are presented to the older patient becomes vitally important. Additionally, ethnic and cultural differences may impede treatment of cancer pain. Some cultures give socially acceptable responses when questioned about their pain rather than provide an accurate assessment of their pain.12 Therefore, clinicians must be aware that these differences exist and attempt to determine alternative ways to communicate and respond to the needs of these patients.12 Pain-related behaviors are often blunted in the presence of cognitive impairment; providers should look for subtle behavior and physiologic changes.11 The Pain Assessment in Advanced Dementia (PAINAD) scale may be useful under these circumstances (TABLE 4). This scale evaluates breathing independent of vocalization, negative vocalization, facial expression, body language, and consolability.14 Scores range from 0 to 10 with a higher score indicative of more severe pain.

There are generally three broad categories of cancer-related pain: somatic pain, visceral pain, and neuropathic pain (TABLE 5).8 Somatic pain is generally localized pain; patients are usually able to describe this type of pain well to their health care providers as a sharp, aching, or throbbing pain.15,16 It is often a direct result of the tumor itself or may result from treatments such as surgery or chemotherapy.8 Bony pain as well as mucositis are examples of somatic pain.8,16 Nonsteroidal anti-inflammatory drugs (NSAIDs) may be especially effective with this type of pain. Oral opioids may be beneficial as well.16 In the case of mucositis resulting from chemotherapy, viscous lidocaine dosed around the clock or before meals is effective. In addition, providers should evaluate the patient’s mouth for herpetic or fungal infections and offer appropriate treatment.8 Visceral nociceptors are sensitive to stretch; stretch receptors can be found in the muscular layers of the hollow viscera between the muscularis mucosa and submucosa, and in the serosa of solid organs.15

Visceral pain is not well localized. It may be described as deep, dull, achy, gnawing, burning, squeezing, cramping, or a sensation of pressure.8,15 Opioid therapy is effective management for this type of cancer-related pain. Surgery to remove the cause of the pain as well as autonomic nerve blocks should also be considered.8,16
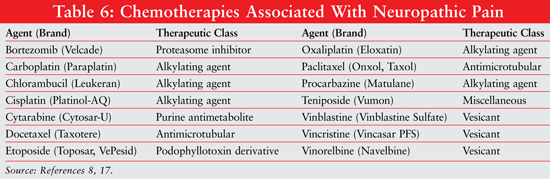
Neuropathic pain is a consequence of neural injury and may directly result from solid tumors impinging on nerve fibers.8,16 Neuropathic pain can be present in any type of cancer but appears to be more prevalent in head and neck cancer and pancreatic cancer, as well as cancers involving the spinal cord.8 Some tumors invade the spinal cord itself while other cancers result in spinal cord bone metastases, causing cord compression.8 Neuropathic pain can also result from an acute infection, myelosuppression, other comorbidities, and chemotherapy.8 Several chemotherapy agents are known to cause neuropathic pain (TABLE 6).8,17 Patients must therefore be appropriately counseled and educated to minimize the incidence and extent of neuropathic pain. Neuropathic pain appears to respond well to tricyclic antidepressants or anticonvulsants. In general, neuropathic pain does not respond well to opioids other than methadone due to its N-methyl-D-aspartate receptor antagonist properties.8,16 Spinal cord compression should be treated as a medical emergency, and the treatment of choice is rapid administration of a corticosteroid such as dexamethasone. Corticosteroids decrease edema associated with the tumor and surrounding tissue, thus alleviating pain symptoms.
