The last several decades have seen a significant rise in the use of opioid medications. A recent study demonstrated an equally significant increase in the number of drug overdose deaths, with opioid medications suspected in 14,800 deaths in 2008 and representing approximately 73% of all prescription drug overdose deaths.1 There is also a likely increase in the number of patients developing opioid dependence.
SUBSTANCE ABUSE, ADDICTION, AND DEPENDENCE
Substance abuse, as described by the Diagnostic and Statistical Manual of Mental Disorders (Fourth Edition, Text Revision) (DSM-IV), is a maladaptive pattern of substance use that leads to clinically significant impairment or distress. DSM-IV criteria for substance abuse appear in TABLE 1.2 Addiction is characterized by compulsive drug use and drug seeking despite adverse social, psychological, and/or physical consequences.3 Physical dependence is a state that develops during regular drug use in which a withdrawal syndrome results upon drug cessation.3 Dependence differs from addiction and substance abuse in that it occurs as a result of normal adaptations to chronic drug exposure.

OPIOID DEPENDENCE
Over the last two decades, there has been a steady increase in the total number of opioid prescriptions. From 1991 to 2010 the number of opioid prescriptions increased sixfold, from 30 million to 180 million prescriptions.4 Concurrent with this growth in opioid prescriptions has been an increase in diversion and nonmedical opioid use. In 2009, an estimated 5.3 million persons were taking pain relievers for nonmedical uses.4 In 2010, nearly 5% of the U.S. population aged 12 years and older was taking an opioid pain reliever for nonmedical purposes.1
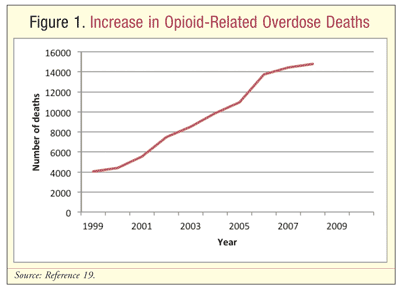
In the U.S. in 2007, nearly 100 people died each day from a drug overdose.1 According to the CDC, death rates from opioid overdoses have more than tripled since 1999.5 Increased opioid use has contributed to the increase in overdose-related deaths (FIGURE 1). Opioid pain relievers currently account for more overdose deaths than heroin and cocaine combined.1
Not everyone receiving opioids will develop an addiction; however, certain biological and environmental risk factors increase the likelihood of substance abuse. Males are 1.5 times more likely to become dependent on opioids.2 Psychological conditions such as depression, anxiety, attention-deficit/hyperactivity disorder or attention-deficit disorder, and posttraumatic stress disorder confer an increased risk of addiction, as the sufferer may use drugs to cope with the condition.6 Peer pressure and lack of family involvement are environmental risk factors that may predispose a person to substance abuse.6
Opioids are safe and effective for pain management when taken as prescribed. However, these drugs can cause severe adverse effects—including drowsiness, slurred speech, confusion, respiratory depression, and even death—in cases of abuse.2 Opioid withdrawal symptoms may occur following dose reduction in chronic users or upon abrupt cessation. The onset of withdrawal symptoms varies depending upon the specific opioid used and occurs from hours to days after cessation.2,7 For example, with heroin, withdrawal symptoms can occur within 1 to 3 hours, while with methadone they may not appear for 3 to 5 days. Withdrawal symptoms are listed in TABLE 2.

OPIOID PHARMACOLOGY
An important concept in opioid dependence is the pharmacology associated with these medications. Three major types of opioid receptors are involved in analgesia: mu, delta, and kappa receptors.8 Activation of mu receptors produces analgesia, sedation, euphoria, respiratory depression, constipation, and physical dependence. Activation of kappa receptors produces mild analgesia and less respiratory depression; delta receptor activation may induce analgesia with fewer adverse effects. Opioids have varying effects on these receptors. Agonist agents produce analgesia, whereas antagonists attenuate analgesia.
Opioid classification is determined by receptor activity. Classes include full agonists, partial agonists, agonist-antagonists, and antagonists.8 Full agonists (morphine, fentanyl, oxycodone, hydrocodone, methadone) activate mu receptors, producing analgesia. Buprenorphine—a partial agonist—has a strong affinity for mu receptors, but produces mild analgesia that is accompanied by an analgesic ceiling effect. The mu receptors’ strong affinity may displace other mu receptor agonists. Agonist-antagonists (pentazocine, butorphanol, nalbuphine) typically act as antagonists on mu receptors and as agonists on kappa receptors, leading to milder analgesic effects with fewer adverse effects. Caution should be exercised in the use of partial agonists or agonist-antagonists in patients currently receiving full agonists because opioid withdrawal syndrome could be precipitated. The antagonists naloxone and naltrexone block mu and kappa receptor activation, preventing analgesia; they are used mostly as reversal agents.
