US Pharm. 2013;38(2):HS2-HS8.
ABSTRACT: Acute decompensated heart failure (ADHF), an exacerbation of chronic cardiac, pulmonary, and/or renal dysfunction, accounts for most of the $39 billion spent on chronic heart failure. The pharmacotherapy of ADHF has not changed recently, but studies have indicated that most therapy is efficaciously, though not economically, equivalent. Evidence-based, inpatient pharmacotherapy is guided by understanding of the Forrester Hemodynamic subsets and their pathophysiology. Clinical success is dependent not only upon proper inpatient treatment, but also upon the utilization of a discharge team. Pharmacists can bridge the gap between hospital visits and outpatient care by recommending and providing continuity of care.
In 2010, the cost of treating heart failure (HF) in the United States was estimated at $39.2 billion.1,2 Nearly 1 million patients are hospitalized each year for exacerbations, and HF currently ranks as the most common reason for hospital admission in adults over 65 years of age.2 Acute decompensated heart failure (ADHF) can result from poorly controlled, chronic HF and/or cardiac, pulmonary, or renal dysfunction. The inpatient cost of care for these patients is extremely high, accounting for about 60% of total HF expenditures.3
In addition to the financial impact of HF, longevity and quality of life are severely impacted in patients with acute exacerbations. Hospital admission is associated with an increased risk of mortality in HF patients; mortality occurs in 15.5% of patients, and one-third of patients are never discharged from the hospital.2 ADHF is a crippling disease burdening not just patients, but also the health care system.
ADHF pharmacotherapy is a battleground for hospitals seeking to control costs in an era of reduced payments and increased expenses. In October 2012, the Centers for Medicare and Medicaid (CMS) began cutting reimbursements to hospitals with high rates of patient readmissions in three high-cost disease states: myocardial infarction (MI), pneumonia, and HF. If patients with these conditions are readmitted within 30 days, CMS will reduce total base reimbursement by 1% annually, until a maximum 3% reduction by 2014.4 Soon, financial loss will motivate the nearly 2,000 hospitals suffering the highest penalties to institute new policies and follow-up care for patients discharged with a diagnosis of HF.
Fortunately, a 2011 analysis by CMS showed that the rate of HF admissions had actually decreased over a 10-year period.1 In 2008, there were 229,000 fewer admissions than expected given observed trends over the past several decades.1,5 Researchers attributed this decline to “reductions in the incidence of coronary artery disease, improved control of blood pressure, increased use of evidence-based therapies, and possibly changes in admission thresholds.” The rate of 1-year all-cause mortality, however, remained high at approximately 30% for patients hospitalized due to HF.1,5 Black males and certain regions in the country fared worse in rates of hospitalization; researchers hypothesized that differences in accessibility or quality of care could contribute to this disparity.1 The best outcomes (reduced costs and readmission rates) are seen with a multidisciplinary approach that includes physicians, pharmacists, nurses, nutritionists, and physical therapists.6 This article is a brief review of ADHF pharmacotherapy with an additional look at pharmacoeconomic considerations and discharge controversies.