As discussed above, opioids can be very useful in the treatment of both somatic and visceral cancer pain, particularly as the likelihood of pain and the severity of pain increases with advanced disease.13 Several factors must be considered when selecting an opioid. These factors include pain intensity, pharmacokinetic and formulary considerations, response to previous opioid therapy, other comorbidities, and route of administration.18 If the patient’s cancer pain is continuous, then treatment should also be continuous and opioid therapy should be dosed around the clock rather than on an as-needed basis in order to maintain an appropriate level of analgesia throughout the day.18 Occasionally, pain symptoms “break through” the regular scheduled opioid regimen requiring a rescue dose, which is generally 5% to 15% of the 24-hour dose.18 Depending upon the number of rescue doses needed and the circumstances involved, a patient’s maintenance opioid regimen may need to be titrated. Doses are generally titrated by 33% to 50% of the pre-existing dose.18
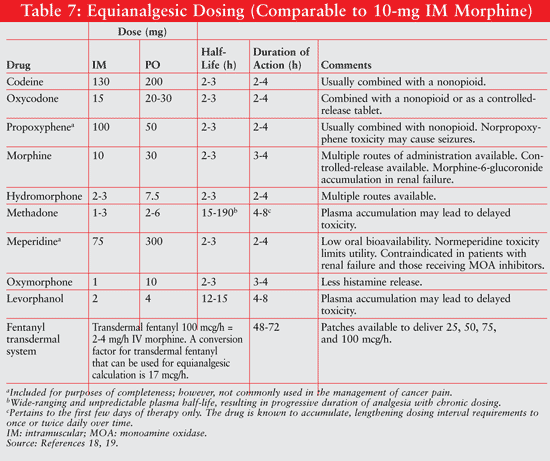
Clinicians may need to switch opioids to improve pain control, reduce opioid toxicity or side effects, improve dosing convenience or change routes of administration, in which case an equianalgesic table, which is a listing of opioids at doses that produce comparable amounts of analgesia, would be useful (TABLE 7).19 The calculation can be set up as a ratio or as a proportion. One would identify the equianalgesic dose of the current drug from the equianalgesic table, over the actual 24-hour dose of the current drug equated to the equianalgesic dose of the new drug, from the equianalgesic table, over the 24-hour dose of the new drug. Solve this equation for the 24-hour dose of the new drug. Divide this number by the appropriate dosing interval for the new drug to obtain your new scheduled dose.19 Depending upon the circumstances and the amount of pain relief on the current drug, the dose of the new drug may or may not need to be decreased to account for cross tolerance between agents; however, some patients may initially require only one-third to one-half of the calculated equianalgesic dose, particularly if pain was optimally controlled on the current regimen.19 It should be emphasized that there are many versions of equianalgesic tables available with their own subtle nuances. These tables are intended to serve only as a guide or tool and are not intended to replace or substitute sound clinical judgment and patient-specific dosing.
An opioid regimen may also need to be changed if a patient reports an adverse reaction to a particular agent. In general, true allergic reactions to opioids are rare. In most cases, the reaction is a side effect rather than an allergic reaction and will generally resolve on its own.20 Depending upon the severity of the reaction, the drug may or may not need to be changed. If the reaction is limited to flushing, itching, sweating, hives, or mild hypotension, the opioid can typically be continued with an antihistamine or dose reduction. If the reaction is more serious or more extensive involving rash, severe hypotension, bronchospasm, or angioedema, it is prudent to change opioid classes.21 There are three classes of opioids, including phenanthrenes (both hydroxylated [morphine, codeine] and nonhydroxylated types [buprenorphine, butorphanol, hydrocodone, hydromorphone, levorphanol, oxycodone, oxymorphone]); phenylpiperidines (alfentanil, fentanyl, sufentanil); and diphenylheptanes (methadone, propoxyphene).21 Understanding which opioid class a particular drug belongs to is important and will assist the clinician should one have to switch opioids due to reports of an allergic reaction, particularly a serious reaction. For example, if angioedema was reported following administration of morphine sulfate, one should avoid all phenanthrenes, both hydroxylated and nonhydroxylated. Opioids belonging to either the phenylpiperidines or diphenylheptanes class may therefore be more suitable options for this patient because the chemical structure of the opioids within these two classes are sufficiently different to minimize further risk of angioedema.