TREATMENT
The treatment of opioid dependence includes several modalities. Office-based or specialized treatment centers are the core component of the treatment of opioid dependence.9-11 Long-term treatment may include maintenance therapy with opioid agonists or antagonists. Additional treatment options include abstinence-based treatment approaches with nonpharmacologic adjunctive treatment (e.g., Narcotics Anonymous). The following are pharmacologic agents used for treatment (TABLE 3).
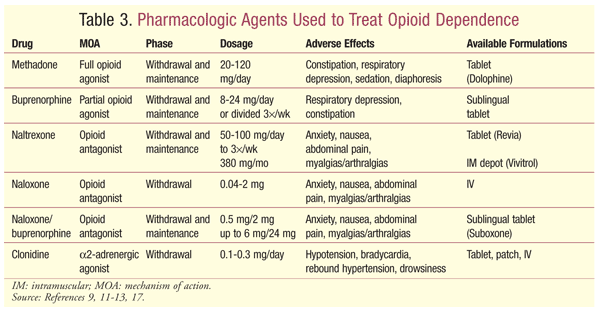
Methadone: A synthetically derived opioid, methadone is listed as a Schedule II medication by the Controlled Substances Act. Methadone is a mu receptor agonist with additional N-methyl-D-aspartate receptor antagonism.8 Its half-life ranges from 8 to 59 hours with a much shorter analgesic effect, necessitating thrice-daily dosing for analgesia. Dosing for maintenance therapy is typically once daily. Methadone’s long half-life allows it to stay bound to mu receptors, preventing withdrawal symptoms while keeping additional opioids from binding to the receptor, thereby blocking euphoria. Methadone is metabolized through the CYP450 system. Drug interactions should be anticipated and evaluated appropriately.12
Buprenorphine: Buprenorphine is a partial mu receptor agonist.8 It has a strong mu receptor affinity, but low analgesic potential because of its ceiling or plateau effect. This effect also may render buprenorphine overdoses safer compared with methadone. The role of buprenorphine in maintenance therapy is due to its strong mu receptor affinity, which ultimately blocks additional opioids from binding to the receptor. Buprenorphine produces sufficient agonist effects to allow discontinuation of the abused opioids without causing withdrawal effects. If buprenorphine is given to a patient while opioids are coupled to mu receptors, it may displace other opioids, potentially leading to withdrawal symptoms. Because of this potential, buprenorphine or buprenorphine combination therapy should be initiated only after opioid withdrawal symptoms become apparent or under direct medical supervision.
Naloxone/Naltrexone: Naloxone and naltrexone are competitive antagonists with high affinity to mu and kappa receptors.8 Use of these agents leads to a rapid displacement of opioids from receptors, causing withdrawal symptoms; therefore, the patient should be opioid free at the time of initiation to avoid inducing acute withdrawal symptoms. These medications are used as abuse deterrents. Naloxone has low oral bioavailability and is rapidly inactivated because of first-pass metabolism. It typically is combined with buprenorphine (Suboxone) to prevent abuse through crushing and parenteral injection. Parenteral administration of naloxone has a rapid onset with full receptor reversal, and the drug is used primarily for acute opioid reversal in overdose settings. Naltrexone has higher bioavailability, permitting oral use, and is available as an intramuscular depot formulation.
Clonidine: Clonidine is a centrally acting alpha-2 agonist that suppresses noradrenergic activity. Opioid use also produces noradrenergic inhibition, and an increase in noradrenergic activity is seen during phases of opioid withdrawal. Clonidine replaces noradrenergic inhibition previously provided by opioids while attenuating withdrawal symptoms.9,13 Clonidine may be used for acute opioid withdrawal and the transition to long-term maintenance therapy. Hypotension and sedation may limit the use of clonidine.
Levo-Alpha-Acetyl-Methadol: This agent, an opioid antagonist, was removed from the U.S. market in 2003 based on reports of life-threatening ventricular rhythm disorders.10
History and Regulations
Methadone replacement, the first treatment for opioid dependence, has been available since the 1960s.14 Concerns regarding methadone diversion by both patients and physicians led to the Narcotic Addiction Treatment Act (NATA) of 1974. NATA authorized the Drug Enforcement Administration (DEA) to register with the federal government any physician wishing to prescribe methadone for detoxification or maintenance therapy.15 Although NATA helped control methadone diversion, it ultimately limited treatment options for many individuals.