Pathophysiology
ADHF is defined as a sudden worsening of HF symptoms and is usually caused by cardiogenic pulmonary edema with rapid fluid accumulation in the lungs, although it can occur without pulmonary edema.7 Hypertension, ischemia, and/or ventricular dysfunction causes a decrease in cardiac output, which leads to an activation of the neurohormonal pathway. The sympathetic system increases norepinephrine to improve peripheral perfusion via vasoconstriction and activates the renin-angiotensin-aldosterone system to increase renal perfusion through water retention.8 An acute increase in left ventricular filling pressure causes protein-poor fluid to leak into the lung alveoli and interstitium, but no compromise of pulmonary membrane integrity occurs. Compensatory mechanisms increase heart rate and systemic vascular resistance in an attempt to improve cardiac output, and a vicious cycle ensues.7,8
Common causes of ADHF include left ventricular or diastolic dysfunction with or without coronary artery disease (CAD) or valvular abnormalities. Although most patients hospitalized with ADHF have a worsening of preexisting HF, up to 20% of patients have no prior diagnosis of HF.9
ADHF can also occur in patients without any preexisting cardiac disease, including conditions such as severe hypertension, fluid overload, severe renal disease, or renal artery stenosis. Factors precipitating an event involve a change in the flow of blood through the heart; ADHF can be induced by hypertensive crisis, MI or ischemia, atrial obstruction, acute mitral regurgitation, fluid overload, or nonadherence to HF medications. Certain medications, such as beta-blockers, nondihydropyridine calcium channel blockers, and nonsteroidal anti-inflammatory drugs (NSAIDs), can also precipitate ADHF.7,10

Classification
ADHF can be categorized into hemodynamic stages depending on cardiac index and pulmonary capillary wedge pressure. The stages are classified as the Forrester Hemodynamic subsets (TABLE 1).11 Cardiac index (CI) indicates the degree of perfusion; patients are characterized as either “warm” or “cold” depending on the presence of hypoperfusion. A CI of <2.2 L/min/m2 warrants a classification of “cold,” indicating hypoperfusion. Signs and symptoms consistent with hypoperfusion include fatigue, hypotension, cool extremities, decreased renal function, and altered mental status. Pulmonary capillary wedge pressure (PCWP) indicates fluid status; patients are classified as either “dry” or “wet” depending on the presence of edema, which is defined as a PCWP >18 mmHg. Signs and symptoms of volume overload include cough, dyspnea, paroxysmal nocturnal dyspnea, elevated jugular venous pressure, peripheral edema, ascites, hepatomegaly, and splenomegaly.
The most common presentation, constituting two-thirds of ADHF admissions, is subset II (warm and wet).8 In comparison to subset I (warm and dry), subset IV (cold and wet) increases the rate of death by two and one-half times.12 Three key laboratory parameters indicate an increased risk of inpatient mortality according to the Acute Decompensated Heart Failure National Registry (ADHERE) trial: a blood urea nitrogen of ≥43 g/dL, systolic blood pressure <115 mmHg, and/or serum creatinine >2.75 mg/dL.13
Pharmacotherapy
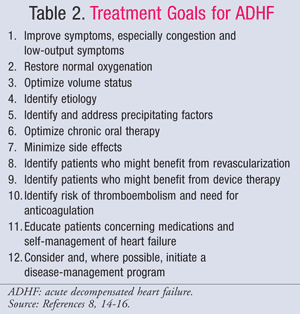
Pharmacologic management of ADHF is guided by the patient’s hemodynamic status, which is evaluated using CI and PCWP values (TABLE 1).11 The goals and medications for treatment of ADHF are summarized in TABLES 2 and 3.8,14-16 While the standard of care has not changed significantly over the past several years, a new focus on the pharmacoeconomic impact of HF has shown that not all treatments are equal. Pharmacotherapy should be chosen based on the diagnosed subset, either treating fluid overload or hypoperfusion. Patients in subset IV are classified as having both fluid overload and perfusion dysfunction and, therefore, require therapy for both symptoms.
Subset II or IV—Wet: Intravenous (IV) loop diuretics are the mainstay of ADHF treatment for patients presenting with fluid overload (PCWP >18 mmHg), despite conflicting data regarding mortality and morbidity benefit. A meta-analysis of 14 small trials showed decreased mortality and hospitalizations in diuretic-treated patients17; however, a recent analysis of observational data from the ADHERE trial showed that hospitalized patients treated with IV diuretics had a longer overall length of stay and a longer ICU length of stay.18 Regardless, the 2009 focused update of the American College of Cardiology/American Heart Association (ACC/AHA) guidelines for treatment of HF recommends early administration of IV diuretics.14 Bolus doses of loop diuretics are preferred. Furosemide is most commonly used; however, bumetanide and torsemide are equally efficacious. If patients have inadequate response to the maximum recommended bolus dose of a loop diuretic, continuous infusion of furosemide may be started at 5 mg/h and titrated to 20 mg/h as needed.14
The superiority of administering furosemide as continuous infusion has yet to be established; the Diuretic Optimization Strategies Evaluation (DOSE) trial, which studied 308 hospitalized patients randomized to receive either bolus or IV furosemide, did not show a significant difference between safety and efficacy endpoints, but smaller studies have indicated a small increase in urinary output with continuous infusion.19 The unmanaged effects of rapid diuresis, such as electrolyte imbalances and renal dysfunction, may lead to prolonged hospitalization beyond the resolution of ADHF symptoms, so electrolytes and serum creatinine should be monitored frequently.