An additional rate-limiting step in the use of opioids for management of cancer pain is constipation. Constipation is the most common adverse reaction reported and can be managed with the concomitant administration of a softening agent and a cathartic.18 Constipation will not self resolve and is best managed empirically at the onset of treatment as a prophylactic measure rather than waiting for the complaint to present as activation of mu opioid receptors in the colon inhibits peristalsis.22 Opioid-induced sedation and opioid-induced delirium present commonly and can be managed appropriately in many cases with a dose reduction without compromising pain control (TABLES 8, 9).18


Pharmacists are integral to the successful management of pain in individuals diagnosed with cancer. Though the World Health Organization does offer providers some very broad generalities and suggestions with respect to the treatment of pain based upon the severity of the patient’s symptoms, it is the pharmacist whose knowledge and understanding of drug therapies, drug interactions, pharmacokinetics, and pharmacodynamics who can facilitate a treatment plan unique to the circumstances and needs of the patient.4 Their thorough and detailed assessment skills combined with their extensive knowledge of drug therapy make them a valued member of the interdisciplinary management team. In addition, their ability to communicate effectively with both patients and health care professionals alike enhances their ability to make appropriate and safe drug recommendations.
REFERENCES
1. American Cancer Society. Statistics for 2008. Cancer Facts & Figures 2008.
www.cancer.org/downloads/STT/2008CAFFfinalsecured.pdf.
2. Jemal A, Murray T, Samuels A, et al. Cancer statistics. CA Cancer J Clin. 2003;53(1):5-26.
3. Lipman AG, ed. Pain Management for Primary Care Clinicians. Bethesda, MD: American Society of Health-System Pharmacists, Inc; 2004:141-151.
4. Portenoy R. Cancer pain management. Semin Oncol. 1993;20(2):19-35.
5. Levy MH. Pharmacologic treatment of cancer pain. N Engl J Med. 1996;335:1124-1132.
6. Deandrea S, Montanari M, Moja L, et al. Prevalence of undertreatment in cancer pain. A review of published literature. Ann Oncol. 2008;19(12):1985-1991.
7. Managment of Cancer Pain. Clinical Practice Guideline No. 9. AHCPR Publication No. 94-0592. Rockville, MD: Agency for Healthcare Policy and Research; March 1994.
8. Brundage DM. Cancer pain management in adults: a pharmacist’s perspective. J Pharmacy Practice. 2003;16(4):284-290.
9. DiPiro JT, Talbert RL, Yee GC, eds. Pharmacotherapy: A Pathophysiologic Approach. 6th ed. New York, NY: The McGraw-Hill Companies, Inc; 2005:1091.
10. Twycross RG. Pain and analgesics. Curr Med Res Opin. 1978;5:497-505.
11. Lipman AG, ed. Pain Management for Primary Care Clinicians. Bethesda, MD: American Society of Health-System Pharmacists, Inc; 2004:27-41.
12. Miaskowski C, Cleary J, Burney R, et al. Guideline for the Management of Cancer Pain in Adults and Children. 3rd ed. Glenview, IL: American Pain Society; 2005:19-38.
13. Managment of Cancer Pain. Evidence Report/Technology Assessment No. 35. AHCPR Publication No. 02-E002. Rockville, MD: Agency for Healthcare Policy and Research; October 2001.
14. Warden V, Hurley AC, Volicer L. Development and psychometric evaluation of the pain assessment in advanced dementia (PAINAD) scale. J Am Med Dir Assoc. 2003;4:9-15.
15. Boswell MV, Cole BE, eds. Weiner’s Pain Management: A Practical Guide for Clinicians. 7th ed. Boca Raton, FL: CRC Press; 2006:371-379.
16. Chang HM. Cancer pain management. Med Clin North Am. 1999;83(3):711-736.
17. Lacy CRF, Armstrong LL, Goldman MP, et al. Drug Information Handbook. 13th ed. Hudson, OH:
Lexi-Comp, Inc; 2005.
18. Cherny NI. The management of cancer pain. Ca Cancer J Clin. 2000;50(2):70-116.
19. Gordon DB, Stevenson KK, Griffie J, et al. Opioid equianalgesic calculations. J Palliat Med. 1999;2(2):209-218.
20. Gilbar PJ, Ridge AM. Inappropriate labeling of patients as opioid allergic. J Oncol Pharm Practice. 2004;10:177-181.
21. Cupp M. Analgesic options for patients with allergic–type opioid reactions. Pharmacist’s Letter/Prescriber’s Letter. 2006;22(220201).
22. Lipman AG, ed. Pain Management for Primary Care Clinicians. Bethesda, MD: American Society of Health-System Pharmacists, Inc; 2004:67.
To comment on this article, contact rdavidson@jobson.com.