In the U.S., the treatment of opioid dependence with opioid medications is governed by Federal Regulation 42 CFR Part 8.16 Methadone for opioid dependence is restricted to certified opioid treatment programs (OTPs) or licensed inpatient hospital detoxification units. Methadone may be used to treat pain; however, a prescriber may not use the 40-mg tablets for this indication. The 40-mg formulation is indicated only for opioid dependence detoxification and maintenance therapy and is distributed only to hospitals and facilities authorized to treat opioid dependence.10 To lessen the incidence of abuse by IV injection, only the oral formulation of methadone is permitted for use in opioid dependence. Practitioners wishing to use methadone for maintenance and detoxification must obtain a separate DEA registration number. In addition to federal regulations, individual state laws may mandate other restrictions or requirements for methadone treatment facilities. Patients are generally required to pick up doses of methadone at the OTP on a daily basis, although under some circumstances (e.g., patient compliance) small quantities of take-home methadone doses may be dispensed.
In May 2001, the Department of Health and Human Services (DHHS) announced a new system for methadone maintenance-treatment monitoring and regulation. Oversight of these programs shifted from the FDA to the Substance Abuse and Mental Health Services Administration’s (SAMHSA) Center for Substance Abuse Treatment (CSAT). SAMHSA, a branch of the DHHS, was established in 1992. Congress directed SAMHSA to target substance abuse and mental health disorders in those people most in need. Under this system, programs must be accredited by CSAT-approved accreditation bodies and the designated state authority.17 This system is based upon best-practice guidelines and permits less variability in the standard of care provided.
The restriction of methadone to OTPs and specialized treatment centers may cause patients not wishing others to know that they attend a methadone clinic for maintenance to limit their treatment. This concern led to the development of the Drug Addiction Treatment Act (DATA) of 2000, which was specifically designed to provide alternative treatment options for opioid dependence. DATA allows physicians who meet certain qualifications to prescribe Schedule III, IV, and V medications (primarily buprenorphine) approved for the treatment of opioid dependence to patients needing treatment for their addiction. Physicians must also indicate that they have the ability to refer patients to appropriate counseling if needed. Originally, DATA permitted a physician to treat up to 30 patients at any given time, but this changed in 2007, so that after 1 year a physician may request permission to treat up to 100 patients.17 Physicians who meet qualifications to prescribe buprenorphine receive a special DEA number beginning with an X. For the medication to be dispensed by a pharmacy, this number must appear on every prescription.10
Several regulations related to the prescribing and administration of medications used to treat opioid dependence have developed over time. Practitioners who are not specifically registered to conduct a narcotics treatment program may administer—but not prescribe—narcotics to a patient in acute withdrawal while making necessary referrals.18 Unlicensed practitioners may provide methadone for dependence or withdrawal for a maximum of 3 days while working on referral to a licensed OTP.
Treatment Phases
Methadone and buprenorphine are the most common maintenance therapy options. With methadone, an initial dose is given and further dosing is titrated based upon withdrawal symptoms experienced 2 to 4 hours after administration. Methadone maintenance treatment typically lasts 12 months or up to 2 years, and doses generally range from 80 to 120 mg daily.17
Maintenance therapy with buprenorphine is classified into three phases: induction, stabilization, and maintenance. The goal of the induction phase is to find the smallest dose of buprenorphine that allows the patient to discontinue opioids without experiencing withdrawal symptoms, usually over 3 to 5 days. In general, buprenorphine should be initiated 12 to 24 hours after short-acting opioids and 24 to 48 hours after long-acting opioids. It is recommended that the patient be observed during initial dosing since buprenorphine may induce withdrawal symptoms. The stabilization phase begins when a patient no longer experiences withdrawal symptoms. Dose adjustments may be necessary to prevent further cravings and minimize side effects. Most patients remain in this phase for 1 to 2 months, and the need for counseling and ancillary services should be assessed. The final treatment phase, maintenance, is also the longest phase and may last from months to years, depending upon the patient. The goal of maintenance is to identify social and family issues associated with the patient’s use of opioids. The physician should continue patient monitoring during this phase while devising a plan in anticipation of relapse.17
Office-Based Approaches
DATA, as previously described, enables physicians to offer office-based treatment of opioid dependence using buprenorphine as monotherapy or combined therapy. This model involves coordination of care services in a physician’s office, versus the traditional OTP at a specific treatment facility and with limited physician involvement. Office-based treatment using buprenorphine increases treatment availability for patients while allowing services to be tailored to the patient’s needs. It also preserves the patient’s autonomy so that it is not necessary to visit a methadone clinic. In determining whether this treatment approach is best for a given patient, the practitioner must take into consideration patient-specific characteristics, physician training and experience, and regulatory issues. Patients best suited for office-based treatment include those who have been enrolled in a maintenance program for 1 to 5 years, are clinically stable with no evidence of illicit drug use, have no untreated psychiatric conditions, have stable financial support, are compliant with treatment, and have no history of relapse.11
CONCLUSION
The use of opioid medications has been on the rise over the last several decades. There is also a likely increase in the number of patients developing opioid dependence. For these reasons, it is important for pharmacists to be educated on this class of medications and fully understand their proper use and administration. When used for maintenance treatment of opioid dependence, methadone may be prescribed and administered only in an OTP accredited by SAMHSA-CSAT. The 40-mg formulation of methadone is indicated only for the detoxification and maintenance treatment of opioid dependence. For buprenorphine to be dispensed by the pharmacy, all prescriptions must include the physician’s special DEA number beginning with the letter X. Knowing these important regulatory points will help pharmacists ensure that opioid medications are being prescribed and utilized appropriately.