Another strategy to increase diuresis is to add a second diuretic with a different mechanism of action such as metolazone or spironolactone. Metolazone, a thiazide-like diuretic, is utilized most frequently for acute volume control; however, spironolactone and other aldosterone antagonists are preferred in chronic HF therapy.20-22
In patients with continued hypertension and/or persistent symptoms despite aggressive diuresis, a vasodilator may be added to reduce pulmonary congestion. Sodium nitroprusside, nesiritide, and IV nitroglycerin are venous vasodilators that quickly relieve congestive symptoms by reducing PCWP. If a patient presents with low cardiac output and high systemic vascular resistance, venous vasodilators or inotropes are recommended, although vasodilators are the preferred therapy. Due to the risk of cyanide toxicity from prolonged, high-dose infusions of nitroprusside, the preferred vasodilators are nesiritide and nitroglycerin. Of the two, nesiritide produced a greater improvement in global clinical status over nitroglycerin in the Vasodilation in the Management of Acute Congestive Heart Failure (VMAC) trial despite the drug’s propensity to cause hypotension.13 Therefore, nesiritide is the preferred agent in a patient with fluid overload and low cardiac output.
The utility of nesiritide has been questioned because of its high acquisition cost.23 Clinical outcomes data are lacking, but one pharmacoeconomic model shows that the high initial cost of nesiritide is offset by the lower rate of hospital readmission within 21 days when compared to patients who received dobutamine.24 Similarly, a retrospective analysis of ADHERE data showed that patients treated with nitroglycerin or nesiritide were discharged from the hospital sooner than patients treated with dobutamine or milrinone and also had a lower mortality risk.13 Since more critically ill patients were treated with inotropes, it is difficult to attribute the incidence of increased mortality to the drug.
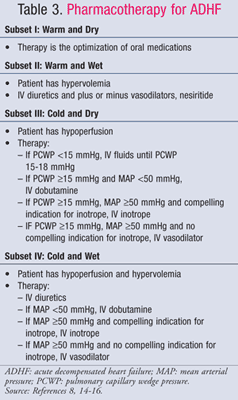
Subset III or IV—Cold: Dobutamine and milrinone are the inotropes of choice to increase perfusion and preserve end-organ perfusion in patients with decreased cardiac output. Compelling indications for inotropes include systolic blood pressure <90 mmHg, symptomatic hypotension despite adequate filling pressures (PCWP 15-18 mmHg), worsening renal function, or inadequate response to IV vasodilators. Prudent selection of patients is necessary because data suggest poor outcomes occur when inotropes are given to patients with preserved cardiac output. The OPTIME-CHF trial showed a nonsignificant increase in mortality and arrhythmias when milrinone infusions were given to patients as a part of standard care.25 Milrinone should be chosen to treat patients receiving concomitant beta-blockers since it exerts an effect on cardiac output and systemic vascular resistance via phosphodiesterase inhibition, whereas dobutamine is a beta1 agonist. Dobutamine, however, is the best choice for hypotensive patients, as milrinone can cause significant drops in blood pressure.15
Discharge Planning
The final step in the treatment of ADHF is discharge planning and follow-up. Both the ACC/AHA and the Heart Failure Society of America (HFSA) guidelines advocate the importance of proper discharge planning.14,15 Mainstays of evidence-based discharge care include treating exacerbating factors, such as discontinuing nondihydropyridine calcium channel blockers or NSAIDs, achieving optimal volume state, optimizing pharmacologic therapy, and transitioning to outpatient oral therapy (TABLE 4).15 The ACC/AHA guidelines recommend giving written instructions or educational material to patients and/or caregivers at discharge addressing the following: activity level, diet, discharge medications, follow-up appointment, weight monitoring, and what to do if symptoms worsen.14 Guidelines also indicate the benefit of initiating mortality-lowering oral therapy for HF during hospital admission, such as angiotensin-converting enzyme (ACE) inhibitors and beta-blockers. Studies show that guideline-based care decreases mortality,26 but fails to show reduction in readmission rates for patients with HF.27,28

Discharge support, or hospital-based case management, is now being utilized for discharge planning. Discharge support is a multidisciplinary, team-based approach to care that deals with medication support, patient education, follow-up planning, and coordination of information. A meta-analysis of 18 randomized, controlled trials (RCT) analyzing discharge planning with postdischarge support involving 3,304 patients found a significant reduction in all-cause readmissions (relative risk [RR] 0.75, 95% CI 0.68-0.88).29 The external validity of the meta-analysis is difficult to determine but may offer some cost-effective strategies. One strategy is from an RCT of 223 HF patients that compared traditional discharge support with educational materials to a 1-hour, one-on-one teaching session with a nurse educator prior to discharge.30 The nurse intervention group had lower risks of rehospitalization or death 180 days after discharge and was estimated to save $2,823 per patient. Conversely, large-scale observational studies have found no relationship between patients receiving educational materials and decreased readmission rates.26,31 Current ACC/AHA guidelines do not explicitly state how to discharge patients after ADHF other than by giving educational materials.14 Guideline recommendations may not provide discharge procedures that help hospitals comply with CMS expectations.
