US Pharm. 2013;38(1):HS-6-HS-8.
Prostate cancer is a heterogeneous disease that is often slow growing. However, if the cancer is advanced or left untreated in the early stages, the cancer cells may metastasize from the prostate to other parts of the body, particularly the bones and lymph nodes.1 The symptoms of prostate cancer closely resemble benign prostatic hyperplasia. These symptoms include frequent urination, nocturia (increased urination at night), difficulty starting and maintaining a steady stream of urine, hematuria (blood in the urine), and dysuria (painful urination). Approximately one-third of patients diagnosed with prostate cancer have one or more such symptoms, while two-thirds have no symptoms.2
Epidemiology
According to the American Cancer Society (ACS), prostate cancer is the second most common type of cancer found in American men and the second leading cause of cancer death in men.3 One in every 6 men will be diagnosed with prostate cancer during their lifetime, and 1 in 36 will die from the disease. This year, more than 241,700 new cases of prostate cancer will be diagnosed.4 Studies demonstrate that African Americans are at a higher risk of developing prostate cancer.5
Traditional Diagnosis
Once men reach age 50 years, most physicians will order a prostate-specific antigen (PSA) level and a digital rectal examination (DRE). However, in men who are at a higher risk, screenings should begin at the age of 45.5 Traditionally, DRE has been recommended for the detection of prostate cancer because of low cost, ease of performance, and safety. Though the specificity of the DRE is ≥85%, its use as a single screening method has minimal effect on the prevention of the disease. As such, the combination of DRE and PSA is a better method in detecting prostate cancer than DRE alone.5
Since the institution of serum PSA as a screening method, much effort has been expended to optimize early detection of prostate cancer. The normal range of PSA is ≤4 ng/mL. If the PSA level is between 4 and 10 ng/mL, further testing involving invasive procedures may be needed to distinguish benign prostatic hyperplasia from prostate cancer.5 Although PSA levels conventionally have a greater specificity for prostate enlargement than cancer detection, each year in the United States more than one million men undergo biopsies as a result of PSA elevations, and most of these biopsies are negative. Additionally, repeat biopsies are ordered in approximately 30% of patients who are suspected of having prostate cancer.4 Controversy still exists regarding whether to repeat a prostate biopsy in patients with a previous negative biopsy.
Repeat Biopsy Concerns
Unnecessary repeat biopsies are an area of concern due to a high rate of complications and associated costs.6 Patients who undergo repeat biopsies are subject to serious complications such as septicemia, urinary infections, bladder perforation, reduced sexual activity, and severe pain.7 Moreover, research has demonstrated that men who undergo prostate biopsy experience a moderate or higher level of pain and report pain scores of 5 or more on a 0 to 10 scale.8
PCA3 Gene
As of late, the PCA3 gene has proven to be a vital marker in the detection of prostate cancer.9 The PCA3 gene produces a noncoding RNA that is only expressed in human prostate tissue and overexpressed in 95% of prostate cancer cells.10,11 Due to prostate-specific expression, PCA3 RNA is very useful as a tumor marker and has been proven to be more accurate than traditional PSA testing in patients who have undergone one or more previous negative prostate biopsies.
PROGENSA
On February 13, 2012, the FDA approved the PROGENSA PCA3 [prostate cancer gene 3] Assay—the first molecular test to assist in determining whether a repeat biopsy is required when used with other clinical information and laboratory data.12 The PROGENSA PCA3 Assay is approved for use in men who have had previous negative prostate biopsies. The assay is not indicated for use in men who have evidence of atypical small acinar proliferation on their last biopsy.13 The PROGENSA PCA3 Assay should be employed using the first-void urine. Once the first-void urine is collected, a portion of the urine sample is transferred to a tube containing urine transport medium, encouraging the lysis and stabilization of RNA. Instructions for use are found in TABLE 1.14 Candidates for the PCA3 assay are men who have experienced one or more previous negative biopsies and who are being considered for repeat biopsy by a urologist.15

PCA3 Score: This test utilizes the PCA3 score that is derived from the ratio of PCA3 RNA to PSA RNA and provides the degree of probability a prostate biopsy will result in prostate cancer. The score may range from less than 5 to greater than 100. A PCA3 score of less than 25 is associated with a decreased likelihood of a positive biopsy. Though the PCA3 score does not rule out cancer, when used in conjunction with PSA, DRE, age, and prostate size, it allows a better estimate to be made regarding the risk of biopsy-detectable prostate cancer.16
Efficacy: A clinical trial was conducted to show whether the PCA3 Assay was helpful in determining prostate cancer. Inclusion criteria consisted of men over the age of 50 years, who had one or more previous negative biopsies and were recommended for a repeat biopsy.17 The study excluded patients with a history of prostate cancer and patients taking medications that might influence PSA levels. Drugs that affect PSA levels are found in TABLE 2.