REFERENCES
1. CDC. Vital signs: overdoses of prescription opioid pain relievers—United States, 1999–2008. MMWR. 2011;60:1487-1492.
2. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders (Fourth Edition, Text Revision). Washington, DC: American Psychiatric Association; 2000:199-273.
3. National Institute on Drug Abuse. Prescription Drugs: Abuse and Addiction.
NIH Pub No 11-4881. Revised October 2011.
www.drugabuse.gov/sites/default/files/rrprescription.pdf. Accessed
December 30, 2011.
4. National Institute on Drug Abuse. Prescription Drug Abuse—May 2011: A Research Update from the National Institute on Drug Abuse. Washington, DC: Public Information and Liaison Branch: Office of Science Policy and Communications; 2011.
5. Warner M, Chen LH, Makuc DM. Increase in fatal poisonings
involving opioid analgesics in the United States, 1999-2006.
www.cdc.gov/nchs/data/databriefs/db22.htm. Accessed December 30, 2011.
6. National Institute on Drug Abuse. Drugs, Brains and Behavior: The Science of Addiction. NIH Pub No 10-5605. Revised August 2010. www.drugabuse.gov/sites/default/files/sciofaddiction.pdf. Accessed January 12, 2012.
7. Doering PL. Substance-related disorders: overview and depressants,
stimulants, and hallucinogens. In: DiPiro JT, Talbert RL, Yee GC, et
al, eds. Pharmacotherapy: A Pathophysiologic Approach. 8th ed. New York, NY: McGraw-Hill Medical; 2011:1118.
8. Trescot AM, Boswell MV, Atluri SL, et al. Opioid guidelines in the management of chronic non-cancer pain. Pain Physician. 2006;9:1-39.
9. Amass L, Ling W, Freese TE, et al. Bringing buprenorphine-naloxone
detoxification to community treatment providers: the NIDA Clinical
Trials Network field experience. Am J Addict. 2004;13:S42-S66.
10. Office of Diversion Control. Section 1306.07: administering or
dispensing of narcotic drugs.
www.deadiversion.usdoj.gov/21cfr/cfr/1306/1306_07.htm. Updated June
2005. Accessed January 3, 2012.
11. Fiellin DA, O’Connor PG. Office-based treatment of opioid-dependent patients. N Engl J Med. 2002;347:817-823.
12. Dolophine (methadone hydrochloride) prescribing information. Columbus, OH: Roxane Laboratories, Inc; October 2006.
13. Gold MS, Pottash AC, Sweeney DR, Kleber HD. Opiate withdrawal
using clonidine. A safe, effective, and rapid nonopiate treatment. JAMA. 1980;243:343-346.
14. Gouldin WM, Kennedy DT, Small RE. American Pain Society. Methadone: history and recommendations for use in analgesia. APS Bulletin. 2000;10. www.ampainsoc.org/pub/bulletin/sep00/upda1.htm. Accessed January 8, 2012.
15. DuPont RL, Dormer RA, Nightingale SL. Treatment of narcotics addiction with narcotic drugs. JAMA. 1976;235:1565-1566.
16. DEA announces restrictions on distribution of methadone. AAFP News Now.
www.aafp.org/online/en/home/publications/news/news-now/clinical-care-research/20071219methadone.html.
Accessed January 20, 2012.
17. Code of Federal Regulations Title 42: Public Health. Part
8—certification of opioid treatment programs.
http://ecfr.gpoaccess.gov/cgi/t/text/text-idx?c=ecfr&sid=771a576d0abe318644b27139a55336a1&rgn=div5&view=text&node=42:1.0.1.1.10&idno=42.
Accessed January 31, 2012.
18. Nicholls L, Bragaw L, Ruetsch C. Opioid dependence treatment and guidelines. J Manag Care Pharm. 2010;16(1 suppl B):S14-S21.
19. Data brief 81: Drug poisoning deaths in the United States,
1980–2008. CDC/NCHS, National Vital Statistics System.
www.cdc.gov/nchs/data/databriefs/db81_tables.pdf#3. Accessed January 26,
2012.
To comment on this article, contact rdavidson@uspharmacist.com.