While educational materials at discharge did not show a mortality benefit, each additional oral agent used for the treatment of HF, such as a beta-blocker or aldosterone antagonist, can decrease hospitalizations and/or mortality by 25% to 35%. ACC/AHA guidelines suggest that many HF patients are discharged before optimal volume status is achieved or sent home without the benefit of life saving therapies, which perpetuates the quick readmission of such patients.14 A hospital pharmacist should be an advocate for patients in the discharge support team by assisting with medication reconciliation, discontinuing contraindicated medications, and recommending evidence-based HF pharmacotherapy. Medications should be initiated as soon as possible, ideally in the hospital setting; the initiation of these drugs may exacerbate HF symptoms precipitating ADHF, which further highlights the importance of timely follow-up for this patient population.
Conclusion
ADHF is a burden to patients, health care providers, and payers. The pharmacotherapy for ADHF has not changed recently, but the utilization of discharge support teams and rapid follow-up after discharge provide opportunities for pharmacist intervention, both in and out of the hospital. Community pharmacists can play a large role in the continuity of care, helping transition patients from hospital to primary care. Pharmacists are poised to offer proper guideline-based recommendations, assist with medication reconciliation, and provide discharge education. With the recent changes in CMS reimbursement, pharmacists need to take a larger role in the treatment of this chronic disease.
REFERENCES
1. Winslow R, Wang SS. Heart failure puts fewer in the hospital. Wall Street Journal. October 19, 2011. http://online.wsj.com/article/SB10001424052970203658804576639360226103584.html. Accessed September 1, 2012.
2. Lloyd-Jones D, Adams RJ, Brown TM, et al. Heart disease
and stroke statistics—2010 update. A report from the American Heart
Association. Circulation. 2010;121:e46–e215.
3. Michota FA Jr, Amin A. Bridging the gap between evidence and practice in acute decompensated heart failure management. J Hosp Med. 2008;3(suppl):S7-S15.
4. Rau J. Medicare to penalize 2,217 hospitals for excess readmissions. Kaiser Health News.
August 13, 2012.
www.kaiserhealthnews.org/Stories/2012/August/13/medicare-hospitals-readmissions-penalties.aspx.
Accessed September 1, 2012.
5. Chen J, Normand ST, Wang Y, Krumholz HM. National and
regional trends in heart failure hospitalization and mortality rates for
Medicare beneficiaries, 1998-2008. JAMA. 2011;306:1669-1678.
6. Amin A. Who is managing acute decompensated heart failure? The need for a multidisciplinary approach. J Hosp Med. 2008;6(suppl):S1-S6.
7. Pinto DS, Lewis S. Pathophysiology of acute decompensated heart failure. In: Basow DS, ed. UpToDate. Waltham, MA: UpToDate; 2012.
8. Crouch MA, Didomenico RJ, Rodgers JE. Applying
consensus guidelines in the management of acute decompensated heart
failure. ACPE Program #204-000-06-407-H01. Bethesda, MD: ASHP Advantage;
2007.
9. Joseph SM, Cedars AM, Ewald GA, et al. Acute decompensated heart failure: contemporary medical management. Tex Heart Inst J. 2009;36:510-520.
10. Pinto DS, Lewis S. Evaluation of acute decompensated heart failure. In: Basow DS, ed. UpToDate. Waltham, MA: UpToDate; 2012.
11. Forrester JS, Diamond G, Chatterjee K, et al. Medical
therapy of acute myocardial infarction by application of hemodynamic
subsets. N Engl J Med. 1976;295:1356-1362.