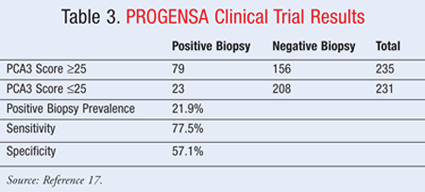
The pivotal clinical study was a prospective, multicenter trial from 14 community-based urology clinics. Data collected included patient age, DRE result, race, family history, serum PSA level, and number of previous negative biopsies (TABLE 3).17 In the study, there were a total of 495 eligible patients; however, 466 patients had a valid PCA3 score and available biopsy results. Of those patients the median age was 67 years; 77% had no family history of prostate cancer, while 102/466 patients had a positive repeat biopsy during the study. When testing the specificity, precision, and sensitivity of the product, tests were conducted using female urine as a control. The authors concluded that the test was helpful in making informed decisions about repeat biopsies for patients. Importantly, the negative predictive value was 90%, meaning that a PCA3 score <25 correctly predicted a negative repeat biopsy 90% of the time.17
Conclusion
Men should discuss with their clinician the benefits and risks associated with prostate screenings and the need for repeat biopsies. Once a decision has been made to move forward with screenings, the PROGENSA PCA3 Assay is a practical choice because it helps to minimize associated risks. More information about the PCA3 Assay may be found at www.gen-probe.com/ products-services/progensa-pca3.
REFERENCES
1. Prostate cancer introduction. Health
Central.
www.healthcentral.com/prostate/prostate-cancer-introduction-000033_1-145.html.
Accessed October 28, 2012.
2. Miller DC, Hafez KS, Stewart A, et
al. Prostate carcinoma presentation, diagnosis, and staging: an update
from the National Cancer Data Base. Cancer. 2003;98(6):1169-1178.
3. What are the key statistics about
prostate cancer?
www.cancer.org/cancer/prostatecancer/detailedguide/prostate-cancer-key-statistics.
Accessed October 31, 2012.
4. Kader K. Genetic test may reduce need
for repeat biopsy for prostate cancer.
http://health.ucsd.edu/news/releases/Pages/2012-07-17-genetic-test-may-predict-prostate-cancer.aspx. Accessed October 30, 2012.
5. DiPiro JT. Pharmacotherapy: A Pathophysiologic Approach. New York, NY: McGraw-Hill Medical; 2011.
6. Busby JE. Determining variables for
repeat prostate biopsy. Nature Publishing Group.
www.nature.com/pcan/journal/v7/n2/full/4500708a.html. Accessed October
25, 2012.
7. Diaz G. Prostate biopsy: 18 reasons to avoid it. www.drgdiaz.com/prostate/prostatebiopsy.shtml. Accessed October 29, 2012.
8. Mayo Clinic. What causes men pain in
prostate biopsy and best method to alleviate it. Medical News Today.
MediLexicon International. www.medicalnewstoday.com/releases/52060.php.
Accessed October 25, 2012.
9. Urological Sciences Research
Foundation. PCA3: a genetic marker of prostate cancer.
www.usrf.org/news/PCA3/PCA3.html. Accessed October 30, 2012.
10. FDA approves PCA3 gene test to aid
decisions about repeat prostate biopsies.
http://labtestsonline.org/news/120323pca3. Accessed October 25, 2012.
11. PCA3 utility. PCA3.org. www.pca3.org/pro/pca3/pca3-utility-0. Accessed October 29, 2012.
12. FDA. Medical devices. PROGENSA® PCA3 Assay. www.fda.gov/MedicalDevices/
ProductsandMedicalProcedures/Device
ApprovalsandClearances/Recently-Approved
Devices/ucm294907.htm. Accessed October 21, 2012.
13. Hologic-Genprobe. www.gen-probe.com. Accessed October 21, 2012.
14. Physician Brochure for the PROGENSA
PCA3 Assay. Gen-Probe. www.gen-probe.com/pdfs/pi/L692-MKT.pdf. Accessed
October 26, 2012.
15. PCA3 Assay, prostate biopsies,
PROGENSA, prostate cancer urine test, repeat prostate biopsies,
urine-based molecular test, positive biopsy, negative biopsy, prostate
cancer diagnosis, cancerous prostate tissue, PCA3, urology, atypical
small acinar proliferation, ASAP.
www.gen-probe.com/products-services/progensa-pca3. Accessed October 24,
2012.
16. PCA3 score. www.windsorurology.co.uk/Clinical_Information/PCA3_score.aspx. Accessed October 26, 2012.
17. PROGENSA PCA3 Assay for in Vitro
Diagnostic Use. Gen-Probe.
www.gen-probe.com/pdfs/pi/502083-EN-RevB.pdf. Accessed October 28, 2012.
To comment on this article, contact rdavidson@uspharmacist.com.