12. Nohria A, Mielniczuk LM, Stevenson LW. Evaluation and monitoring of patients with acute heart failure syndromes. Am J Cardiol. 2005;96:32G-40G.
13. Abraham WT, Adams KF, Fonarow GC, et al. In-hospital
mortality in patients with acute decompensated heart failure requiring
intravenous vasoactive medications: an analysis from the Acute
Decompensated Heart Failure National Registry (ADHERE). J Am Coll Cardiol. 2005;46:57-64.
14. Hunt SA, Abraham WT, Chin MH, et al. 2009 focused
update incorporated into the ACC/AHA 2005 guidelines for the diagnosis
and management of heart failure in adults: a report of the American
College of Cardiology Foundation/American Heart Association Task Force
on Practice Guidelines. Circulation. 2009;119;e391-e479.
15. Heart Failure Society of America. Evaluation and
management of patients with acute decompensated heart failure: HFSA 2010
comprehensive heart failure practice guideline. J Card Fail. 2010;16:e134-e156.
16. Colucci W. Treatment of acute decompensated heart failure: Components of therapy. In: Basow DS, ed. UpToDate. Waltham, MA: UpToDate; 2012.
17. Faris R, Flather MD, Purcell H, et al. Diuretics for heart failure. Cochrane Database Syst Rev. 2012;(1):CD003838.
18. Gheorghiade M, Filippatos G. Reassessing treatment of acute heart failure syndromes: the ADHERE registry. Eur Heart J. 2005;7:813-819.
19. Felker GM, Lee KL, Bull DA, et al. Diuretic strategies in patients with acute decompensated heart failure. N Engl J Med. 2011;364:797-805.
20. Effectiveness of spironolactone added to an
angiotensin-converting enzyme inhibitor and a loop diuretic for severe
chronic congestive heart failure (the Randomized Aldactone Evaluation
Study [RALES]). Am J Cardiol. 1996;78:902-907.
21. Pitt B, Remme W, Zannad F, et al. Eplerenone, a
selective aldosterone blocker, in patients with left ventricular
dysfunction after myocardial infarction. N Engl J Med. 2003;348:1309-1321.
22. Zannad F, McMurray JJ, Krum H, et al; EMPHASIS-HF
Study Group. Eplerenone in patients with systolic heart failure and mild
symptoms. N Engl J Med. 2011;364:11-21.
23. Gerhard T, Zineh I, Winterstein AG, Hartzema AG.
Pharmacoeconomic modeling of nesiritide versus dobutamine for
decompensated heart failure. Pharmacotherapy. 2006;26:34-43.
24. Lissovoy G, Stier DM, Ciesla G, et al. Economic
implications of nesiritide versus dobutamine in the treatment of
patients with acutely decompensated congestive heart failure. Am J Cardiol. 2003;92:631-633.
25. Cuffe MS, Califf RM, Adams KF Jr, et al. Short-term
intravenous milrinone for acute exacerbation of heart failure: a
randomized controlled trial. JAMA. 2002;287:1541-1547.
26. Kfoury AG, French TK, Horne BD, et al. Incremental
survival benefit with adherence to standardized heart failure core
measures: a performance evaluation study of 2958 patients. J Card Fail. 2008;14:95-102.
27. Patterson ME, Hernandez AF, Hammill BG, et al. Process
of care performance measures and long-term outcomes in patients
hospitalized with heart failure. Med Care. 2010;48:210.
28. Fonarow GC, Abraham WT, Albert NM, et al. Association
between performance measures and clinical outcomes for patients
hospitalized with heart failure. JAMA. 2007;297:61-70.
29. Phillips CO, Wright SM, Kern DE, et al. Comprehensive
discharge planning with postdischarge support for older patients with
congestive heart failure: a meta-analysis. JAMA. 2004;291:1358-1367.
30. Koelling TM, Johnson ML, Cody RJ, Aaronson KD.
Discharge education improves clinical outcomes in patients with chronic
heart failure. Circulation. 2005;111:179-185.
31. Jha AK, Orav EJ, Epstein AM. Public reporting of discharge planning and rates of readmissions. N Engl J Med. 2009;361:2637-2645.
To comment on this article, contact rdavidson@uspharmacist.com.





